Vennila Krishnan, PT, PhD1, Matthew Kindig, MS3 and Mehdi M. Mirbagheri, PhD2,3,4
From the 1Department of Physical Therapy, California State University at Long Beach, CA, 2Department of Biomedical Engineering and Medical Physics, Faculty of Medicine, Tehran University of Medical Sciences,3Sensory Motor Performance Program, Rehabilitation Institute of Chicago, 4Department of Physical Medicine and Rehabilitation, Northwestern University, Chicago, IL, USA
OBJECTIVE: Ankle joint control plays an important role in independent walking. This study investigated the effects of robotic-assisted locomotor training on impaired ankle joint control in individuals with chronic incomplete spinal cord injury.
METHODS: Sixteen individuals with incomplete spinal cord injury underwent 12 one-h sessions of robotic-assisted locomotor training for 4 weeks, while 16 individuals with incomplete spinal cord injury served as inactive controls. Changes in ankle control measures, torque and co-activation were evaluated during maximal voluntary contractions in dorsi- and plantar-flexion. Changes in walking performance measures using Timed Up and Go (TUG), 10-m walk (10MWT) and 6-min walk (6MWT) tests were evaluated at 2 time points: baseline and after 4 weeks.
RESULTS: Maximal voluntary contractions torque during both dorsi- and plantar-flexion contractions improved markedly in the robotic-assisted locomotor training group compared with baseline. Furthermore, after the training, co-activation during the dorsi-flexion maximal voluntary contractions decreased in the training group compared with controls. In addition, the training group significantly improved walking mobility (TUG) and speed (10MWT) compared with baseline. Finally, correlation analysis indicated a significant linear relationship between maximal voluntary contraction torques and walking performance measures.
CONCLUSION: These findings provide evidence that robotic-assisted locomotor training improves ankle joint control, which may translate into enhanced walking performance in individuals with chronic incomplete spinal cord injury.
Key words: ankle motor control; co-activation; electromyography; SCI; robotic intervention; MVC; torque.
J Rehabil Med 2016; 48: 00–00
Correspondence address: Mehdi M. Mirbagheri, Physical Medicine and Rehabilitation, Sensory Motor Performance Program, Northwestern University, Rehabilitation Institute of Chicago, 60611 Chicago, IL, USA. E-mail: mehdi@northwestern.edu
Accepted Jun 29, 2016; Epub ahead of print Sep 14, 2016
INTRODUCTION
In individuals with incomplete spinal cord injury (iSCI), independent walking is negatively influenced by, in part, decreased strength (1) and increased co-activation (2) of the muscles around the ankle joint (3). Appropriate ankle joint control plays an important role in independent walking, as it is crucial to maintain body weight support during gait (4). In particular, the dorsi-flexor (DF) muscles that act around the ankle joint play a crucial role in controlling the foot trajectory during the swing phase to ensure adequate foot clearance (5). The plantar-flexor (PF) muscles are critical for propulsive force generation at the end of the stance phase (4, 6). However, in iSCI, both impaired voluntary DF control and PF hyper-excitability produce excessive ankle plantar-flexion during the swing phase, and improper positioning of the lower limb during heel strike (7, 8). Given the mechanical power deficits during gait in subjects with chronic iSCI (3), improving paretic ankle control may considerably improve walking performance.
Positive results from training with the Lokomat after SCI have been reported, but need to be confirmed (9–11). Fundamentally, the motorized exoskeleton replaces the manual assistance provided by therapists with repetitive, guided and task-specific stepping (12), by moving the individuals’ legs in a pre-programmed, physiological gait pattern. The advantages of using such systems include providing training for a longer duration with more physiological and reproducible gait patterns, and quantification of walking performance over the course of recovery (13).
To date, the effectiveness of a robotic-assisted locomotor training (RALT) protocol has mostly been quantified using clinical walking performance measures, such as walking speed (14), endurance, and functional independence (15). Very few studies have investigated its effects on individual joints, specifically the ankle joint (11, 16). Consequently, the purpose of this study was to quantify the effects of a 4-week RALT protocol on ankle motor control during MVC contraction (as assessed by the net joint torque and muscular co-activation), and on overall walking performance measures (as assessed by clinical walking assessments) in chronic iSCI. We hypothesized that, compared with a matched-control group, the RALT group would show improved torque, reduced co-activation of the ankle joint, and enhanced walking performance. We also expected that there would be a positive correlation between the ankle control measures and the walking performance measures.
METHODS
A total of 32 ambulatory subjects with chronic iSCI with incomplete motor function loss were recruited to this study. Sixteen of the participants underwent a 4-week RALT protocol (mean 50.81 years (standard deviation (SD) 7.93 years)) and 16 were assigned to the control group (49.4 years (SD 11.28 years)). The groups were matched by age, time since injury, muscle tone at the ankle plantar-flexors (as assessed by Modified Ashworth Scale (MAS) (17)) and walking ability (Walking Index for Spinal Cord Injury II (WISCI II) (18, 19)). Detailed demographic characteristics for both groups are shown in Table I.
The following inclusion criteria were met by the participants: (a) motor-incomplete SCI with an American Spinal Injury Association impairment scale (AIS) classification of C or D, (b) level of injury above T10, (c) partial to full ambulatory capacity (i.e. the ability to take at least 2 steps independently), (d) passive range of motion of the lower extremity within functional limits for ambulation, and (e) medical clearance to participate in the experiment. Subjects were recruited from the outpatient service of the Rehabilitation Institute of Chicago. The experimental procedure was approved by the Northwestern University Institutional Review Board and participants provided their informed consent.
Robotic-assisted locomotor training protocol
The RALT group participated in locomotor training using the Lokomat 3 times a week over 4 weeks, for a total of 12 training sessions, as has been used previously (11). Each session lasted 1 h, including set-up time, with 30–45 min of training (Fig. 1).
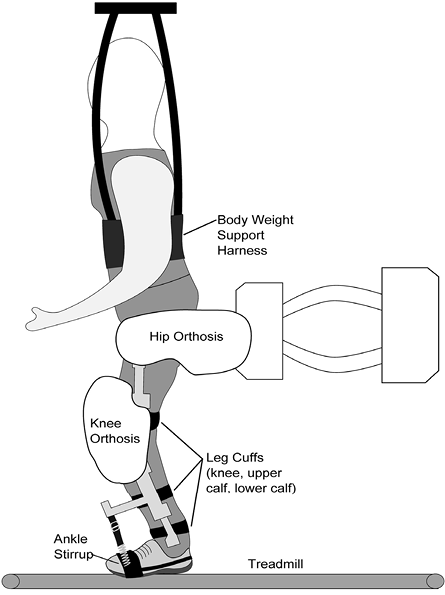
Fig. 1. Robotic-assisted locomotor training (RALT) device.
For the RALT protocol, in accordance with the individual’s tolerance, the treadmill speed was increased from 1.5 to 3.0 km/h, over the course of training. In addition, the guidance force was progressively reduced during the training (from full to 20% assistance), as tolerated by the subject. During the training session, the ankle was held in a neutral position (90° angle between the leg and foot) by spring-supported stirrups that supported the plantar surface of the foot and connected to the knee orthosis of the exoskeleton. The upright weight-bearing position on the neutral foot with a slight closed kinematic tibial tilt provided a stimulation to both dorsi- and plantar-flexors to activate while walking. A mirror was placed before the patient to allow for self-monitoring of his/her leg movements during the training, and the physical therapist provided encouraging verbal feedback throughout the training.
Experimental set-up and instrumentation
The participants’ ankle motor control was evaluated using MVC assessments on the first day of training and on the last day after training. Each participant was seated in an experimental chair with their thigh strapped to the chair base (Fig. 2), and their bare foot secured to a rigid footrest attached to the rotational axis of a servomotor. The seat, footrest, and motor were adjusted, such that the ankle was kept in the neutral position, while the knee joint was flexed at 60°. A 6-axis torque transducer recorded net joint torque of the ankle. Torque data were sampled at 1 kHz by a 16 bit A/D converter (National Instruments, Austin, TX, USA), and passed through a 230 Hz anti-aliasing filter on-line. The isometric testing procedure from the experimental setup is valid (11, 14, 20). Surface electromyography (EMG) was recorded from the tibialis anterior (DF muscle) and gastrocnemius (PF muscle) via bipolar surface electrodes (DE 2.1 Single Differential Surface EMG Sensor, Delsys, Inc., Boston MA, USA). Prior to the experiment, the skin area was cleaned by isopropyl alcohol. EMG signals were sampled at 1 kHz, amplified (gain 10,000), high-pass filtered at 10 Hz and low-pass filtered at 230 Hz.
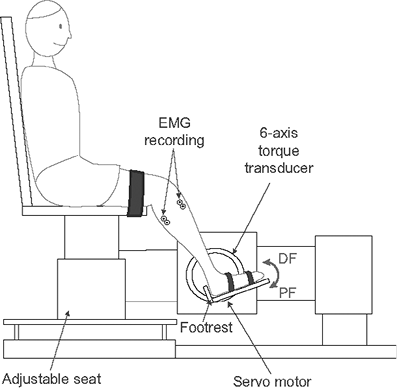
Fig. 2. Experimental apparatus used to evaluate ankle isometric maximal voluntary contractions.
Procedure
Participants were instructed to perform a 5-s MVC “as hard as possible” in each direction by lifting their toes up (DF) or by pushing down with their toes onto the footrest (PF). The MVC during the dorsi-flexion contraction determined the isometric torque for the DF (TDF), while MVC during the plantar-flexion contraction determined the isometric torque for the PF (TPF).
Data analysis
Data analysis was performed using Matlab (The Mathworks, Natick, MA, USA). Prior to further analyses, torque signals were low-pass filtered with a cut-off frequency of 5 Hz. Isometric torque was calculated as the mean torque during the middle 3-s window for each contraction direction (TDF and TPF). Baseline noise of the initial 1-s, when the subject was relaxed, was subtracted from the respective filtered EMG signals. To facilitate comparisons across subjects, EMG values for each muscle were normalized to the peak activity of the respective muscles for each subject. Finally, the co-activation for dorsi-flexion (CADF) and plantar-flexion (CAPF) during MVC was calculated based on previous literature (21). To assess the participants’ functional ambulation capacity, a total of 3 clinical evaluations were performed pre- and post-training: the Timed Up and Go test (TUG) (22), the 10-m walk test (10MWT) (23), and the 6-min walk test (6MWT) (24). The participants walked with their usual walking aids. The isometric torques (TDF and TPF), co-activations (CADF and CAPF) and the clinical evaluations were performed at the same 2 time points as the MVC, i.e. before training (baseline) and after 4-weeks. Baseline scores for both RALT and control groups are shown in Table I.
|
Table I. Baseline demographic characteristics and clinical scores of the robotic-assisted locomotor training (RALT) and control groups |
|||
|
Baseline demography |
RALT (n = 16) |
Control (n = 16) |
p-value |
|
Age, years, mean (SD) [range] |
50.81 (7.93) [37–70] |
50.02 (9.57) [28–68] |
0.73 |
|
Lesion duration, years, mean (SD) [range] |
11.38 (9.65) [2–36] |
10.35 (9.79) [1–36] |
0.42 |
|
WISCI II (0–20), years, mean (SD) [range] |
15.31 (4.17) [9–20] |
14.50 (4.31) [9–20] |
0.70 |
|
TUG, s, median (IQR) [range] |
18.23 (18.51) [9.7–126.1] |
44.57 (41.37) [8.7–115.0] |
0.14 |
|
10MWT, s, median (IQR) [range] |
17.08 (37.71) [0.05–1.06] |
31.94 (48.47) [0.06–1.49] |
0.59 |
|
6MWT, m, median (IQR) [range] |
229.64 (87.36) [25.5–346.0] |
126.51 (152.95) [20.7–501.2] |
0.34 |
|
Sex (F-M), n (%) |
8 (50) – 8 (50) |
4 (25)–12 (75) |
|
|
Level of lesion (C-T), n (%) |
9C (56), 7T (44) |
9C (56), 7T (44) |
0.72 |
|
Ankle flexors MAS (0–4), median (IQR) |
2 (1) |
2 (1) |
0.10 |
|
F: female; M: male; C: cervical; T: thoracic; MAS: Modified Ashworth Scale; WISCI II: Walking Index for Spinal Cord Injury II; TUG: Timed Up and Go; 10MWT: 10-m walk test; 6MWT: 6-min walk test, SD: standard deviation; IQR: interquartile range. |
|||
Statistical analysis
Data were presented as mean and standard deviations within each group. Kolmogorov-Smirnov tests of normality were used to test the normality of the data. Whenever normality was violated, non-parametric tests were used in the analysis. To test the homogeneity between the control and RALT groups at the time of inclusion, Mann-Whitney tests were performed on the following continuous variables: age, lesion duration, WISCI II, TUG, 10MWT and 6MWT. χ2 tests were performed on the categorical (level of lesion) and ordinal variables (MAS).
Paired sample t-tests were used to compare the values at baseline and at 4 weeks for both the RALT and the control groups separately. Effect sizes were assessed with standardized response means (SRM) (25) for torque and co-activation data, where the mean pre-post RALT training change was divided by the standard deviation of the change. Based on Cohen’s criteria, SRM values of 0.20, 0.50 and 0.80 indicate small, moderate and large effects, respectively (25). The pre-post RALT training change was determined by subtracting the baseline from the 4-week measure.
To determine the effect of the RALT on the walking performance (second hypothesis), Wilcoxon signed-rank tests were performed on baseline and 4-week data independently for each group for each of the walking assessments (TUG, 10MWT and 6MWT) as they were non-normally distributed. We also used Spearman’s rank correlation analysis to determine the relationships between the 3 walking capacity measures (i.e. TUG, 10MWT and 6MWT) and the ankle’s isometric and co-activation measures at baseline. All statistical tests were performed using SPSS v.21 (SPSS Inc., Chicago, IL, USA) with a significance (alpha) level of 0.05.
RESULTS
Demographic characteristics
Both the RALT and the control groups were similar in demographic and clinical measures at the baseline evaluation. There was no difference between the groups in terms of age (p = 0.73), duration of lesion (p = 0.42), level of lesion (χ2 = 0.1, p = 0.72), walking capacity measured by WISCI II (p = 0.70), plantar muscle tone assessed by MAS (χ2 = 7.7, p = 0.10), TUG (p = 0.14), 10MWT (p = 0.59) and 6MWT (p = 0.34).
Isometric torque, co-activations and clinical measures
The RALT group (Fig. 3) showed a mean increase of 20.7 % (SD 8.2) in TDF (p < 0.05), and a mean increase of 22.4 % (SD 9.4) in TPF (p < 0.05), with a moderate training effect size (SRM = 0.57) for both. In contrast, for the control group, no statistically significant difference was found between baseline and 4 weeks for both TDF (p = 0.86) and TPF (p = 0.14). The RALT group showed a reduction in the co-activation of the PF muscle after 1 month of RALT training; this training effect was moderate (SRM = 0.53). We did not see any statistically significant reduction between baseline and 4 weeks for the control CADF (p = 0.64) and CADF (p = 0.31) or for the RALT CAPF (p = 0.62).
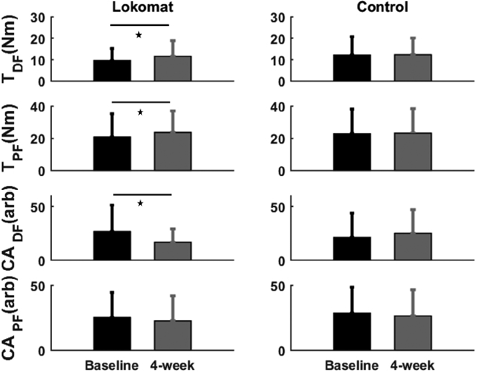
Fig. 3. Baseline and 4-week mean values in isometric maximal voluntary contractions (MVC) torque and co-activation of the ankle measures for robotic-assisted locomotor training (RALT) and control group. Bars represent the standard deviation of the group mean. *Significance at p < 0.05 between pre- and post-tests.
The mean TUG showed a significant reduction of 13.9% (SD 3.2) (Fig. 4) in the time needed to perform the task (p < 0.05) with a moderate effect size (SRM=0.54) for the RALT group. The 10MWT exhibited a significant increase in the mean over-ground gait speed (0.08 m/s (SD 0.02)) after training (p < 0.05). This change corresponded to a mean improvement of 13.0 % (SD 2.6), with a moderate effect size (SRM = 0.71). However, no significant change (p = 0.33) was observed in walking endurance (6MWT). For the Control group, no statistically significant differences were found between the pre- and post-tests for TUG (p = 0.54), 10MWT (p = 0.45) and 6MWT (p = 0.34).
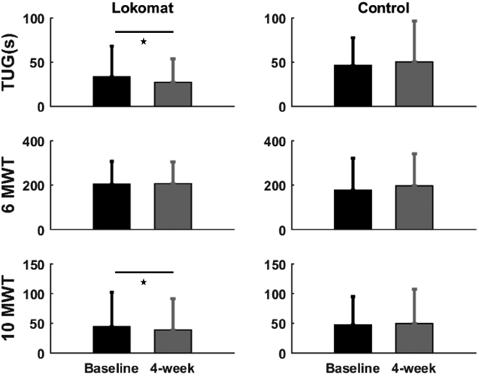
Fig. 4. Baseline and 4-weeks mean values in clinical measures for the robotic-assisted locomotor training (RALT) and the control group. Bars represent the standard deviation of the group mean. *Significance at p < 0.05 between pre- and post-tests. TUG: Timed Up and Go test; 10MWT: 10-m walk test; 6MWT: 6-min walk test.
Relationship between ankle control measures and walking performance
In order to show the important role of ankle control measures in walking performance, we combined the baseline measures of the intervention and control groups and determined the correlation between the ankle control measures (torque and co-activation) and the walking performance (TUG, 10 MWT and 6 MWT). The correlation coefficients are shown in Table II. All of the walking performance tests (walking mobility assessed by the TUG, over-ground walking speed measured by the 10MWT, and walking endurance assessed by the 6MWT) showed significant linear correlations with TDF and TPF (p < 0.05 for all). The correlation test implied that the muscle strength of the ankle is related to the clinical measures of walking. However, the walking performance measures did not show any correlations with the co-activation tests.
|
Table II. Correlations (r values) show a linear relationship between clinical walking performance measures and torques at baseline for both groups combined |
||
|
TDF |
TPF |
|
|
TUG |
–0.51* |
–0.42* |
|
10MWT |
–0.62* |
–0.53* |
|
6MWT |
0.57* |
0.46* |
|
*p < 0.05. TUG: Timed Up and Go; 10MWT: 10-m walk test; 6MWT: 6-min walk test. TDF and TPF: isometric torque in dorsi- and plantar-flexion directions. |
||
DISCUSSION
In this study, we evaluated changes in ankle control measures (torque and co-activation) during MVCs, along with walking performance in individuals with chronic incomplete SCI with and without RALT. For both the groups, clinical gait and ankle control measures were assessed at baseline and after 4 weeks to evaluate ankle impairment and walking performance. The RALT group showed increased maximal torque during both dorsi- (TDF) and plantar-flexion (TPF) contractions after the training. Moreover, co-activation during dorsiflexion decreased in the RALT group after training, whereas there were no changes in co-activation seen in plantarflexion. Regarding the clinical walking performance measures, RALT significantly improved walking mobility (TUG) and over-ground walking speed (10MWT) with no change in walking endurance after the training. Lastly, significant correlations were observed between the maximal isometric torques (TDF and TPF) and the walking performance measures.
Robotic-assisted locomotor training effects on ankle isometric torque
The RALT group generated ~20% more isometric torque from their baseline level during both dorsi- and plantar-flexion MVC contractions. This is consistent with a previous study that reported increased torque in the PF muscle after manually assisted locomotor training (26). Our study extends that result by finding that RALT can improve voluntary torque for both the DF and PF muscle groups.
Spinal cord injury causes a reduction in neural drive in individuals with iSCI, which causes reduced strength and muscle atrophy (27). Specifically, in the ankle joint, a deficit in cortico-spinal voluntary activation of the DF muscle (7, 28), along with spinally mediated changes, such as an increased reciprocal inhibition from spastic triceps surae muscles (29), could lead to weakness and reduced ability to produce torque (30). Nevertheless, we know that exercise training is beneficial in chronic iSCI as it improves skeletal muscle strength (31). By guiding the lower limb in a physiological step pattern and allowing upright weight-bearing on the neutral positioned foot with a closed kinematic tibial tilt would have provided a stimulation to both dorsi- and plantar-flexors to activate while walking; a condition known to promote the recovery of function (32).
Robotic-assisted locomotor training effects on co-activation at the ankle joint
The level of co-activation of the PF muscle decreased in RALT compared with the controls after training. Increased antagonist co-activation in the ankle joint is prevalent in individuals with iSCI during dynamic controlled movement (33) as well as in isometric contractions. During maximal isometric contractions, antagonist co-activation tends to reduce the efficiency of the agonist, increase the metabolic cost, and reduce the maximal net torque available to the joint (34). It has also been reported that specific agonist training would be a source of improvement in performance and skill (35). We believe that through the RALT protocol’s weight bearing training, the level of PF co-activation could have decreased, which, in turn, might have improved the efficiency of the agonist (36).
Robotic-assisted locomotor training effects on walking performance
Four weeks of RALT showed an improvement in the walking speed (10MWT) and walking mobility (TUG) in individuals with chronic iSCI compared with baseline values. This is in line with the observations seen with robotic-assisted gait devices that show a beneficial effect on walking capacity (15, 37). Of particular importance is the change seen in walking speed in our study after RALT training. The mean increase in speed in 10MWT was approximately 0.08 m/s in the RALT group after 4 weeks, which was larger than the minimally important difference of 0.05 m/s reported by Musselman after BWSTT (38). Key factors that could have contributed to this notable improvement in walking speed and mobility are: (i) increased DF torque due to increased DF strength and decreased PF co-activation allowing for adequate foot clearance during the swing phase (39), and (ii) increased PF torque due to the strengthened ankle PF muscle, which provides most of the energy required for body propulsion (6). The lack of changes in the 6MWT may be explained by the nature of the test itself, as it is an endurance-based test. In addition, the short duration of training in our study would have also contributed a lack of changes. Previous RALT studies, however, have shown substantial gains in walking endurance. The reason for the difference in this study could be the short duration of training compared with longer training durations in the other studies (15, 40).
Study limitations
This study had some limitations. The control group was inactive and therefore the increase in function in the RALT group could have been due to the intervention group being more active. The EMG activation of DF and PF muscles during training was not assessed. Finally, we focused on only the ankle joint; future research could be examine other relevant joints involved in ambulation, such as the knee and hip joints.
Conclusion
In this study of individuals with chronic iSCI, 12 training sessions of RALT resulted in ~20% increases in net joint torque, decreased co-activation in the ankle joint, and increased walking mobility (assessed by TUG) and speed (assessed by 10MWT). These improvements in the ankle joint after RALT are clinically meaningful given the ankle’s role in a functional task such as walking. The correlation between ankle torque and walking measures is a noteworthy observation that positively emphasizes the importance of the ankle muscles in gait.
ACKNOWLEDGEMENT
The authors would like to thank X. Niu and D. Varoqui for their assistance with running the experiments and operating the RALT. The study was supported by the National Institute of Health [5R01HD059895].
The authors declare no conflicts of interest.
REFERENCES
