Wouter J. Harmsen, MSc1,2, Gerard M. Ribbers, MD, PhD1,2, Bart Zegers, MD1,2, Emiel M. Sneekes, MSc2, Majanka H. Heijenbrok-Kal, PhD1,2, Ladbon Khajeh, MD3, Fop van Kooten, MD, PhD3, Sebastiaan J. C. M. M. Neggers, MD, PhD4 and Rita J.G. van den Berg-Emons, PhD1,2
From the 1Rijndam Rehabilitation Institute, Rotterdam, 2Department of Rehabilitation Medicine, 3Department of Neurology and 4Department of Endocrinology, Erasmus MC University Medical Center Rotterdam, Rotterdam, The Netherlands
OBJECTIVE: To assess cardiorespiratory fitness in patients following an aneurysmal subarachnoid haemorrhage and to explore this in fatigued and non-fatigued patients.
DESIGN: Cross-sectional case-control study.
SUBJECTS/PATIENTS: A total of 28 patients, 6 months post aneurysmal subarachnoid haemorrhage, and 28 sex- and age-matched controls.
METHODS: Cardiorespiratory responses to a progressive cardiopulmonary exercise test on a cycle ergometer were obtained using indirect calorimetry. Fatigue was assessed using the Fatigue Severity Scale.
RESULTS: Mean peak oxygen uptake (V̇O2peak) was significantly lower in patients (22.0 (standard deviation (SD) 6.2) ml/kg/min) than in controls (69% of controls, p < 0.001). All other cardiorespiratory fitness parameters were also lower, with peak levels ranging from 62% to 77% of matched controls. Mean V̇O2peak was 19.4 (SD 4.1) ml/kg/min in fatigued patients (63% of matched controls, p < 0.001) and 23.9 (SD 6.9) ml/kg/min in non-fatigued patients (74% of matched controls, p = 0.002).
CONCLUSION: Cardiorespiratory fitness is impaired after aneurysmal subarachnoid haemorrhage, both in fatigued and non-fatigued patients. This finding may have implications for treatment.
Key words: subarachnoid haemorrhage; physical fitness; cardiopulmonary exercise test; fatigue.
J Rehabil Med 2016; 48: 00–00
Correspondence address: Wouter Harmsen, Rehabilitation Medicine, Erasmus MC, 3015 CE Rotterdam, The Netherlands. E-mail: w.harmsen@erasmusmc.nl
Accepted Jun 16, 2016; Epub ahead of print Aug 26, 2016
INTRODUCTION
Aneurysmal subarachnoid haemorrhage (a-SAH) is a potential life-threatening stroke, which accounts for 5% of all stroke cases (1, 2). Advances in diagnostic and surgical management have increased the chance of surviving an a-SAH, which reaches 65% (3). With a growing number of patients surviving an a-SAH, understanding its long-lasting sequelae becomes increasingly important.
With only one-third of survivors being able to fully resume their previous occupation, a-SAH has a significant impact on society, patients’ families and on patients themselves (3, 4). Long-term cognitive and emotional problems may be present (5). One of the most distressing complaints is fatigue, which has a prevalence ranging from 31% to 90% (6). Fatigue may persist for up to 7 years after a-SAH (7), and is associated with many factors, including sleep disturbance, anxiety, emotional problems, passive coping styles, and cognitive impairment (6). Another interesting hypothesis is that the presence of fatigue may be a result of impaired cardiorespiratory fitness, triggered by physical deconditioning (6, 8). Associations between cardiorespiratory fitness and fatigue have been reported among several non-stroke patient groups (9–12).
Cardiorespiratory fitness is defined as the ability of the cardiorespiratory system to supply oxygen to the skeletal muscles during sustained physical activity (13). In patients with stroke not caused by a-SAH, the cardiorespiratory fitness is almost half that of sex- and age-matched controls (14). In these patients, the beneficial effects of improved cardiorespiratory fitness are well-recognized, and exercise training has become an integral component of stroke rehabilitation (15). Such training contributes to cardiovascular health and seems to provide a physical reserve to fatigue (14, 16). However, to our knowledge, cardiorespiratory fitness has not yet been investigated after a-SAH and deserves attention in rehabilitation medicine.
The primary goal of this study was to assess cardiorespiratory fitness in patients after a-SAH, and to explore this in fatigued and non-fatigued patients. Cardiorespiratory responses to a progressive cardiopulmonary exercise test were compared with that of sex- and age-matched controls. We hypothesized that cardiorespiratory fitness will be impaired in a-SAH, particularly in fatigued patients.
METHODS
Setting and participants
The present case-control study, entitled HIPS-Rehab, was part of a longitudinal observational study, Hypopituitarism In Patients after Subarachnoid haemorrhage (HIPS) (17). HIPS-Rehab focuses on a-SAH related consequences from a rehabilitation perspective and includes measurements on physical fitness and physical activity. In the present study we used data measured at 6 months post a-SAH. The study was approved by the Medical Ethics Committee of Erasmus University Medical Centre, and all participants gave written informed consent.
Patients with a-SAH admitted to the Department of Neurology of the Erasmus Medical Centre, the Netherlands, between June 2009 and June 2012 were eligible for inclusion when they were discharged from the intensive care unit (ICU) and aged ≥ 18 years. Diagnosis of a-SAH was confirmed by computerized tomography (CT) of the brain and in cases with negative CT, by lumbar puncture. Presence and location of the aneurysm was determined by CT angiography and/or digital subtraction angiography. Patients were excluded if they met any of the following criteria: (i) hypothalamic or pituitary disease diagnosed prior to a-SAH; (ii) history of cranial irradiation; (iii) trauma capitis prior to a-SAH; (iv) other intracranial lesion apart from a-SAH; or (v) other medical or psychiatric condition or laboratory abnormality that may interfere with the outcome of the study. Additional exclusion criteria regarding the physical fitness measurements were: aged ≥ 70 years, and not eligible for maximal exercise testing, as determined by the treating physician and the Physical Activity Readiness Questionnaire (PAR-Q) (18).
Since appropriate Dutch reference values for cardiorespiratory fitness were not available, we included a comparison group. Each patient was individually paired with a control subject with respect to sex and age (± 5 years). Controls were recruited by advertisement and screened on medical contraindications for physical exercise testing by a physician using the PAR-Q (18), and a questionnaire concerning medical history and sports participation (developed for this study and available on request).
Safety procedure
This was the first study to specifically investigate cardiorespiratory responses to progressive cardiopulmonary exercise testing (CPET) in patients after a-SAH. Therefore, we implemented a supplementary safety protocol, which matches the recommendations for conducting CPET after stroke (19). First, all patients were screened for absolute and relative medical contraindications to physical exercise testing by a neurologist and a sports physician. If there was any suspicion of underlying cardiovascular or pulmonary pathology, progressive CPET was not carried out. In addition, resting blood pressure was recorded twice prior to CPET with a few minutes of rest between measurements. During CPET, blood pressure was measured using an automatic system, heart function was monitored using a 12-lead electrocardiogram[a] (Oxycon Pro, CareFusion, Houten, The Netherlands) and a sports physician provided emergency back-up.
Cardiorespiratory fitness
CPET was performed using a progressive ramp protocol on an electronically braked cycle ergometer[b] (Jaeger ER800, Jaeger Toennies, Breda, The Netherlands), which is frequently performed after stroke and determined to be feasible in a selected group of patients who had undergone pre-test medical screening (19). Gas exchange analyses were performed during CPET by indirect calorimetry using the Oxycon Pro[a] Oxycon Pro, CareFusion, Houten, The Netherlands, a breath-by-breath oximetry analysing system.
Participants were instructed to abstain from consuming food or caffeine prior to CPET. Before each measurement, volume and gas calibrations were performed. After calibrations, participants warmed up for 4 min without resistance, after which a ramp protocol was implemented with the goal of having the participants reach their maximum physical effort within 10–14 min. Resistance increased automatically every 10 s and varied by sex (female: 12 W/min, male: 16 W/min). Participants were instructed to pedal at a rate of 60–70 revolutions per min. Strong verbal encouragement was provided, to continue for as long as possible until maximal voluntary exhaustion. CPET was terminated when participants reached volitional fatigue, or if they were unable to maintain target pedal rate. CPET could also be terminated because of medical complications, as prescribed in the guidelines of the American College of Sports Medicine (ACSM) for exercise testing (e.g. sustained ventricular tachycardia, ST elevation, moderately severe angina, reduction in systolic blood pressure of >10 mmHg from baseline blood pressure) (13).
The following peak cardiorespiratory responses were analysed: (i) oxygen consumption (VO2peak), the criterion standard for cardiorespiratory fitness, which was expressed in absolute VO2peak (ml/min) and relative V̇O2peak per kg body mass (ml/kg/min) and per kg fat-free mass (ml/kg FFM/min); (ii) ventilation (VEpeak), a measure of pulmonary capacity (l/min); (iii) oxygen pulse (VO2peak/HRpeak), a measure of cardiac output (ml/beat); (iv) heart rate (HR) in beats/min; (v) respiratory rate (RR) in breaths/min; (vi) respiratory exchange ratio (RER) (VCO2/VO2); and (vii) the ventilatory equivalent for oxygen (VEpeak/VO2peak), a measure of ventilatory efficiency (l/min). Cardiorespiratory responses were analysed at peak physical work rates, defined as the highest mean values recorded during 30 s of exercise.
In addition, the ventilatory anaerobic threshold was estimated by 2 assessors using the ventilatory equivalent method (20); defined as the moment at which the ventilatory equivalent for CO2 (VE/VCO2) and end-tidal CO2 partial pressure (PETCO2) remained stable, and the VE/VO2 and the end-tidal O2 partial pressure (PETO2) increased disproportionally.
Only participants who fulfilled at least one of the objective criteria for maximal physical exertion were included in the final analyses. The following criteria were used to determine maximal physical exertion: (i) RER of >1.0 (19) or (ii) HRpeak within 10 beats/min (bpm) of the age predicted maximum heart (HRmax) rate calculated from the formula of Tanaka et al. (21):
HRmax = 208– (0.7 * age)
As β-blocker medication reduces maximal heart rate by 25–30% (22), the equation was adjusted for patients with β-blocker medication.
HRmax = 0.7 [208– (0.7 * age)]
Fatigue
Fatigue was measured using the Fatigue Severity Scale (FSS) (23). The FSS is a brief and simple instrument consisting of 9 statements about fatigue, scored on a 7-point scale that ranges from 1 (strongly disagree) to 7 (strongly agree). The total score is the mean of the 9 item scores. Higher scores indicate more impact of fatigue on daily life. Fatigue was defined as a score of more than 1 standard deviation (SD) above the mean score for healthy individuals (FSS score ≥ 4.0) (24). The FSS is a widely used measure of fatigue and has been validated for stroke patients in a large Swiss cohort (25).
Clinical and personal characteristics
The following patients’ characteristics were collected: (i) severity of a-SAH, determined by the World Federation of Neurologic Surgeons (WFNS) grade (low-grade: I–III or high-grade: IV–V) (26) and the Glasgow Coma Scale (GCS) score (27), (ii) location of the aneurysm (anterior or posterior circulation), (iii) treatment procedure (surgical clipping or endovascular treatment), (iv) neurological comorbidities (paresis or spasticity), (v) presence of secondary health complications (re-bleed; delayed cerebral ischaemia; hyponatraemia; hydrocephalus; and growth hormone deficiency, defined as an insufficient growth hormone (GH) response to a GH-releasing hormone (GHRH-arginine test) (17). In addition, we collected data on follow-up care (i.e. discharge to the patients’ home environment, a SAH outpatient aftercare clinic, or an inpatient rehabilitation centre).
Body anthropometry was examined in both patients and controls; (i) body mass index (BMI) was calculated from height and body mass (kg/m²), (ii) waist circumference (cm) was measured midway between the lowest rib and the iliac crest while standing, and (iii) thickness of 4 skinfolds (biceps, triceps, subscapular, and supra-iliac region) was measured twice at the left side of the body with a Harpenden Skinfolds Caliper (Burgess Hill, UK). The mean of 2 measurements was used to predict percentage body fat, from which the fat-free mass (FFM) was calculated (28).
Furthermore, a questionnaire regarding participation in sports and/or moderate intensive daily physical activities (including recreational activities, such as cycling, swimming and walking) was completed (developed for this study and available on request).
Statistical analyses
All data are expressed as means with standard deviation (SD) unless otherwise indicated. To compare data for participants with non-participants, independent t-tests were used for continuous data and χ2 tests were used for categorical data. To compare participants’ characteristics and CPET data of patients with that of control subjects, paired samples t-tests were applied for continuous data, χ2 tests for categorical data. Secondly, CPET data of fatigued (FSS score ≥ 4.0) and non-fatigued patients (FSS score < 4.0) were explored with descriptive statistics and compared with that of controls, using paired samples t-tests. A probability value of p < 0.05 was considered statistically significant. To adjust for multiplicity we applied a Bonferroni correction. All analyses were performed using IBM SPSS Statistics, version 20 (SPSS Inc., Chicago, IL, USA).
RESULTS
Descriptive results
Of the 241 patients admitted to the ICU with a diagnosis of a-SAH, 84 were included in the HIPS, of whom 52 volunteered to participate in the HIPS-Rehab (Fig. 1, flow diagram). Patients in the HIPS-Rehab (n = 52) did not differ from non-participants (n = 32) for the severity of a-SAH, location of the aneurysm, treatment procedure, and the presence of secondary health complications.
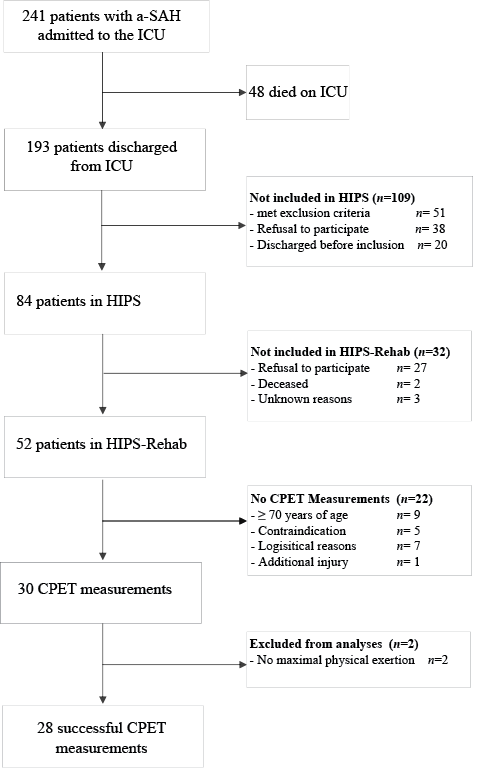
Fig. 1. Consort flow diagram. a-SAH: aneurysmal subarachnoid haemorrhage; ICU: intensive care unit; HIPS: hypopituitarism in patients after subarachnoid haemorrhage; CPET: cardiopulmonary exercise testing.
Of the 52 patients in the HIPS-Rehab, 28 performed successful CPET measurements. Table I presents the patients’ characteristics. The neurological scores showed that 23 patients (82%) had low-grade a-SAH and mean GCS-score was 13.6 (SD 2.1). The majority had a ruptured aneurysm in the anterior circulation (64%) and most underwent endovascular coiling (79%). None of the patients had a paresis or showed signs of spasticity. All were discharged to their home environment, and 11 were redirected to a SAH outpatient aftercare clinic (including a visit to the rehabilitation specialist, neuropsychologist and specialized nurse). Nine of the 28 patients were on beta-blocker medication. According to the medical pre-screening procedure, there were no other cardiovascular or pulmonary pathologies detected. Except that the included patients were younger (mean difference 7.8 years (SD 3.0), p = 0.012), they did not differ from those who were excluded (n = 24) from the final analyses.
|
Table I. Descriptive characteristics of patients with aneurysmal subarachnoid haemorrhage (a-SAH) |
|
|
Patient characteristics |
Patients with a-SAH (n = 28) |
|
Age, years, mean (SD) |
53 (10) |
|
Gender, males, n (%) |
8 (29) |
|
WFNS grade, n (%) I II III IV V |
14 (50) 9 (32) 0 (0) 4 (14) 1 (4) |
|
Glasgow Coma Scale, mean (range) |
13.6 (5–15) |
|
Location of aneurysm, n (%) Anterior circulation Posterior circulation |
18 (64) 10 (36) |
|
Aneurysm treatment, n (%) Clipping Endovascular |
6 (21) 22 (79) |
|
Complications following a-SAH, n (%) |
|
|
Re-bleed |
0 |
|
Delayed cerebral ischaemia |
7 (25) |
|
Hyponatraemia |
4 (14) |
|
Hydrocephalus |
9 (32) |
|
Growth hormone deficient |
2 (7) |
|
WFNS: World Federation of Neurologic Surgeons grading system for subarachnoid haemorrhage scale; SD: standard deviation; SAH: subarachnoid haemorrhage. |
|
Characteristics of patients and controls are presented in Table II. BMI and waist circumference were higher in patients than in controls. Patients participated less frequently in sports and/or moderate intensive daily physical activities than controls (43% vs 96%, respectively; χ2 = 19.0, p < 0.001). Furthermore, fatigue was more frequently present in patients than in controls (χ2 = 9.54, p = 0.002).
|
Table II. Characteristics of patients with aneurysmal subarachnoid haemorrhage (a-SAH) and control subjects |
||||
|
Patients post a-SAH (n = 28) |
Controls (n = 28) |
95% CI for the difference |
p-value |
|
|
Age, years, mean (SD) |
53 (9) |
52 (9) |
– |
|
|
Gender, males, n (%) |
8 (29) |
8 (29) |
– |
|
|
Weight, kg, mean (SD) |
74.5 (10.2) |
72.2 (9.1) |
–2.9 to 7.5 |
0.367 |
|
Body mass index, kg/m2, mean (SD) |
27.1 (3.5) |
24.1 (2.1) |
1.3 to 4.8 |
0.001 |
|
Percentage body fat, %, mean (SD) |
35.3 (6.9) |
32.7 (6.0) |
–0.76 to 5.0 |
0.067 |
|
Waist circumference, cm, mean (SD) |
92.5 (8.5) |
83.2 (8.3) |
5.1 to 13.4 |
< 0.001 |
|
Fat free mass, kg, mean (SD) |
48.1 (8.0) |
48.9 (8.8) |
–4.6 to 2.2 |
0.475 |
|
Participation in sports and/or moderate intensive physical activity, n (%)a |
12 (43) |
27 (96) |
< 0.001 |
|
|
Fatigue FSS-score, mean (SD) |
3.5 (1.5) |
2.5 (1.0) |
0.34 to 1.7 |
0.005 |
|
Non-fatigued (< 4.0), n (%) |
16 (57) |
26 (93) |
0.002 |
|
|
Fatigued (≥ 4.0), n (%) |
12 (43) |
2 (7) |
||
|
aParticipation in sports and/ or moderate intensive physical activities (such as cycling, swimming and walking) at least once a month. FSS-score: Fatigue Severity Scale score; 95% CI: 95% confidence interval. |
||||
Cardiorespiratory fitness
Table III presents CPET outcome of patients and controls. All CPET measurements were terminated because of volitional fatigue; no adverse medical complications were observed. Fifteen patients (54%) terminated the test because of dyspnoea and 13 (46%) because of muscular fatigue; in controls this was 19 (68%) and 11 (32%), respectively. Patients had a mean V̇O2peak of 22.0 (SD 6.2) ml/kg/min, which was 69% of that in controls (p < 0.001). All other cardiorespiratory responses, except VEpeak/VO2peak, were also lower in patients than in controls (Table III). CPET measurements showed that patients had a mean HRpeak of 154 (22), which reflected 90% of predicted HRmax. Mean RERpeak was 1.14 (0.08). Although patients exercised to acceptable cardiorespiratory limits, these values were lower than in controls, HRpeak = 165 (14); RERpeak = 1.22 (0.08).
|
Table III. Cardiorespiratory fitness in patients with aneurysmal subarachnoid haemorrhage (a-SAH) and control subjects |
||||
|
Patients post a-SAH (n = 28) Mean (SD) |
Controls (n = 28) Mean (SD) |
95% CI for the difference |
p-value |
|
|
VO2peak, ml/min |
1,636 (514) |
2,285 (561) |
–910.6 to –386.4 |
< 0.001** |
|
V̇O2peak, ml/kg/min |
22.0 (6.2) |
31.5 (6.2) |
–12.4 to –6.7 |
< 0.001** |
|
V̇O2peak, ml/kg FFM/min |
34.0 (8.7) |
45.9 (5.9) |
–15.5 to –8.3 |
< 0.001** |
|
VEpeak, l/min |
68.9 (24.7) |
90.1 (25.0) |
–33.8 to –9.4 |
0.001** |
|
VO2peak/HRpeak, ml/beat |
10.9 (2.5) |
13.9 (3.3) |
–3.8 to –1.4 |
< 0.001** |
|
POpeak, Watt |
130.7 (45.3) |
208.7 (55.0) |
–102.3 to –53.6 |
< 0.001** |
|
VEpeak/VO2peak, l/min |
42.1 (6.3) |
39.2 (5.3) |
–0.02 to 5.8 |
0.052 |
|
VAT, V̇O2 in ml/kg/min* |
15.2 (3.9) |
25.7 (8.2) |
–14.7 to –6.4 |
< 0.001** |
|
VAT, %V̇O2peak in ml/kg/min* |
69.0 (7.8) |
79.3 (7.5) |
–16.2 to –4.0 |
0.002** |
|
PO on VAT, Watt* |
79.0 (29.6) |
150.7 (53.7) |
–99.6 to –45.3 |
< 0.001** |
|
RRpeak (breaths/min) |
36.5 (8.1) |
36.8 (8.0) |
–4.8 to 4.1 |
0.872 |
|
HRpeak (bpm) |
154 (22) |
165 (14) |
–20.8 to –2.0 |
0.020 |
|
RERpeak (VCO2/VO2) |
1.14 (0.08) |
1.22 (0.08) |
–0.12 to –0.04 |
< 0.001** |
|
HRpeak % of predicted HRmax |
90 (12) |
96 (7) |
–11.7 to –1.2 |
0.018 |
|
*The ventilatory anaerobic threshold could be determined in 23 patients and was compared with their matched controls (n = 23). **Significantly different after Bonferroni correction to adjust for multiplicity: p = 0.05/14 = 0.004. VO2peak: peak oxygen consumption; VEpeak: peak minute ventilation; VO2peak/HRpeak: peak oxygen pulse; VCO2: carbon dioxide consumption; RER: respiratory exchange ratio; PO: power output; VAT: ventilatory anaerobic threshold; HRpeak: peak heart rate RR, respiratory rate; 95% CI: 95% confidence interval. |
||||
Cardiorespiratory fitness and fatigue
Table IV presents CPET outcome of fatigued and non-fatigued patients, and control subjects. In non-fatigued patients, 9 patients (56%) terminated the test because of dyspnoea and 7 (44%) because of muscular fatigue. In fatigued patients, half of the patients (n = 6) terminated the test because of dyspnoea and the other half because of muscular fatigue. Fatigued patients had a mean V̇O2peak of 19.4 (SD 4.1) ml/kg/min (63% of matched controls, p < 0.001) and non-fatigued patients had a mean V̇O2peak of 23.9 (6.9) ml/kg/min (74% of matched controls, p = 0.002). All other cardiorespiratory responses, except VEpeak in non-fatigued patients, were also lower in patients than in controls; fatigued patients performed at a level ranging from 52% to 70% of controls, and for non-fatigued patients this ranged from 70% to 84% (Table IV).
|
Table IV. Cardiorespiratory fitness in fatigued and non-fatigued patients with aneurysmal subarachnoid haemorrhage (a-SAH) and control subjects |
||||||||
|
Fatigued a-SAH (n = 12) Mean (SD) |
Controls (n = 12) Mean (SD) |
95% CI for the difference |
p-value |
Non-fatigued a-SAH (n = 16) Mean (SD) |
Controls (n = 16) Mean (SD) |
95% CI for the difference |
p-value |
|
|
VO2peak, ml/min |
1,365 (322) |
2,199 (391) |
–1,148.4 to –518.9 |
< 0.001* |
1,840 (544) |
2,349 (667) |
–827.4 to –191.9 |
0.004* |
|
V̇O2peak, ml/kg/min |
19.4 (4.1) |
30.8 (5.0) |
–13.4 to –9.4 |
< 0.001* |
23.9 (6.9) |
32.1 (6.9) |
–12.2 to –4.1 |
0.001 |
|
V̇O2peak, ml/kg FFM/min |
31.1 (6.5) |
46.3 (6.0) |
–19.4 to –11.1 |
< 0.001* |
36.4 (9.6) |
45.7 (6.0) |
–14.9 to –3.6 |
0.004* |
|
VEpeak, l/min |
54.3 (10.9) |
83.8 (18.2) |
–43.8 to –15.2 |
0.001* |
79.8 (26.7) |
94.8 (28.9) |
–30.5 to 0.39 |
0.055 |
|
VO2peak/HRpeak, ml/beat |
9.8 (2.0) |
13.5 (2.0) |
–4.6 to –1.5 |
0.002* |
11.5 (2.6) |
14.2 (4.0) |
–4.4 to –0.80 |
0.008 |
|
POpeak, Watt |
104.0 (26.7) |
200.2 (40.9) |
–123.5 to –68.8 |
< 0.001* |
150.8 (46.6) |
215.1 (64.1) |
–94.3 to –34.5 |
< 0.001* |
|
RRpeak, breaths/min |
34 (8) |
35 (6) |
–6.8 to 4.6 |
0.687 |
38 (8) |
38 (9) |
–6.5 to 6.9 |
0.954 |
|
HRpeak, bpm |
146 (19) |
163 (18) |
–34.7 to –0.77 |
0.042 |
159 (24) |
166 (11) |
–20.0 to 4.7 |
0.208 |
|
RERpeak, VCO2/VO2 |
1.13 (0.09) |
1.19 (0.07) |
0.01 to –0.12 |
0.070 |
1.15 (0.08) |
1.24 (0.07) |
–0.15 to –0.03 |
0.005 |
|
HRpeak, % of HRmax |
85.8 (10.8) |
95.0 (9.1) |
–19.8 to 0.19 |
0.054 |
92.2 (13.0) |
96.8 (5.6) |
–11.7 to 2.4 |
0.183 |
|
VO2peak: peak oxygen consumption; VEpeak: peak minute ventilation; HRpeak: peak heart rate; VO2peak/HRpeak: peak oxygen pulse; POpeak: peak power output; RR: respiratory rate; HRpeak: peak heart rate; RERpeak: peak respiratory exchange ratio; 95% CI: 95% confidence interval. *Significantly different after Bonferroni correction to adjust for multiplicity: p = 0.05/10 = 0.005. |
||||||||
DISCUSSION
To our knowledge, this is the first study to investigate cardiorespiratory responses to a progressive CPET in patients who have had an a-SAH. Cardiorespiratory fitness was significantly lower in patients compared with that of sex- and age-matched controls. Although we hypothesized that the cardiorespiratory fitness would be particularly impaired in fatigued patients, this study shows that VO2peak was also limited in non-fatigued patients. Furthermore, this study demonstrates that progressive CPET can be safely performed in a substantial group of patients after a-SAH if medical pre-screening procedures are implemented.
The present study shows impaired cardiorespiratory fitness after a-SAH; mean V̇O2peak was 69% of matched controls. Compared with patients with stroke not caused by an a-SAH, levels of cardiorespiratory fitness are somewhat higher (14). This can be explained by the fact that the patients in the present study had a relatively good functional outcome; there were no signs of paresis or spasticity. Therefore, they are more likely to preserve their cardiorespiratory fitness (29–31). Furthermore, premorbid physical inactivity, with reduced cardiorespiratory fitness, is a designated risk factor for stroke, whereas it is not for patients with a-SAH (32, 33).
Many factors could contribute to a limited VO2peak after a-SAH. Increased oxygen uptake by the muscle cells in response to physical effort requires increased pulmonary capacity and cardiac output. According to the present findings, patients showed both a lower VEpeak and a lower peak oxygen pulse. This may result from physical deconditioning, a mechanism that is supported by the fact that patients participated less in sports and/or moderate intensive physical activities.
Patients with a-SAH reached ventilatory anaerobic threshold (VAT) at earlier stages of CPET than controls. This implies that anaerobic oxidation, with subsequent accumulation of lactate in the blood, starts during milder physical effort. Because the perceived physical exertion at the VAT is found to be “heavy” (34), reaching the VAT at milder physical effort may impact on daily physical functioning. Although limitations in cardiorespiratory fitness affect daily physical functioning after stroke (15, 35) not caused by a-SAH, further research is needed to investigate its impact after a-SAH.
Although non-fatigued patients had lower V̇O2peak than controls (74% of controls), mean V̇O2peak seems to be more impaired in fatigued patients (63% of controls). This can be explained by the fact that fatigued patients may be more prone to initiate a degenerative cycle of physical deconditioning, in which the avoidance of physical activities reduces the cardiorespiratory fitness, which may further deteriorate fatigue complaints (36). Associations are reported between cardiorespiratory fitness and fatigue among several non-stroke patient groups (9–12). However, research on such relationships post-stroke is limited (8). Longitudinal studies are warranted to investigate the causal relationship between cardiorespiratory fitness and fatigue after a-SAH.
A recent report recommends exercise training as an integral component of stroke rehabilitation (15). The present study indicates that patients with a-SAH may also benefit from exercise training. Previous studies showed that exercise training can improve VO2peak by 9–23% after stroke (37, 38). Moreover, Zedlitz et al. reported on additional benefits of graded exercise training on post-stroke fatigue (16). Future intervention studies are warranted to investigate the beneficial effects of exercise training in patients after a-SAH.
The strength of the present study is the inclusion of a sex- and age-matched healthy comparison group. Patients and controls performed identical CPET measurements, which allowed better interpretation of CPET outcome. Including a comparison group is a commonly used method when studying cardiorespiratory responses in patients, including patients after stroke (39, 40).
Study limitations
Some possible limitations should be discussed. First, selection bias may have occurred. Patients were excluded when they were not eligible to perform CPET. Therefore, we might have excluded those with poor-grade a-SAH. Since these patients are likely to have worse outcome, this might have led to an underestimation of cardiorespiratory limitations. However, 18% of the study population had a poor-grade a-SAH (WFNS grade: IV–V), which is comparable to the occurrence of poor-grade a-SAH in the patient population; 18–24% of the a-SAH patients (41). It should be noted that more female patients were enrolled in the present study; only 8 male patients participated (29%). This can partly be explained by the fact that females have a higher risk of a-SAH than males (42); the incidence of a-SAH in the Netherlands is 7.6 for men and 11.2 for women per 100,000 persons per year (43). Because controls volunteered to participate, they might be more likely to take part in sports or moderate intensive daily physical activities. This might have led to an overestimation of the cardiorespiratory limitations after a-SAH. No appropriate norm data are available on participation in sports and/or moderate intensive daily physical activities. However, Dutch data on sports participation solely (75% of the Dutch population participates in sports activities) (44), suggest that our control group is representative for the Dutch population. Another limitation is that we do not know whether impaired cardiorespiratory fitness is a consequence of a-SAH or whether it was already present prior to the index event. However, premorbid physical inactivity and, inherently, impaired cardiorespiratory fitness is not a known risk factor for a-SAH. Considering the invasive CPET measurements, the present sample size is worth mentioning. However, the subdivision based on the presence of fatigue leads to smaller subgroups, which may be considered as a possible limitation. Because fatigue is one of the most distressing complaints, and cardiorespiratory fitness has not yet been investigated after a-SAH, we considered it important to differentiate between fatigued and non-fatigued patients.
In conclusion, cardiorespiratory fitness is limited in patients after a-SAH, both in fatigued and non-fatigued patients. These findings may indicate a physical component in fatigue after a-SAH. Longitudinal studies are warranted to investigate the course of fatigue and its physical determinants. Further research on interventions aiming at improving cardiorespiratory fitness after a-SAH seems necessary.
ACKNOWLEDGEMENT
This study was supported by the Dutch Brain Foundation.
REFERENCES
