Simone Thomas, MSc1,5, Jane H. Burridge, PhD2, Marcus Pohl, MD3, Frank Oehmichen, MD4 and Jan Mehrholz, PhD1,5
From the 1Wissenschaftliches Institut, Private Europäische Medizinische Akademie der Klinik Bavaria in Kreischa, Kreischa, Germany, 2Neurorehabilitation Research Group, Faculty of Health Sciences, University of Southampton, UK, 3Helios Klinik Schloss Pulsnitz, 4Klinik Bavaria in Kreischa, Kreischa and 5Department of Public Health, Medizinische Fakultät‚ Carl Gustav Carus’, Technische Universität Dresden, Germany
OBJECTIVES: To describe the time course of recovery of sit-to-stand function in patients with intensive-care-unit-acquired muscle weakness and the impact of recovery.
METHODS: A cohort study in post-acute intensive care unit and rehabilitation units. Patients with chronic critical illness and intensive-care-unit-acquired muscle weakness were included. Sit-to-stand function was measured daily, using a standardized chair height, defined as 120% of the individual’s knee height.
RESULTS: A total of 150 patients were recruited according to the selection criteria. The primary outcome of independent sit-to-stand function was achieved by a median of 56 days (interquartile range Q1–Q3 = 32–90 days) after rehabilitation onset and a median of 113 days (Q1–Q3=70–148 days) after onset of illness. The final multivariate model for recovery of sit-to-stand function included 3 variables: age (adjusted hazard ratio (HR) = 0.96 (95% CI 0.94–0.99), duration of ventilation (HR=0.99 (95% CI 0.98–1.00) and Functional Status Score for the Intensive Care Unit (FSS-ICU) (adjusted HR=1.12 (95% CI 1.08–1.16)).
CONCLUSION: Rapid recovery of sit-to-stand function for most patients with intensive-care-unit-acquired muscle weakness was seen. The variables older age and longer duration of ventilation decreased, and higher FSS-ICU increased the chance of regaining independent sit-to-stand function.
Key words: intensive care myoapthy; intensive care neuropathy; rehabilitation; sit-to-stand function; muscle weakness.
J Rehabil Med 2016; 48: 00–00
Correspondence address: Jan Mehrholz, Wissenschaftliches Institut, Private Europäische Medizinische Akademie der Klinik Bavaria in Kreischa GmbH, An der Wolfsschlucht 1-2, 01731 Kreischa, Germany. E-mail: jan.mehrholz@klinik-bavaria.de
Accepted Jun 29, 2016; Epub ahead of print Sep 26, 2016
INTRODUCTION
Muscle weakness is common after treatment of critical illness in intensive care units (ICU), with a prevalence rate greater than 25% (1, 2). Such acquired muscle weakness is characterized by profound weakness that is greater than would normally be expected from prolonged bed rest. It is therefore designated as clinically detected weakness in critically ill patients in whom there is no plausible aetiology other than critical illness (2). Acquired weakness of limb muscles significantly limits activities of daily living (ADL) that require fundamental activities such as sit-to stand or standing and walking (3–5), and consequently increases morbidity and delays rehabilitation and recovery from critical illness (6). Although full recovery has been reported in approximately 50% of people with ICU-acquired muscle weakness, improvement is related to the severity of the condition. People with severe weakness may take months to improve, and may even remain severely affected (7, 8). Focused, individual rehabilitation of people with ICU-acquired muscle weakness is therefore important (2, 9), and physical intervention strategies have been described in the literature (2, 8, 10, 11). A major goal of rehabilitation is to mobilize these patients to enable them to improve ADL. An important prerequisite of many ADL, and particularly of those involving walking, is functional sit-to-stand, i.e. getting up from a chair independently to be able to walk (12).
Recent longitudinal studies have described the recovery of critically ill people (13–16), but we are not aware of studies of recovery of sit-to-stand function in patients with chronic ICU-acquired muscle weakness, and the determinants for recovery remain unknown.
Therefore the aim of the General Weakness Syndrome Therapy (GymNAST) study was to describe the time course of sit-to-stand function in patients with ICU-acquired muscle weakness and to develop a multivariate risk factor model for recovery of sit-to-stand function (17).
METHODS
Between January 2013 and March 2015 we screened all patients admitted to our post-acute ICU and recruited those who met the following criteria, as previously described (17):
Inclusion criteria:
• patient is in the post-acute phase and is chronically critically ill, defined as > 21 days ICU treatment including mechanical ventilation and at least 14 more days of ICU treatment (17, 18),
• ICU-acquired muscle weakness, defined as a Medical Research Council (MRC) total score (upper and lower limb) of less than 48 points (2),
• muscle weakness pathology, e.g. a clinical diagnosis of critical illness myopathy (CIM) and critical illness polyneuropathy (CIP) (19),
• age 18 years or over,
• Richmond Agitation Sedation Scale (RASS) score of –1 to 2 (20),
• written informed consent of the patient or his/her legal guardian,
Exclusion criteria:
• patients receiving palliative care,
• co-morbidities of the trunk or lower limbs interfering with upright posture and walking (e.g. amputation or fracture of lower limb),
• other neuromuscular or neurological disease and/or syndromes causing weakness (e.g. Guillain–Barré syndrome, myasthenia gravis, porphyria, Lambert-Eaton syndrome, amyotrophic lateral sclerosis, vasculitic neuropathy, cervical myelopathy and botulism),
• severe physical co-morbidity before becoming critically ill (e.g. frailty due to neurological conditions, and not being able to sit-to-stand or to walk),
From the first day of admission to our post-acute ICU and rehabilitation units all patients (including those on mechanical ventilation) received individual treatment, including physiotherapy and occupational and other appropriate therapies (21). The amount of therapy and methods were dependent on the severity of critical illness and medical indication. However, the start, content and amount of treatment in the early acute stage were not measured.
Measures and outcomes
Sit-to-stand function, the primary outcome of the GymNAST study (17), was defined as one successful, independent sit-to-stand from a standardized chair (chair height 120% of knee height). We followed all patients for 1 year or until sit-to-stand function was reached, whichever was sooner and analysed sit-to-stand function as the first time point when sit-to-stand function was performed successfully (time to event). If a patient was discharged, died, or lost to follow-up we recorded this information (censoring).
We used the following secondary outcomes (21):
• ADL measured with the Barthel Index (BI; 10 items) (22),
• clinical severity (e.g. mechanical ventilation, dysphagia, tracheostomy) measured with the Early Rehabilitation Barthel Index (ERBI) (23),
• muscle strength of the upper limb (shoulder, elbow and wrist) and lower limb (hip, knee and ankle) using the Medical Research Council (MRC) sum score (2),
• summed grip strength of both hands (measured with a dynamometer) (24),
• Functional Status Score for the Intensive Care Unit (FSS-ICU) (25),
• Physical Function ICU Test (scored) (PFIT-s) (26),
• pain using a numeric pain rating scale
• “functional reach” forward as a measure of sitting and standing balance (distance in cm) (27),
• cognitive measures (Montreal Cognitive Assessment (MoCA) (28) and clock drawing test (CDT) (29),
• walking speed (m/s) and distance (in m) walked in 6 min (6-MWT) (6).
All assessments were administered by trained, experienced therapists. The primary outcome was measured daily, and secondary outcomes were measured from baseline (T0) every 2 weeks up to 8 weeks (T4). Baseline was defined as the first admission to our post-acute hospital or to our inpatient rehabilitation centre, respectively (T0). The duration of the primary illness was defined as the time between first admission to the acute hospital due to the onset of primary illness until T0 (baseline) or until the observation of the primary outcome. The duration of study was therefore the time between T0 and observation of the primary outcome.
Ethical considerations
This study was conducted according to the principles of the Declaration of Helsinki, with approval from the local ethics commission (Sächsische Landesärztekammer, EK-BR-32/13-1/106755). The study was registered before publication (German Register of Clinical Trials, DRKS00006528).
Statistical analysis
Descriptive and inference statistics were applied dependent on the type of test and data distribution (30). Global alpha level was set at 0.05.
The probability of regaining sit-to-stand function was calculated using the Kaplan–Meier method (31). Univariate and multivariate Cox regression analysis with a selection of possible predictor variables for the primary outcome was used as described below (32, 33).
Univariate analysis. Univariate Cox regression analysis was performed for the following variables: age at baseline, body mass index (BMI), sex, duration of illness, number of medical tubes (the sum of catheters and vascular access), duration of mechanical ventilation, number (the sum) of secondary diagnoses, and all secondary outcomes.
Multivariate analysis and model building. All statistically significant variables (alpha level of 0.2 for selection) were candidate predictor variables, and those with the highest global χ2 score were selected first and entered into a multivariate regression analysis (32, 33). To remain in the multivariate model a variable had to be significant at the 0.1 level (33). The multivariate models was then compared with the remaining variables on the global score χ2 statistic (best subset selection) and on the Akaike’s information criterion (AIC) and Schwarz’s Bayesian criterion (SBC) for our final multivariate model (32, 33). The effects of the final multivariate model were expressed as hazard ratios (HRs) with 95% confidence intervals (95% CI). SAS/STAT 9.3 was used for all statistical procedures (SAS Institute Inc., Cary, NC, USA) and proportional hazards assumptions were tested with the implemented function.
RESULTS
A total of 150 patients with ICU-acquired muscle weakness were included between January 2013 and March 2015 in our GymNAST study (Fig. 1), as described recently (21). The baseline demographic and clinical characteristics of the patients (at T0) are shown in Table I.
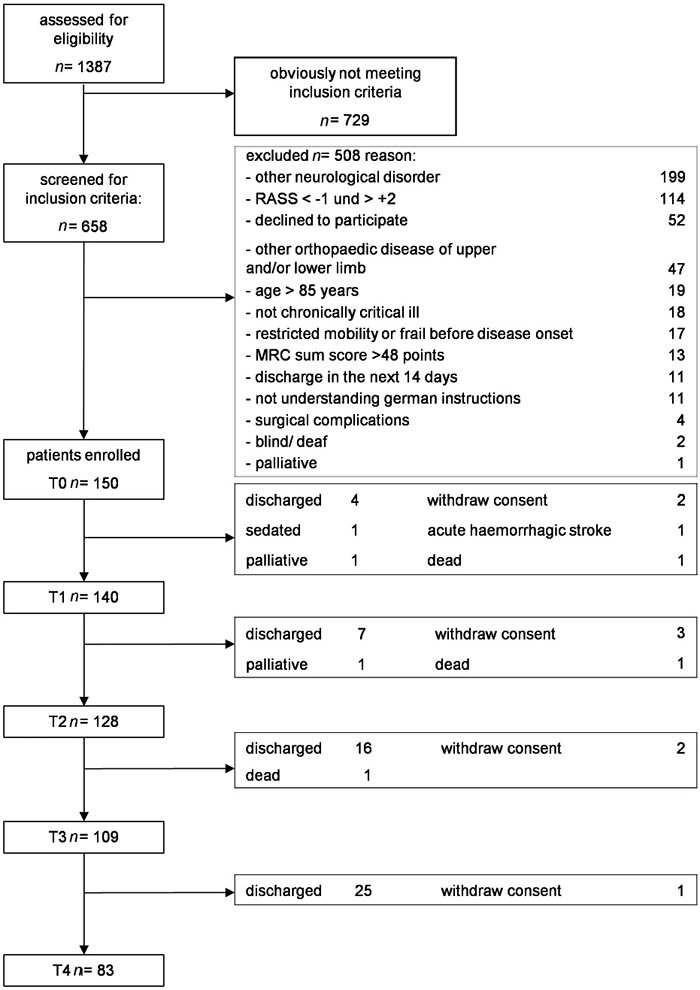
Fig. 1. Study flow-chart.
|
Table I. Patient characteristics (n = 150) |
|||||
|
Variable |
n |
Median (Q1–Q3) |
Mean (SD) |
Patients with high FSS-ICU score (≥ 16 points) Mean (SD) |
Patients with low FSS-ICU score (< 16 points) |
|
Patient demographics |
|||||
|
Age, years |
71 (63–75) |
69.26 (9.0) |
70.1 (7.9) |
67.8 (10.3) |
|
|
Female, n (%) |
50 (30) |
25 (29) |
20 (31) |
||
|
BMI |
27 (25–32) |
29.1 (8.3) |
30.1 (9.4) |
27.8 (6.3) |
|
|
Patients recruited at post-ICU |
121 (81) |
61 (81) |
60 (80) |
||
|
Patients recruited at inpatient rehabilitation centre |
29 (19) |
14 (19) |
15 (20) |
||
|
Duration of illness, days |
41 (30–60) |
49.1 (29.1) |
51.4 (31.3) |
46.1 (25.8) |
|
|
Duration of mechanical ventilation, days |
53 (38–84) |
65.2 (45.1) |
77.9 (49.4) |
54.5 (52.6) |
|
|
Primary ICU diagnosis |
|||||
|
Sepsis |
82 (55) |
40 (53) |
42 (56) |
||
|
Pneumonia |
29 (19) |
15 (20) |
14 (19) |
||
|
Cardiac |
21 (14) |
10 (13) |
11 (15) |
||
|
Other |
18 (12) |
9 (12) |
9 (12) |
||
|
Dialysis |
45 (30) |
24 (32) |
21 (28) |
||
|
Independent variables |
|||||
|
Apache II (range 0–34) |
16 (14–19) |
16.5 (4.1) |
17.3 (4.6) |
15.4 (3.1) |
|
|
Barthel Index (range 0–100) |
5 (0–25) |
14.7 (19.2) |
4.1 (8.0) |
27.5 (21.0) |
|
|
MRC sum score (range 0–30) at baseline, upper limb |
10 (8–12) |
0.5 (0.8) |
8.2 (2.5) |
10.8 (1.9) |
|
|
MRC sum score (range 0–30) at baseline, lower limb |
9 (7–10) |
0.5 (0.8) |
7.0 (2.4) |
9.9 (1.6) |
|
|
MOCA score (range 0–30) at baseline |
16 (10–20) |
14.3 (7.0) |
11.5 (7.9) |
17.0 (4.6) |
|
|
FSS-ICU (range 0–35) at baseline All patients |
16 (8–23) |
15.8 (9.8) |
|||
|
Patients without successful sit-to-stand recovery |
11 (6–17) |
12.2 (8.5) |
|||
|
Patients with successful sit-to-stand recovery |
22 (13–28) |
19.9 (9.6) |
|||
|
ERBI item 1: intensive care supervision |
121 (81) |
76 (89) |
45 (70) |
||
|
ERBI item 2: tracheostomy tube management and supervision |
120 (80) |
76 (89) |
44 (69) |
||
|
ERBI item 3: intermittent (or continuous) mechanical ventilation |
103 (69) |
67 (79) |
36 (56) |
||
|
ERBI item 4: confused patient (in need of supervision) |
3 (2) |
3 (4) |
0 (0) |
||
|
ERBI item 5: behavioural disturbances (patient being a danger to |
8 (5) |
4 (5) |
4 (6) |
||
|
ERBI item 6: severe impairment of communication |
41 (21) |
33 (39) |
8 (13) |
||
|
ERBI item 7: dysphagia patient in need of supervision |
54 (64) |
27 (42) |
|||
|
ERBI: Early Rehabilitation Barthel Index; FSS-ICU: Functional Status Score for the Intensive Care Unit; PFIT-s: Physical Function ICU Test (scored); MRC: Medical Research Council; VAS: visual analogue scale; MoCA: Montreal – Cognitive Assessment; BMI: body mass index. |
|||||
Recovery of sit-to-stand function was achieved after a median of 56 days (interquartile range Q1–Q3 = 32–90 days) after rehabilitation began and after a median of 113 days (Q1–Q3 = 70–148 days) after onset of illness. Sit-to-stand function improved significantly over time (Fig. 2a and 2b). We present the time course of recovery of sit-to-stand function as the time from study onset (Fig. 2a) and as the time after onset of illness (Fig. 2b). We present univariate Cox proportional hazards for regaining sit-to-stand function of all predictor variables in Table II. Our final multivariate adjusted Cox proportional hazards model included: age (adjusted hazard ratio (HR) = 0.96 (95% CI 0.94–0.99), duration of ventilation (HR = 0.99 (95% CI 0.98–1.00), and FSS-ICU (HR) = 1.12 (95% CI 1.08–1.16; see Table III), explaining a total variance for regaining sit-to-stand function of 21%.
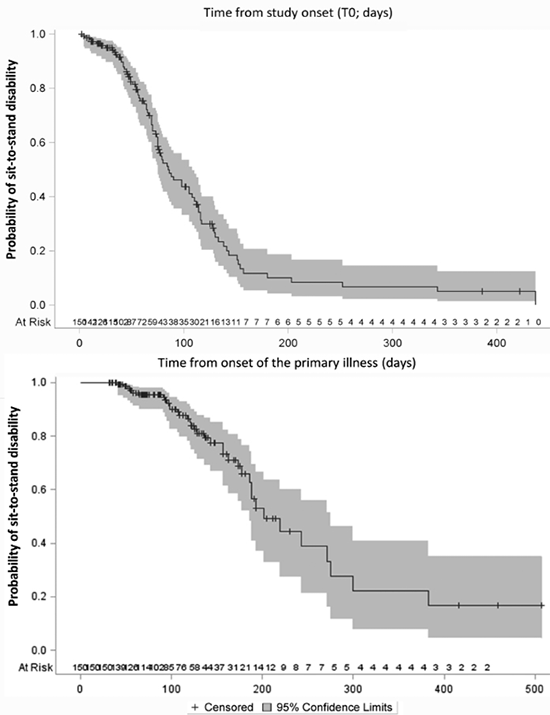
Fig. 2. Time course of recovery of sit-to-stand function from: (a) study onset (T0); and (b) onset of the primary illness in days.
|
Table II. Summary of the univariate Cox proportional hazards for regaining sit-to-stand function of all potential predictor variables |
||||
|
Variable (at T0) |
χ2 |
p-value |
HR |
95% CI |
|
Age, years |
19.43 |
< 0.001 |
0.940 |
0.915–0.966 |
|
Body mass index |
4.44 |
0.035 |
0.961 |
0.927–0.997 |
|
Sex, male |
1.15 |
0.284 |
1.376 |
0.768–2.465 |
|
Duration of illness, days |
1.02 |
0.312 |
0.994 |
0.983–1.006 |
|
Medical tubes (catheters and vascular access), n |
2.35 |
0.126 |
0.858 |
0.706–1.044 |
|
Duration of mechanical ventilation, days |
9.61 |
0.002 |
0.988 |
0.980–0.996 |
|
Secondary diagnosis, n |
0.67 |
0.413 |
0.980 |
0.934–1.028 |
|
ERBI item 1: intensive care supervision |
1.30 |
0.255 |
1.010 |
0.993–1.027 |
|
ERBI item 2: tracheostomy tube management and supervision |
2.18 |
0.140 |
1.012 |
0.996–1.029 |
|
ERBI item 3: intermittent or continuous mechanical ventilation |
0.08 |
0.778 |
0.998 |
0.981–1.015 |
|
ERBI item 4: confused patient (in need of supervision) |
0.00 |
0.983 |
1.359 |
0.01–5.047 |
|
ERBI item 5: behavioural disturbances (patient being a danger to himself or others) |
6.91 |
0.009 |
0.968 |
0.945–0.992 |
|
ERBI item 6: severe impairment of communication |
5.64 |
0.018 |
1.034 |
1.006–1.064 |
|
ERBI item 7: dysphagia patient in need of supervision |
0.31 |
0.576 |
0.997 |
0.986–1.008 |
|
Ability to reach forward, cm |
0.19 |
0.666 |
0.993 |
0.964–1.024 |
|
Functional Status Score for the Intensive Care Unit score |
10.64 |
0.001 |
1.182 |
1.069–1.308 |
|
Physical Function ICU Test (scored) score |
2.06 |
0.151 |
1.253 |
0.921–1.704 |
|
Grip strength, kg |
0.28 |
0.595 |
0.977 |
0.897–1.064 |
|
Medical Research Council sum score |
||||
|
Upper limb |
4.09 |
0.043 |
0.754 |
0.574–0.991 |
|
Lower limb |
1.90 |
0.169 |
0.847 |
0.668–1.073 |
|
Visual analogue scale, mm |
1.69 |
0.193 |
0.971 |
0.928–1.015 |
|
Montreal Cognitive Assessment |
1.21 |
0.272 |
1.038 |
0.971–1.109 |
|
HR: hazard ratio; CI: confidence interval; ERBI: Early Rehabilitation Barthel Index. |
||||
|
Table III. Description of the final multivariate Cox proportional hazard model for regaining sit-to-stand function. Higher age, longer duration of weaning from mechanical ventilation at baseline, and lower scores on the Functional Status Score for the Intensive Care Unit (FSS-ICU) scale at baseline negatively affects the timing of recovery of sit-to-stand function. The explained variance of the variables in the final model for regaining sit-to-stand function is 21% |
||||
|
Variable |
χ2 |
p-value |
HR |
95% CI |
|
Age, years |
7.25 |
0.01 |
0.96 |
0.94–0.99 |
|
Duration of weaning, days |
4.18 |
0.04 |
0.99 |
0.98–1.00 |
|
FSS-ICU score |
32.43 |
< 0.001 |
1.12 |
1.08–1.16 |
|
HR: hazard ratio; 95% CI: 95% confidence interval. |
||||
DISCUSSION
Our study found that 50% of all included patients were able to sit-to-stand without help a median of 56 days after rehabilitation and after a median duration of illness of 113 days. Age, duration of ventilation, and FSS-ICU score at study onset explained the recovery of sit-to-stand function. To our knowledge, this is one of the first studies to explain the recovery of sit-to-stand function in patients with ICU-acquired muscle weakness. The study therefore provides a more detailed understanding of the pattern and implications for rehabilitation of activities such as sit-to-stand among people with chronic ICU-acquired muscle weakness.
A recent study investigated functional recovery at 6 months among 192 mechanically ventilated ICU patients (approximately 50% had ICU-acquired weakness) (34). In contrast to our study, the authors did not describe the detailed recovery of sit-to-stand function. Our participants all had ICU-acquired muscle weakness, were chronically ill, and the results are therefore not directly comparable with other trials in the field of ICU research.
Only those patients who were able to perform the a priori defined assessments were included in the study. Thus, the study might be limited by excluding some sedated or very agitated patients, and therefore may limit the generalizability of the results to the entire chronically critically ill population. In addition, one could argue that diagnosis of CIP and CIM as major causes of acquired muscle weakness requires clinical and electrophysiological investigation (35). A possible limitation is therefore that we did not always provide an electrophysiological examination. Furthermore, future studies should investigate different rehabilitation strategies, e.g. electrostimulation to improve function (36).
A further limitation could be our method of clinical assessment of muscle strength itself (37). Future research should take into account other measures, such as muscle mass assessment using ultrasound.
Although we showed good recovery of sit-to-stand function, we have not identified which, if any, interventions were effective in achieving this function. A recent Cochrane Review described a lack of randomized trials of people with ICU-acquired muscle weakness with a defined diagnosis of CIP or CIM (38), indicating that little is known about which therapies are effective. Future studies should include people with ICU-acquired muscle weakness with a defined diagnosis of CIP and/or CIM and investigate interventions for improving sit-to-stand function, which is an important prerequisite for ADL.
In conclusion, recovery of sit-to-stand function in people with ICU-acquired muscle weakness occurs after 2 months of rehabilitation. Higher age, longer duration of weaning from mechanical ventilation at baseline, and lower scores on the FSS-ICU scale at baseline negatively affect the timing of recovery of sit-to-stand function.
REFERENCES
