Adriana Costa-Ribeiro, PhD1, Ariadne Maux, PT2, Thamyris Bosford2, Yumi Tenório2,
Déborah Marques, MSc2, Maíra Carneiro, MSc2, Michael A. Nitsche, PhD3,4,5,
Alberto Moura Filho, PhD6 and Kátia Monte-Silva, PhD2
From the 1Department of Physical Therapy, Universidade Federal da Paraíba – UFPB, Paraíba, 2Department of
Physical Therapy, Applied Neuroscience Laboratory, Universidade Federal de Pernambuco – UFPE, Pernambuco,
Brazil, 3Department of Clinical Neurophysiology, Georg August University, Gottingen, 4Leibniz Research Centre for Working Environment and Human Resources, Dortmund, 5Department of Neurology, University Medical Hospital
Bergmannsheil, Bochum, Germany and 6Department of Physical Therapy, Laboratory of Kinesiology and Functional
Assessment, Universidade Federal de Pernambuco – UFPE, Pernambuco, Brazil
OBJECTIVE: To investigate the dopamine-dependent effect of combining transcranial direct current stimulation (tDCS) with visually cued gait training on cortical excitability and functional mobility in individuals with Parkinson’s disease.
DESIGN: A pilot, randomized, double-blind, controlled trial.
METHODS: Twenty-two patients with Parkinson’s disease were randomly assigned to 2 groups: (i) active anodal tDCS over the supplementary motor area (experimental group), or (ii) sham tDCS (control group). After tDCS, both groups participated in a visually cued gait training. Functional mobility was evaluated with the Timed Up and Go test (TUG). Cortical excitability was assessed by active motor threshold and motor-evoked potential amplitudes elicited by transcranial magnetic stimulation in patients in on and off medication states.
RESULTS: In the TUG test both groups achieved improvements either in on or off medication condition compared with baseline. However, for both medication conditions, these gains were maintained only in the experimental group during 1-month follow-up, compared with baseline. In the experimental group, enhancement of cortical excitability was observed at post-intervention and 1-month follow-up (both only for the “on” phase) compared with baseline.
CONCLUSION: These findings suggest that tDCS, independent of dopaminergic medication state, might prolong the positive effect induced by cued gait training on functional mobility.
Key words: transcranial direct current stimulation; cued gait training; cortical excitability; functional mobility; Parkinson’s disease.
J Rehabil Med 2016; 48: 00–00
Correspondence address: Kátia Monte-Silva, Department of Physical Therapy, Applied Neuroscience Laboratory, Universidade Federal de Pernambuco – UFPE. Av. Jornalista Anibal Fernandes, s/n, Cidade Universitária, Recife, Pernambuco, Brazil. E-mail: monte.silvakk@gmail.com
Accepted Jun 29, 2016; Epub ahead of print Sep 9, 2016
Introduction
Parkinson’s disease (PD) commonly impairs some aspects of gait, such as the amount and velocity of movement. The use of temporal or spatial stimuli to encourage movement in patients with PD, namely cued strategies, has been recommended in physiotherapy guidelines (1). Several randomized clinical trials on these strategies have shown some improvements in walking speed and step length (1). However, despite promising results, evidence regarding the effectiveness of coupling physical therapy with visual cued strategies to treat gait disorders in this population is unclear (2); indeed, patients react to the therapy in different ways (2).
Transcranial direct current stimulation (tDCS) has been used recently to enhance the responsiveness of the brain to therapies. The combination of tDCS with traditional therapeutic interventions may result in improvement in clinical outcomes in stroke patients (3). We have demonstrated positive effects on functional mobility of combining tDCS with cueing gait training in outpatients with PD (4). Further knowledge of the efficacy of combining tDCS with motor therapies, and on the manner in which the interaction occurs, would be a key factor in the successful implementation of tDCS as an adjuvant therapy in neurorehabilitation. However, to date, these mechanisms are incompletely understood.
Since the use of levodopa modifies the activity of motor cortico–subcortical networks and thus may significantly influence the effects of tDCS (5), it is important to explore whether the results of combining tDCS with standard therapy is modified by the level of dopamine. We hypothesized that, if the effects of tDCS are dependent on dopamine level, then the beneficial effect of tDCS on the responsiveness of the brain to cued gait training should be abolished in patients with PD who are off medication. Thus, the current study investigated the effects of combining tDCS with cued gait training on cortical excitability and functional mobility of individuals with PD, tracking these effects daily during sessions with and without dopaminergic medication.
Methods
Participants
A randomized controlled trial was performed with the following inclusion criteria: patients with PD Hoehn and Yahr (H&Y) stages I–III while “on” medication, aged 40–80 years, walking independently, medication (levodopa dose ≥ 300 mg daily), and no cognitive impairment. Exclusion criteria were: neurological diseases, visual disorders, unstable medication, metallic implants, inability to walk 10 m, severe freezing (> 15 points) according the Freezing of Gait Questionnaire, and participation in physical therapy elsewhere.
The study was approved by the local Research Ethics Committee and publicly registered (ClinicalTrials.gov: NCT 02250690).
Interventions
Subjects were allocated to groups through the use of an online randomization program (6). Groups received tDCS (2 mA) for 13 min (experimental group, EG) or 30 s (control group, CG). Studies have shown that 13 min of anodal tDCS is sufficient to produce lasting effects (for up to 1 h). The anode was placed 2 cm anterior to the vertex, in the sagittal midline, and the cathode above the supraorbital area over the hemisphere of the more affected side, as determined by patient report.
Sham tDCS was performed by current flow for 30 s in the same positions as for active stimulation. The electrodes were kept on the scalp for the same time (13 min) as for the active tDCS condition.
After tDCS, participants underwent gait training, conducted by physiotherapists. Subjects were instructed to walk at the step length indicated by white strips (visual cue) along the 6.5-m walkway (4). Gait training lasted for 30 min per session, 24 min of active training, with a 6 min interval (i.e. 2 min rest for every 8 min of training). The rehabilitation programme was delivered in 10 sessions over a period of 4 weeks, with a minimum interval of 48 h between sessions, 3 times per week, at the same time of day in the “on” phase (approximately 1 h after drug intake). Session number (training minimum 4 weeks), duration (30 min) and frequency (3 times per week) were chosen according to the recommendations of the European Physiotherapy Guideline for Parkinson Disease for treadmill training (1).
Outcome measures
Functional mobility and bradykinesia were assessed with the Timed Up and Go test (TUG) test and Upper-Limb Motor Task (UL-MT), respectively (4). Motor cortex excitability was determined by the amplitude of motor evoked potential (MEP) and active motor threshold (aMT) according to Groppa et al. (7). These measures were performed only in the most affected hemisphere.
MEP, aMT and TUG test were assessed at baseline, 48 h after the 10-session training and 1-month follow-up in the “off” phase. One hour after drug intake, outcome measures were taken again. In addition, during sessions, cortical excitability (aMT and MEP) and motor function (TUG and UL-MT) were evaluated before and after each session while participants were in the “on” phase (Fig. 1).
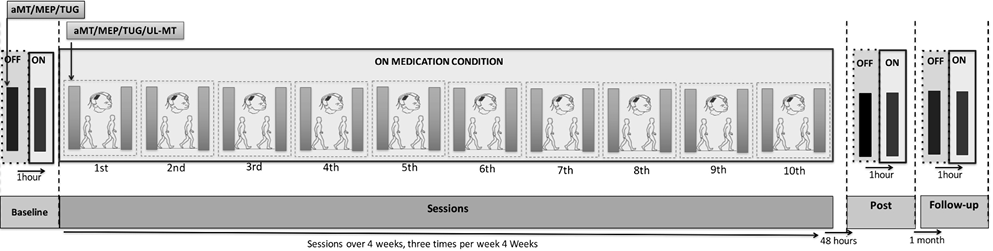
Fig. 1. Experimental course.
Statistical analysis
Data normality was verified with the Shapiro-Wilk test. No data (except aMT) met the criterion of normality.
To investigate cortical excitability changes during and after sessions, repeated-measures analysis of variance (ANOVA) (2 × 13) was used for aMT, with time (baseline, post-1st, 2nd, 3rd, 4th, 5th, 6th, 7th, 8th, 9th and 10th sessions, 48 h post-intervention and 1-month follow up) as the within-subject factor, and group (control and experimental) as the between-subject factor. Friedman’s test (values of 13 time measurements) was used for MEP, TUG and UL-MT.
To investigate the dopamine-dependent effects, ANOVA 3 × 2 (aMT), with time as the within-subject factor, and group as the between-subject factor, or Friedman test (MEP and TUG) for each medication condition, were used separately. Post hoc comparisons were carried out using independent t-test or Mann–Whitney test and paired t-test or Wilcoxon test.
To explore an association between cortical excitability and functional mobility changes, Spearman’s correlation coefficients were calculated. Significance level was set at an alpha value of p < 0.05.
Results
Twenty-four subjects were randomized to the EG (n = 12) and the CG (n = 12). Sample characteristics are shown in Table I.
Friedman’s test revealed significant changes in the TUG scores under “on” (χ2 = 24.364; p < 0.001) and “off” (χ2 = 15.636; p < 0.001) medication conditions. Post hoc tests revealed a decrease in TUG scores compared with baseline in both groups. However, only in the EG were these gains maintained until 1-month follow up compared with baseline (Table II). No significant differences were found between groups for “on” and “off” medication conditions (Table SI1). Figs 2A and 2B show the changes in TUG score (χ2 = 71.511; p < 0.001) and UL-MT score (χ2 = 89.946; p < 0.001) throughout the sessions. TUG scores decreased in both groups compared with baseline. The CG improved significantly after the seventh session, and the EG improved after the second session. However, these gains were maintained during 1-month follow up only in the EG. Compared with baseline, the results of the UL-MT showed decreases in the time test in both groups. No significant differences between groups were found for either outcome.
|
Table I. Sample characteristics |
|||
|
Experimental group (n = 11) |
Control group (n = 11) |
p-value |
|
|
Sex, male, n (%) |
8 (72.7) |
7 (63.6) |
1.000a |
|
Age, years, mean (SD) |
61.1 (9.1) |
62.0 (16.7) |
0.808b |
|
Disease duration, years, mean (SD) |
6.1 (3.8) |
6.28 (3.74) |
0.750b |
|
LED, mg/day, mean (SD) |
740.9 (924.3) |
890.9 (836.0) |
0.694b |
|
Body mass index, kg/m2, mean (SD) |
26.2 (4.46) |
26.0 (3.59) |
0.875b |
|
HY, n (%) |
|||
|
Stage 1 |
1 (9.0) |
1 (9.0) |
0.979a |
|
Stage 1.5 |
1 (9.0) |
1 (9.0) |
|
|
Stage 2 |
3 (27.0) |
3 (27.0) |
|
|
Stage 2.5 |
1 (9.0) |
2 (18.0) |
|
|
Stage 3 |
5 (45.0) |
4 (36.0) |
|
|
MMSE, mean (SD) |
26.4 (3.0) |
25.7 (4.9) |
0.687b |
|
Tremor (presence) n (%) |
8 (72.7) |
9 (81.8) |
0.690b |
|
Falls (presence) n (%) |
8 (72.7) |
7 (63.6) |
0.809a |
|
UPDRS-III (on medication), mean |
19.0 |
19.1 |
0.509b |
|
Depression (HADS), yes, n (%) |
4 (36.3) |
5 (45.4) |
1.000a |
|
aχ2 test; bIndependent sample test. LED: levodopa equivalent dosage; HY: Hoehn & Yahr Modifying Staging scale; MMSE: Mini-Mental State Examination; UPDRS-III: Unified Parkinson’s Disease Rated Scale; HADS: Hospital Anxiety and Depression Scale. |
|||
|
Table II. Differences within and between groups |
||||||||||
|
Differences within groups |
Differences between groups |
|||||||||
|
Post-intervention minus baseline |
1-month follow up minus baseline |
Post-intervention minus baseline |
1-month follow up minus baseline |
|||||||
|
EG Mean (SD) |
CG Mean (SD) |
EG Mean (SD) |
CG Mean (SD) |
EG-CG Mean (95% CI) |
EG-CG Mean (95% CI) |
|||||
|
aMT (%) |
On |
–11.73 (10.35) |
–10.64 (16.83) |
–15.82 (14.46)* |
–0.36 (17.66) |
–1.09 (–13.52 to 11.34) |
–15.46 (–29.82 to –1.10) |
|||
|
Off |
–1.36 (9.26) |
–12.64 (16.45) |
–11.64 (13.21) |
–0.18 (18.74) |
11.28 (–0.59 to 23.15) |
–11.46 (–25.88 to 2.96) |
||||
|
MEP (µV) |
On |
0.22 (0.94) |
0.25 (0.78) |
–0.02 (0.63) |
0.13 (0.78) |
–0.03 (–0.80 to 0.74) |
–0.15 (–0.78 to 0.48) |
|||
|
Off |
–0.05 (0.71) |
0.34 (1.10) |
–0.09 (0.38) |
0.21 (0.81) |
–0.39 (–1.21 to 0.43) |
–0.3 (–0.86 to 0.26) |
||||
|
TUG (s) |
On |
–2.29 (1.80) |
–3.08 (4.15) |
|
–2.06 (2.20) |
0.63 (10.56) |
0.79 (–2.06 to 3.64) |
–2.69 (–9.47 to 4.09) |
||
|
Off |
–1.71 (2.29) |
–3.22 (4.41) |
|
–1.67 (2.39) |
0.32 (10.38) |
1.51 (–1.62 to 4.64) |
–1.99 (–8.69 to 4.71) |
|||
|
Bold values present differences in relation to baseline. *Presents differences between groups (p < 0.05). aMT: active motor threshold; MEP: motor evoked potential; TUG: Timed Up & Go; EG: experimental group; CG: control group; 95% CI: 95% confidence interval; SD: standard deviation. |
||||||||||
In the “on” medication condition, the 2 × 3 ANOVA revealed a significant effect of time×group interaction (F = 2.528; p = 0.004) for aMT. Under the “off” medication condition, no significant effects were found. The 2 × 13 ANOVA conducted for aMT revealed a significant main effect of time×group interaction (F = 2.150; p = 0.015) and time (F = 2.127; p = 0.016) in the “on” medication condition. Compared with baseline, post hoc test revealed a decrease in aMT along the sessions for both groups. However, this reduction was maintained post-intervention and at 1-month follow up only in the EG. The difference between groups was significant at 1-month follow-up (t = 2.245; p = 0.036) (Fig. 2C).
Friedman’s test revealed no significant changes in MEP after sessions and 1-month follow up for groups and for “on” (χ2 = 1.727; p = 0.422) and “off” (χ2 = 0.302; p = 0.860) medication conditions (Table II). Also, at the single session level, no difference was found over time for either group (χ2 = 11.071; p = 0.523) (Fig. 2D).
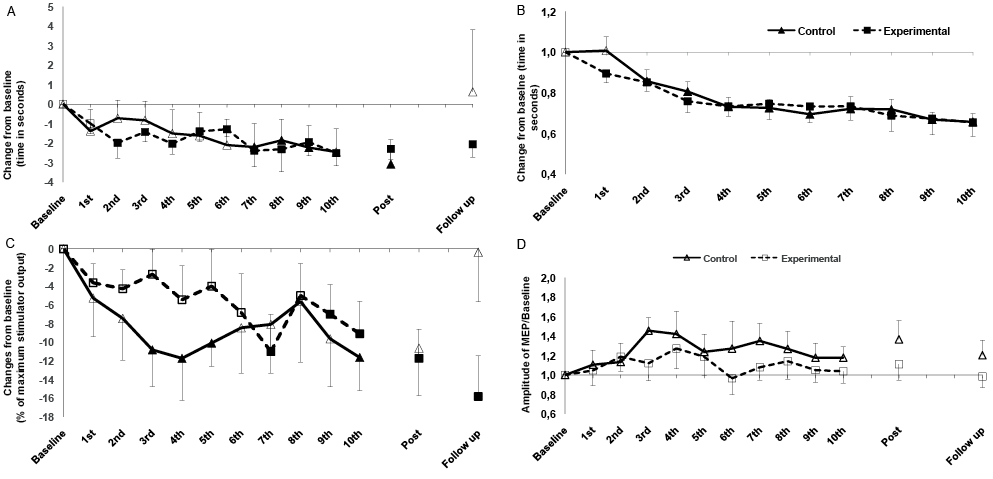
Fig. 2. Intervention-related alterations in: (A) motor function (functional mobility, Timed UP and Go test; TUG); (B) bradykinesia (upper-limb motor task; UL-MT); (C) physiological parameters (active motor threshold; aMT); and (D) motor evoked potential amplitudes (MEP) in patients with Parkinson’s disease in “on” medication states. Solid symbols indicate significant deviations (p ≤ 0.05) from baseline values. *Significant deviations compared with control group (CG) with regard to identical time points.
No significant correlation was found between changes in the clinical (TUG test) and electrophysiological assessment when collapsed data for groups at either post-intervention and follow up were correlated for “on” (aMT r = 0.225; p = 0.143 and MEP r = –0.378; p = 0.08) and “off” (aMT r = 0.067; p = 0.666 and MEP r = –0.185; p = 0.229) medication conditions. Subgroup analyses revealed a positive correlation between aMT-measured cortical excitability and TUG-measured functional gains (r = 0.729; p = 0.011) only for the EG in the “on” medication condition.
Discussion
Functional mobility and bradykinesia
The results of this study show that 10 sessions of cued gait training alone gradually enhances functional mobility and decreases bradykinesia, as observed by a decrease in the TUG test and UL-MT time during sessions. In terms of functional mobility, tDCS did not increase the effect magnitude of cued gait training, but did make its effect faster (from the second session) and more lasting (until 1-month follow-up), compared with sham stimulation, taking into account significant differences vs baseline. The beneficial effect of tDCS in prolonging the cued therapy-induced motor gains of patients with PD in this trial has been reported earlier (4). Here, we observed that the tDCS-induced prolonged after-effect is not affected by dopaminergic medication (i.e. “on” or “off” state).
The current study supports the hypothesis that the combination of stimulation with motor training accelerates and enhances the training effects. Indeed, Parasuraman’s mini-review (8) concluded that tDCS can accelerate learning and enhance performance in motor tasks, and may be a beneficial addition to other training methods. Also, a previous study demonstrated that tDCS alone (8 sessions of anodal tDCS over the motor cortex) improves gait, although only for short time, 24 h after the end of sessions. Similarly, in our study, cued gait training alone improved the functional mobility of patients with PD for only a short time (48 h). Combining both interventions, we observed a prolongation of the beneficial effects on functional mobility (until 1-month follow-up). However, in contrast to the former study, which found that the tDCS-induced positive effects on gait were observed only in the “off “ condition, we found no influence of medication condition on the effects of tDCS. This also suggests more robust effects induced by combined therapy.
The mechanism underlying the tDCS effect in enhancing and accelerating the efficacy of traditional therapies is unknown. One possibility is that, in PD patients, tDCS might induce dopamine release, and thus facilitate rehabilitation. In humans, Khedr et al. (9) reported an enhancement of serum dopamine levels immediately after 6 days of daily 25 Hz transcranial magnetic stimulation over the primary motor cortex, which was correlated with motor performance improvement.
Another possibility is that enhancing cortical excitability prior to therapy might reinforce the positive effects induced by motor practice and translate into improved clinical outcomes compared with traditional therapies alone. This hypothesis was tested in the current study.
Cortical excitability
Cued gait training alone altered cortical excitability, as observed by a decrease in aMT immediately after the end of the therapeutic sessions, but not at 1-month follow-up. Also here, tDCS prolonged this effect. However, in contrast to clinical measurements, regarding neurophysiological parameters, the effects of cued gait therapy and tDCS appeared to be dependent on dopamine level. Interestingly, compared with baseline conditions, therapy-induced excitability enhancement (gait training alone or combined with tDCS) was observed only in the “on” medication condition. These findings are in line with the beneficial effect of levodopa on practice-dependent plasticity (10).
Regarding changes in MEP size, no changes were found after therapeutic sessions in both groups and in the 2 medication conditions. Different response patterns between MEP and MT are not rare (11). The difference in physiological significance of these excitability markers might help to explain these distinct response patterns.
Correlation between cortical excitability and mobility
In accordance with the study by Fregni’s et al. (12), which found a trend toward a positive correlation between improvement in motor function and an increase in cortical excitability in 9 patients, our study found a similar positive correlation was found between changes in cortical excitability and changes in functional mobility in PD. Interestingly, this result was found only for the patient group that received active tDCS. Thus, at least for this condition, physiological alterations are associated with functional gains. A positive correlation between brain stimulation-induced cortical excitability changes and functional gains has also been found in other neurological conditions (13).
Study limitations
An important limitation of this study is the small sample size; thus caution is required in interpretation of the data. Furthermore, since the electrode size was 35 cm2, it is likely that tDCS stimulation was not confined to the targeted supplementary motor area, but also affected the primary motor cortex and premotor areas. A further important limitation was that no accurate assessment was made of the “on” and “off” states. There is a significant variability in clinical practice regarding the timing of the effects of levodopa on motor function.
tDCS was applied before motor training because the previous study by our group found that tDCS applied before, rather than during or after training, optimized motor training-induced plasticity in healthy individuals (14). However, studies have shown that the application of tDCS during task performance is more effective than application of tDCS before the task (15). Thus, further investigation is necessary in order to optimize tDCS protocols in rehabilitation.

1http://www.medicaljournals.se/jrm/content/?doi=10.2340/16501977-2134
REFERENCES
