Manuel Bayon-Calatayud, MD, Ana Maria Benavente-Valdepeñas, MD and Maria del Prado Vazquez-Muñoz, OT
From the Department of Physical and Rehabilitation Medicine, Hospital Virgen de la Salud, Servicio de Salud de Castilla-La Mancha, SESCAM, Toledo, Spain
OBJECTIVE: To investigate the efficacy of mirror therapy in reducing pain and disability in patients with distal radial fractures.
DESIGN: Pilot randomized controlled study.
SUBJECTS: Twenty-two patients with closed distal radial fracture.
METHODS: Patients were randomly assigned to experimental (n = 11) or control (n = 11) groups. Researchers were blinded to group allocation. Both groups received conventional physiotherapy. In addition, the experimental group had 15 sessions of mirror therapy (a daily session, 30 min). The control group received the same amount of conventional occupational therapy. Assessment was made from baseline to post-treatment. Pain was measured on a visual analogue scale (VAS). Active wrist extension and Quick-DASH (Disabilities of Arm, Shoulder and Hand) were used to assess functional recovery.
RESULTS: Pain, disability, and range of motion improved for both groups after intervention. No significant post-treatment differences were found between groups in Quick-DASH (p = 0.409), active wrist extension (p = 0.191) and VAS scores (p = 0.807).
CONCLUSION: There was no significant difference in active wrist extension between groups. Mirror therapy was not superior to conventional occupational therapy in reducing pain and disability.
Key words: distal radius; fracture; mirror therapy; rehabilitation.
J Rehabil Med 2016; 48: 00–00
Correspondence address: Manuel Bayon-Calatayud, Department of Physical and Rehabilitation Medicine, Hospital Virgen de la Salud, Avenida de Barber, 30, ES-45004-Toledo, Spain. E-mail: mrbayon@sescam.jccm.es
Accepted Jun 21, 2016; Epub ahead of print Aug 26, 2016
INTRODUCTION
Distal radial fracture is one of the most common orthopaedic injuries, accounting for up to 15% of all extremity fractures (1). Its annual incidence has been estimated approximately 105/100,000 in men, and 416/100,000 in women, with a 4:1 women/men ratio (2). It is particularly predominant in older populations who have an increased number of falls and a greater bone loss due to osteoporosis.
These fractures are also important because they can be associated with a high incidence of serious complications (complex regional pain syndrome (CRPS), peripheral neuropathies, tendon and ligament injuries) (3–6). Rehabilitation is crucial to diminish pain and speed up functional recovery. However, in many cases, despite appropriate management, patients have persistent pain and functional impairment. There is currently insufficient evidence to determine the best form of rehabilitation to reduce pain and achieve better functional outcomes (7).
Mirror therapy (MT) is a simple inexpensive rehabilitation technique in which a mirror is positioned between the unaffected and affected limbs, blocking the patient’s view of the affected limb. The patient performs exercises using both limbs, and the reflection of the intact limb movement in the mirror creates a visual illusion of enhanced movement capability in the affected limb.
MT was first introduced by Ramachandran & Hirstein to treat phantom limb pain after amputation (8). Since then it has also been applied to treat CRPS (9) and upper limb motor impairment in stroke patients (10–13). However, there is little published research on the application of MT in rehabilitation for orthopaedic disorders. MT has been used after median nerve repair and hand surgery, allowing patients to recover hand coordination, grip strength and active finger flexion (14). However, few studies have been addressed to research whether the application of a MT programme could be useful in patients with distal radial fracture.
The hypothesis on which the current study was based is that mirror visual feedback (MVF) may contribute to improved wrist and hand motor function. This may occur through activation of the superior temporal gyrus, premotor cortex, mirror neurone system, and areas involved with allocation of attention and cognitive control (dorsolateral prefrontal cortex, posterior cingulate cortex, S1 and S2, precuneus) (15).
The aim of this study was to investigate whether an early adjuvant MT programme, added to usual rehabilitation care, is effective in order to increase range of motion (active wrist extension), reduce pain and enhance functional recovery for patients with distal radial fracture.
METHODS
Study participants
This randomized controlled study included 22 participants (7 men and 15 women). Inclusion criteria were: patients with a closed distal radial fracture who followed operative or conservative treatment. Exclusion criteria were: patients with unstable medical conditions, upper limb paresis, apraxia, hemi-neglect, peripheral nerve injuries, cognitive impairment (Spanish version of Mini-Mental State Examination score MMES < 24), open fracture, or other concomitant upper limb orthopaedic disorders. Patients who were participating in other rehabilitation studies were also excluded.
At the beginning of the study 24 patients were assessed as meeting the inclusion criteria, but 2 moved to another hospital and were excluded from the study.
Patients recruited for this study were referred from Toledo, Spain for outpatient rehabilitation, from September 2014 to February 2015. Two physical and rehabilitation medicine specialists determined the participants’ eligibility and collected written informed consent.
The aim of this study, the effects and rules of MT were explained to all participants, who read an information form and signed corresponding written informed consent.
Intervention and assessment
After concluding baseline measurements, patients were randomly assigned (allocation ratio 1:1) to either MT group (n = 11) or control group (n = 11), using a computer-generated random number list. Investigators were blinded to the allocation group. Patients and occupational therapists were not blinded. A physical and rehabilitation medicine specialist who was blinded to the study protocol and not otherwise involved in the trial operated the random number programme.
Participants from the MT group were seated close to a table in which a mirror box (60 × 45 × 25 cm size) was positioned vertically. The affected hand was placed into the mirror box and the unaffected hand was placed in front of the reflective mirror surface. According to this procedure, patients from the MT group practiced active mobilization of the wrist and fingers, grip and grasp training, and task-oriented exercises, during a daily session of 30 min, 5 days a week, for 3 weeks; a total of 15 sessions of treatment. During this procedure the reflection of the movement of the unaffected wrist is perceived as movement of the fractured limb.
Instead of a MT programme, participants from the control group received an equivalent time and intensity of a conventional occupational therapy programme, consisting of the same treatment protocol described above, but without mirror intervention.
In addition, patients from both groups carried out a 30 min conventional physiotherapy programme, consisting of active and passive mobilization, muscle strengthening exercises, and physical methods to relieve pain (transcutaneous electrical nerve stimulation (TENS), ultrasound, heat and cold therapies). The experimental procedure for the present study is shown in Fig. 1.
Fig. 1. Experimental procedure.
Outcomes were measured in terms of pain, disability and recovery of active range of motion, before and after concluding 15 sessions of treatment. Pain was assessed with a visual analogue scale (VAS) with a 10-cm line anchored with words descriptive of the maximal and minimal pain.
Disability was measured using Quick-DASH (Disabilities of Arm, Shoulder and Hand) Spanish version, an 11-ítem questionnaire that measures the severity of symptoms and individual ability to complete a variety of functional tasks. The score ranges from 0 (no disability) to 100 (greatest disability). Finally, wrist range of motion evaluation is represented by active wrist extension, measured using a handheld goniometer.
Sample size and statistical analysis
Power calculations indicated that detecting a 60% difference between groups regarding increase in active wrist extension from baseline to post-treatment (with β = 0.20, and α = 0.05), would require a sample size of 11 subjects for each group.
To compare differences between groups after intervention a non-parametric test was used (Mann–Whitney U test). Data are presented as means (with standard deviation) and medians (with interquartile ranges). Statistical significance was accepted for p-values less than 0.05. Statistical analysis was performed using SPSS 17.0 programme, and there were no missing data.
RESULTS
The demographic and clinical characteristics of participants are shown in Table I.
|
Table I. Characteristics of study participants |
||
|
Characteristics |
Mirror group |
Control group |
|
Total, male/female, n |
11 (3/8) |
11 (4/7) |
|
Age, years, mean (SD) [range] |
61.09 (13.05) [34–78] |
55.36 (18.28) [26–85] |
|
Intra-articular fracture, n |
5/11 |
4/11 |
|
Extra-articular fracture, n |
6/11 |
7/11 |
|
Surgical/conservative treatment, n |
4/7 |
4/7 |
|
Complications, n |
||
|
CRPS |
3/11 |
3/11 |
|
Paresthesias |
5/11 |
6/11 |
|
Comorbidity, n |
||
|
Osteoporosis |
5/11 |
2/11 |
|
SD: standard deviation; CRPS: complex regional pain syndrome. |
||
Active wrist extension improved after intervention for both groups.
The mean and standard deviation (SD) for post-treatment change in active wrist extension for the mirror group was 17° (SD 7°), whereas for the control group it was 13° (SD 11°). Median values with interquartile ranges were 15.0° (IQR 10.0°, 20.0°) for the mirror group, and 10.0° (IQR 5.0°, 15.0°) for the control group.
The Quick-DASH median with interquartile ranges (IQR) at baseline was 56.81 (IQR 31.81, 75.0) for the mirror group, and 54.54 (IQR 52.27, 72.72) for the control group. Mean Quick-DASH change after treatment for the mirror group was –25.6 (SD 16.7) and median –27.27 (IQR –40.91, –6.82), whereas for the control group the mean was –30.57 (SD 7.76) and median was –31.82 (IQR–36.40, –22.72).
Baseline VAS score values for the mirror group were median 5.0 (IQR 4.0, 6.0), whereas for the control group they were 5.0 (IQR 3.0, 7.0). Changes in VAS score after intervention were median –2.0 (IQR –5.0, –1.0) for the MT group, and –2 (IQR –4.0, 0.0) for control group. Comparison between groups of VAS score, Quick-DASH, and wrist extension are shown in Fig. 2.
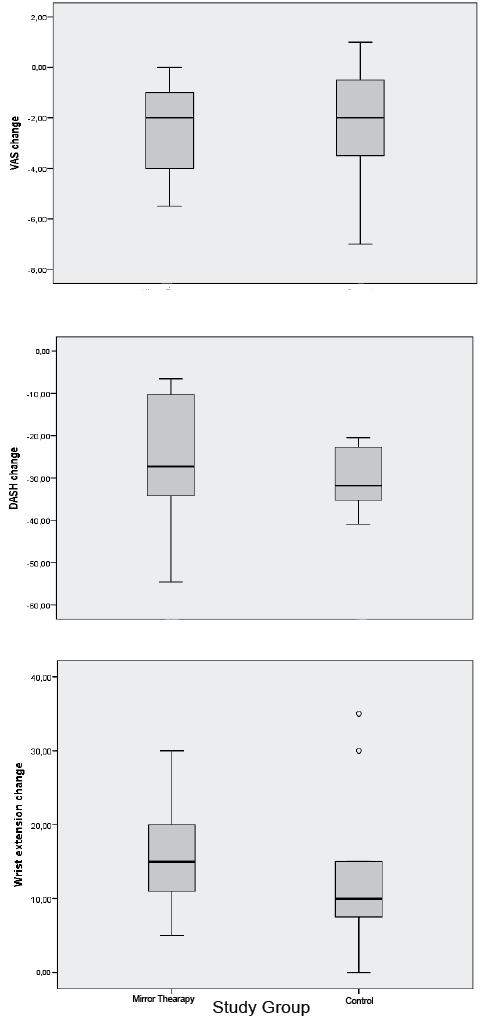
Fig. 2. Comparison between groups of visual analogue scale (VAS), Quick-DASH (Disabilities of Arm, Shoulder and Hand), and wrist extension. Data are presented as median values with interquartile ranges.
According to Mann–Whitney test outcomes, no significant post-treatment differences were found between groups in Quick-DASH (p = 0.409), active wrist extension (p = 0.191) and VAS scores (p = 0.807).
There were 3 cases of CRPS in each group. All participants from the mirror and control CRPS subgroups had improved range of motion and disability scores after intervention. VAS scores were better for the CRPS control subgroup than the MT group. However, due to the small sample size of the CRPS subgroup we cannot draw any significant conclusion.
DISCUSSION
MT could be useful in patients with distal radial fractures for whom active motor rehabilitation may be initially difficult due to pain and mobility restraints.
MT could induce activation of the dorsolateral prefrontal cortex, posterior cingulate cortex, S1, S2, and precuneus, facilitating ipsilateral projections from the contralateral M1 to the affected hand (15). The discharge of these neurones is associated with object-oriented hand actions, such as grasping, holding and manipulating (16, 17).
Moreover, visual illusion during MT could generate positive feedback to the motor cortex, modulating cortical mechanisms of sensation and movement (18, 19). Mirror visual illusion increases primary motor cortex (M1) excitability of the affected wrist behind the mirror (20), and at the same time activates brain areas (precuneus, cingulate cortex) associated with awareness of the affected limb reducing the learned non-use phenomenon (21).
Few studies have been conducted on patients with distal radial fractures who potentially could benefit from MT. In a previous case study a patient with a distal radial fracture was treated with electrical stimulation of wrist extensors added to conventional treatment. Only when a MT programme (including mirror exercises at home) was combined with electrical stimulation of wrist extensors was the patient able to recover active wrist extension (22).
In the present pilot study an early and intensive MT programme was provided in combination with a conventional rehabilitation programme. Participants from both groups improved range of motion, disability and pain scores, but no significant difference was found between groups. MT has previously shown positive results for diminishing pain intensity in patients with CRPS type I, and reducing the learned pain phenomenon (23, 24).
In this study there were the same number of CRPS cases in each group (3 in the experimental group, and 3 in the control group).
Post-treatment active wrist extension, pain, and disability scores were no better for mirror patients compared with controls in the CRPS subgroup. MT might be ineffective in this situation; however, it could be influenced by several factors, including clinical variability in surgical procedures, presence of osteoporosis (1 confirmed case in the MT CRPS subgroup against none in the CRPS control subgroup), moderate paresthesias predominantly in the MT CRPS subgroup (3 cases), and older mean age (55.33 years (SD 22.03) for the MT CRPS group, compared with 48 years (SD 23.64) for the CRPS controls).
Paresthesias were the most frequent complication in this study; half of the total number of participants presented them. Patients with paresthesias from both groups improved active wrist extension after intervention. During MT, viewing the reflection of the unaffected hand touching and manipulating objects induces the patient to do the same things with the affected hand, helping them to overcome the aberrant sensory input from the affected wrist and hand in such way that paresthesias could be reduced.
Outcome variability in the clinical application of MT may depend on the type and characteristics of the fracture, conservative or surgical management, associated comorbidities, such as osteoporosis, MT protocol, and other unknown factors.
The same number of patients within each group followed surgical or conservative treatment. Regardless of fracture management, the MT group showed a non-significant greater increase in active wrist extension than the control group.
Outcomes variability may be also influenced by the characteristics of MT protocols applied. There is currently no agreement about the best protocol for MT in terms of onset, duration, intensity and type of exercise, to obtain best functional outcomes, and the influence of this factor remains unclear.
Adverse effects were clinically infrequent during the application of MT in this study, and only one participant reported occasional feelings of dizziness.
Limitations of this study were the small simple size and the lack of follow-up.
In spite of its small simple size, this pilot study could contribute to calculate sample size for future studies. In this study pre-post-intervention difference between groups regarding active wrist extension is median 5° (IQR 5°, 5°), with a calculated small power ranging between 14% and 59%. An acceptable power (80%) for a study with 22 participants could only be achieved with a pre-post-intervention difference between groups greater than 10º of active wrist extension. Regarding future studies, and taking into account the variability observed in this study, at least 30 participants per group are required to achieve an acceptable power (80%) and to obtain differences between groups in wrist extension of approximately 7°.
Another limitation is the absence of more than immediate post-treatment follow-up. It was not the main purpose of the study to make a follow-up assessment, but an evaluation of MT efficacy at an acute-subacute phase along treatment course. In this way the retention effect of MT remains uncertain; thus future studies should consider including longitudinal follow-up assessment.
In conclusion, the early application of adjuvant MT for rehabilitation of distal radial fractures was not found to be superior to conventional occupational therapy in terms of increasing range of motion, reducing pain and disability. More research is required to confirm the efficacy of MT and to determine the most effective and appropriate protocols to achieve best functional outcomes.
The authors declare no conflicts of interest.
REFERENCES
