Yen-Nung Lin, MD, MSc1,2, Chaur-Jong Hu, MD3,4, Ju-yang Chi, PT, MSc5, Li-Fong Lin, PT, MSc6,7, Tze-Hsun Yen, MD7, Yen-Kuang Lin, PhD8 and Tsan-Hon Liou, MD, PhD2,7
From the 1Department of Physical Medicine and Rehabilitation, Wan Fang Hospital, 2Graduate Institute of Injury Prevention and Control, 3Department of Neurology, Shuang Ho Hospital, 4Department of Neurology, Taipei Medical University, Taipei, 5Graduate Institute of Acupuncture Science, China Medical University, Taichung, 6Institute of Biomedical Engineering, National Yang Ming University, 7Department of Physical Medicine and Rehabilitation, Shuang Ho Hospital and 8Graduate Institute of Nursing, College of Nursing, Taipei Medical University, Taipei, Taiwan
OBJECTIVE: To evaluate the effects of repetitive transcranial magnetic stimulation (rTMS) on mobility among patients with substantial leg impairment after subacute stroke.
DESIGN: Double-blinded, stratified, randomized trial involving a sham control group.
PARTICIPANTS: Patients who developed unilateral hemiplegia after first-ever subacute stroke and underwent inpatient stroke rehabilitation.
METHODS: The 15-day intervention programme used in the present study included the application of rTMS (1 Hz, 15 min) over the leg motor area of the unaffected hemisphere, followed by 45 min physical therapy. Overall, 32 participants were randomly assigned to receive either real rTMS or sham stimulation. Clinical assessments, including the Postural Assessment Scale for Stroke Patients (PASS), balance subscale of the Performance Oriented Mobility Assessment (POMA-b), Fugl-Meyer Assessment, Barthel Index (BI), and Timed Up and Go test, were performed immediately before and after the intervention.
RESULTS: Both groups demonstrated significant improvements in all the test results over time. At the post-test assessment, the patients in the real rTMS group demonstrated greater improvements in the PASS, POMA, and BI scores than did the patients in the sham rTMS group. In addition, a significantly higher number of patients in the real rTMS group regained mobility at the post-test assessment compared with the corresponding number of patients in the sham rTMS group.
CONCLUSION: Application of 1-Hz rTMS may improve mobility among patients with substantial leg impairment after subacute stroke.
Key words: stroke; repetitive transcranial magnetic stimulation; balance; mobility.
J Rehabil Med 2015; 47: 305–310
Correspondence address: Tsan-Hon Liou, Graduate Institute of Injury Prevention and Control, Taipei Medical University, 250 Wu-Hsing Street, Taipei 110, Taiwan. E-mail: peter_liou@s.tmu.edu.tw
Accepted Dec 23, 2014; Epub ahead of print Feb 13, 2015
INTRODUCTION
Application of repetitive transcranial magnetic stimulation (rTMS) to facilitate neural plasticity during stroke treatment has recently gained considerable attention. Numerous clinical studies have investigated the safety and efficacy of using rTMS to treat various stroke symptoms (e.g. hemiplegia, dysphagia, aphasia, and hemi-neglect), and certain studies have also reported promising results (1–5). The possible mechanism through which rTMS acts is based on the interhemispheric competition (IHC) model (6), which explains that patients with stroke experience alterations in cortical excitability and exhibit abnormally high interhemispheric inhibition from the unaffected hemisphere to the affected hemisphere (7, 8). Thus, purposeful modulation of cortical excitability between the hemispheres might optimize neural reorganization. Studies have advocated that the hemispheric activity can be rebalanced by applying high-frequency rTMS over the motor cortex of the affected hemisphere (M1-AH) to enhance the excitability of the affected hemisphere (9–12) or by applying low-frequency rTMS over the motor cortex of the unaffected hemisphere (M1-UH) to inhibit the excitability of the unaffected hemisphere (13–15).
Several rTMS studies have evaluated the upper extremity dysfunction following stroke (2, 16), but few studies have explored the efficacy of applying rTMS on the lower extremities. This discrepancy may be due to the deep location of the leg representation in the motor cortex, which is difficult to target using transcranial magnetic stimulation (TMS); however, Wang et al. suggested that the application of 1-Hz rTMS to the leg representation of M1-UH (M1-UH-Leg) may improve the gait patterns of patients with chronic stroke (17). These results are encouraging despite the small sample size of the study. However, limited information is available regarding the efficacy of rTMS application to treat leg impairment, particularly after subacute stroke.
The establishment of mobility (e.g. stand-up, transfer between bed and chair, and walk) relies on the coordination of muscles in the leg and trunk to achieve a dynamic balance. As in a chain of weight-bearing, the leg is an essential part of upright postural control. Impaired control of a paretic leg increases postural sway, disrupts equilibrium reactions, and impairs postural adjustments during standing and walking. The present study focused on patients who were unable to walk independently after subacute stroke because of substantial leg impairment. Among this patient population, restoration of mobility (e.g. mobility in bed, sit-to-stand motion, walk, and transfer between bed and chair) as soon as possible is the primary goal in an inpatient rehabilitation setting. Therefore, this pilot study explored the potential treatment effects of rTMS on the mobility of patients who sustained substantial leg impairment. Because the knee extensor is a major muscle involved in weight-bearing during upright activities, 1 Hz of rTMS was applied to the quadriceps representation of M1-UH to inhibit its over-excitability and improve functioning of the paretic leg.
METHODS
Participants
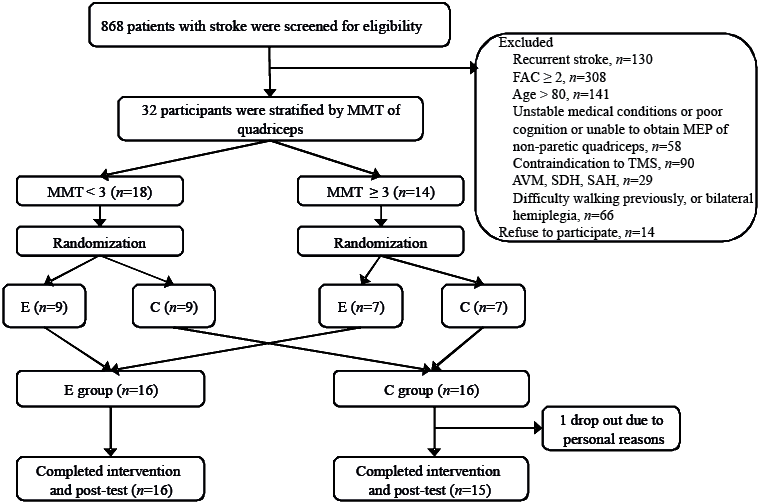
All patients hospitalized for stroke treatment and rehabilitation at a university-affiliated teaching hospital were screened for eligibility during the period from January 2013 to December 2013. Inclusion criteria were: patients presenting with unilateral hemiplegia caused by a first-ever stroke (either infarction or haemorrhage) that occurred 10–90 days earlier; aged 18–80 years; and exhibiting substantial leg impairment, indicated by a functional ambulation classification (FAC) score of 0 to 1 (18). The FAC classifies the ability of ambulation as FAC 0 to FAC 5, wherein FAC 0 indicates non-functional ambulation of a patient and FAC 1 indicates that the patient requires constant manual contact to support the body weight as well as maintain balance during walking. A minimal level of comprehension was required to understand the instructions during the TMS and functional assessments. Therefore, patients who were unable to follow 1-step orders (e.g. “raise your leg,” “extend your knee,” and “stand-up”) were not included. Moreover, patients with a pacemaker, seizure history, intracranial haemorrhage associated with a tumour or arteriovenous malformation, or craniotomy, those who completed a Timed Up and Go (TUG) test (19) within 2 min, or those who had sustained abnormal gait patterns (e.g. limping, unsteady gait, and slow movements) before the stroke were excluded from the study. Furthermore, patients who exhibited lack of motor evoked potentials (MEPs) of the non-paretic leg in the pre-test assessment were excluded.
Study design
Motor deficit severity is a critical predictor for motor recovery after a stroke (20). To ensure an even distribution of this factor between the experimental (Group E) and the control (Group C) groups, the study participants were divided into 2 subgroups according to the muscle strength of their paretic quadriceps based on a Grade 3 cut-off on a manual muscle test (MMT). The grading of muscle power may be subjective, and the synergic contractions of other muscles may interfere in the same; therefore, we extended this definition. When a patient could extend the knee up to 40° from a flexion of 90° in a sitting posture, with both hands placed on both knees and no back support, and could maintain this position for > 10 s, the muscle power of their quadriceps was graded as ≥ 3. The patients in each subgroup were then randomly allocated using a 1:1 ratio (with a block of 2) into Group E or C (Fig. 1). This procedure was designed to increase the power to detect treatment effects, particularly for small samples including a heterogeneous study population (21). The researcher who conducted the randomization subsequently performed the rTMS application. The research protocol was reviewed and approved by the Institutional Review Board of Taipei Medical University and each participant provided written informed consent.
Transcranial magnetic stimulation procedure
During the examination, the participants sat in a wheelchair with a head-rest and leg rests to keep their heads stabilized, with their knees flexed at 45°. Surface electromyography (EMG) electrodes (silver–silver chloride; Ag-AgCl) were placed on the rectus femoris. An active recorder was placed on the midpoint between the anterior superior iliac spine and the patella. A reference recorder was placed on the superior border of the patella. The EMG measured the MEPs of the rectus femoris in response to the TMS delivered using a Magstim Rapid2 stimulator (Magstim Co. Ltd, Carmarthenshire, UK) with a 70-mm figure-8 coil (maximum power 2.2 T) over the contralateral M1. MEP measurement was performed with pre-activation by asking the patient to contract the quadriceps isometrically using 10–20% force of the maximal voluntary isometric contraction. The intensity was initially set at 100% of the machine output (MO) to determine the optimal stimulation site (hotspot). We slowly moved the coil over the scalp (e.g. 5 mm) and stimulated the M1-UH every 5–8 s. The hotspot was located at the site that demonstrated the greatest MEP. Subsequently, we decreased the intensity in a stepwise manner while stimulating the hotspot. The motor threshold (MT; expressed as %MO) was determined by observing the MEPs > 100 μV of the peak-to-peak amplitude in 5 out of 10 responses under the required minimal intensity. The hotspot was marked on the scalp with oil ink and recorded as x, y, in centimetres from the vertex (cz).
Interventions
Interventions were initiated on the weekday following the pre-test and were performed daily for 15 consecutive weekdays. Based on suggestions by Avenanti et al. that a priming physical therapy (PT) with inhibitory rTMS may boost use-dependent plasticity (22), the rTMS and PT sessions were arranged in a time-locked manner. Thus, the participants in Groups E and C received real rTMS and sham rTMS, respectively (1 Hz, 15 min), immediately followed by a 45-min PT session. The rTMS was delivered to the scalp over the hotspot using the Magstim Rapid2 and a 70-mm figure-8 coil. One rTMS session included 900 pulses at an intensity of 130% MT measured using the 70-mm figure-8 coil. When the planned intensity was stronger than 80% MO, an intensity of 80% MO was used. The sham rTMS was delivered using a customized sham coil that produced similar sounds to those produced by a real coil. The PT session, which involved transfer, balance, and ambulation training, was individually tailored to the current functional status of each patient. The physical therapists were blinded to the treatment assignment.
Measurements
Information regarding stroke was obtained using chart and image reports. The baseline assessments for stroke severity included MMT, Brunnstrom stage, and modified Rankin Scale (mRS). The Postural Assessment Scale for Stroke Patients (PASS), which assesses the ability of a patient to maintain or change a given posture (e.g. lying, sitting and standing), was the primary outcome (23). This instrument, which scored between 0 and 36 points, exhibits strong validity and reliability at various stages of stroke recovery and is sensitive to functional changes in both stationary and dynamic balance in patients with severe stroke at early stages after stroke (24). Other outcomes included the balance subscale of the Tinetti Performance-Oriented Mobility Assessment (POMA-b, 0–16 points), which measures the static and dynamic balance in sitting and standing (25) and the TUG, which measures the ability to perform advanced mobility tasks, such as walking and turning (19). In addition, the Barthel Index (BI; 0–100 points) was used to measure the ADL independence. Moreover, the lower-extremity (LE) subscale of the Fugl-Meyer Assessment (FMA-LE; 0–34 points) was used to measure neurological recovery of LE (26). An assessor, who was blind to the intervention assignment, performed all the pre-test and post-test clinical measurements. Any side-effects or discomfort reported during the rTMS sessions were investigated and recorded.
Statistical analysis
All continuous variables were tested using the Kolmogorov–Smirnov statistic, and the results exhibited a normal distribution. The intra-group temporal differences were evaluated using a paired t-test. The intergroup post-test differences were assessed using an analysis of covariance (ANCOVA) with the pre-test data as the covariance. The intergroup comparison of the categorical variables was assessed using χ2 analysis. Optimal mobility improvement was defined as the capability to finish the TUG test within 2 min. The number of patients who achieved optimal mobility improvements was compared between groups with a χ2 test. A 2-tailed p < 0.05 was considered as the level of significance. Intention-to-treat analysis was used. All analyses were performed using SPSS statistical package, Version 17.0.
RESULTS
A total of 32 patients were enrolled in this study; of these, 31 completed the interventions and post-test assessments (Fig. 1). One patient was transferred to another hospital for personal reasons before treatment completion. No significant differences were observed in baseline characteristics between Groups E and C (Table I). A Group E patient reported dizziness, and a Group C patient reported tingling scalp pain after the intervention sessions. These symptoms were tolerable and subsided after the treatment sessions.
|
Table I. Participant characteristics |
|||
|
Group E (n = 16) |
Group C (n = 16) |
p-value |
|
|
Age, years, mean (SD) |
58.3 (10.8) |
62.3 (11.7) |
0.323 |
|
Time since stroke, days, mean (SD) |
40.6 (29.1) |
33.5 (23.8) |
0.458 |
|
Education, years, mean (SD) |
8.6 (5.8) |
9.3 (3.9) |
0.71 |
|
mRS, mean (SD) |
4.6 (0.5) |
4.6 (0.5) |
0.729 |
|
MMT of paretic quadriceps, mean (SD) |
2.4 (1.2) |
2.5 (1.0) |
0.748 |
|
Brunnstrom stage of paretic leg, mean (SD) |
3.0 (0.9) |
3.2 (0.8) |
0.544 |
|
Gender, male/female, n |
10/6 |
11/5 |
1.00 |
|
Diabetes, yes/no, n |
4/12 |
6/10 |
0.704 |
|
Side of lesion, left/right, n |
10/6 |
7/9 |
0.479 |
|
Stroke type, infarct/haemorrhage, n |
12/4 |
10/6 |
0.704 |
|
Location of lesion, cortical/subcortical and deep structures, n |
5/11 |
3/13 |
0.685 |
|
FAC 0/FAC 1, n |
6/10 |
5/11 |
1.00 |
|
SD: standard deviation; mRS: modified Rankin scale; MMT: manual muscle test; E: experimental and C, control groups; FAC: Functional Ambulation Classification. FAC 0 indicated non-functional ambulation and FAC 1 indicated constant manual contact was required to support body weight during walking. |
|||
Table II presents the clinical assessment results. No significant differences were observed in the pre-test parameters between both groups. All test results demonstrated significant post-test improvements in each group. In addition, the post-test assessment revealed significant intergroup differences in the PASS, POMA-b, and BI measurements (p < 0.05). At the pre-test, all the participants were unable to finish the TUG test within 2 min. At the post-test, 11 Group E patients and 4 Group C patients could complete the TUG test within 2 min; this intergroup difference was significant (p = 0.013).
|
Table II. Clinical measurement changes after intervention |
||||
|
Pre-test Median (IQR) |
Post-test Median (IQR) |
Change Median (IQR) |
p-value for the intergroup difference |
|
|
PASS |
||||
|
Group E |
19.0 (9.5) |
28.5 (11.3) |
7.5 (5.5)*** |
0.001 |
|
Group C |
17.5 (10.5) |
24.0 (10.0) |
5.0 (5.0)** |
|
|
POMA-b |
||||
|
Group E |
2.0 (5.0) |
9.5 (8.0) |
4.0 (5.0)*** |
0.043 |
|
Group C |
1.0 (4.0) |
4.0 (7.0) |
1.0 (3.0)** |
|
|
BI |
||||
|
Group E |
40.0 (16.3) |
50.0 (22.5) |
15.0 (18.8)*** |
0.015 |
|
Group C |
32.5 (30.0) |
40.0 (20.0) |
5.0 (15.0)** |
|
|
FMA-LE |
||||
|
Group E |
14.5 (6.8) |
21.0 (6.8) |
3.5 (3.8)*** |
0.459 |
|
Group C |
15.0 (3.8) |
20.0 (8.0) |
3.5 (3.8)* |
|
|
PASS: Postural Assessment Scale for Stroke Patients; POMA-b: balance subscale of the Tinetti Performance Oriented Mobility Assessment; BI: Barthel Index; FMA-LE: lower extremity subscale of the Fugl–Meyer Assessment; E: experimental; C: control groups. Intragroup differences were assessed using a paired t-test: *p < 0.05; **p < 0.05; ***p < 0.001. Intergroup differences at the post-test assessment were assessed using analysis of covariance (ANCOVA) with adjustments for the corresponding pre-test values. |
||||
DISCUSSION
The results of this study add evidence to currently available information regarding the treatment effects of rTMS on lower-extremity disability among patients with subacute stroke. Based on relevant literature review, this is the first study to explore the effects of rTMS application on mobility among subacute stroke patients who exhibit substantial leg impairment. The results indicate that the application of 1-Hz rTMS exerted beneficial effects on the balance, mobility, and ADL independence of these patients. Furthermore, 1-Hz rTMS is a safe and practical option for treating stroke in an inpatient rehabilitation setting. These findings are crucial because mobility is the mainstay of further functional achievements. Restoration of these functions as soon as possible is the primary goal of inpatient rehabilitation.
Determination of the treatment effects of an intervention among patients with subacute stroke may be challenging because of the considerable variations in the prognosis of this population. The effects of other prognostic factors (e.g. stroke severity, type, lesion location, and cognitive function) could be greater than those of the intervention, which would thereby mask the treatment effects. Careful control of the severity of the initial motor deficit may increase the power to detect the treatment effects of the interventions administered. After controlling for initial quadriceps muscle strength, the results indicated that rTMS significantly affected the post-test PASS, BI and TUG results, and marginally affected the post-test POMA results. These findings demonstrated the beneficial treatment effects of rTMS on mobility, which may contribute to improved ADL independence.
Walking is a crucial component of mobility functions. However, to assess pre-test and post-test improvement in walking using quantitative parameters (e.g. walking speed, distance and time) was not possible in this study because all the participants were unable to walk at the pre-test assessment. Therefore, the ability to walk was categorized as “able” or “unable” based on the ability to complete the TUG within 2 min. Thus, all the participants were “unable” to walk at the pre-test assessment, and a significantly higher number of Group E patients were “able” to walk than the Group C patients at the post-test assessment, indicating that a higher number of patients in Group E regained walking ability.
Although both groups exhibited significantly improved FMA-LE scores, indicating LE motor recovery, the results were not significantly different between both groups. The lack of positive neurological results measured using the FMA-LE could obfuscate the mechanisms of functional improvement caused by rTMS. However, one possible explanation might be considered: the FMA measures neurological recovery based on Brunnstrom’s concept of sequential stages of neurological motor recovery in patients with hemiplegic stroke. Therefore, this impairment-based tool can be insensitive to functional improvement (26).
Limited information is available regarding the TMS-related neurophysiology in the leg representation of M1 and, to date, insufficient evidence is available to support the notion of IHC in the leg representation of M1; however, studies based on the IHC model have continued to report positive treatment effects on leg impairment (17, 27). The rationale of applying 1-Hz rTMS was to inhibit the overexcitability of M1-UH through interhemispheric interactions. Wang et al. first applied 1-Hz rTMS to improve gait among patients with chronic stroke; the patients exhibited improved functional performance after rTMS, and the corresponding electrophysiological findings provided further indirect evidence that supported the IHC model. In their study, the excitability of M1-AH-Leg and M1-UH-Leg seemed to be increased and decreased after rTMS, respectively (17). The present study followed the same hypothesis and demonstrated the short-term effects of rTMS on mobility. These findings may strengthen the evidence supporting the use of 1-Hz rTMS on leg impairment after stroke and indicate the feasibility of applying the IHC model to lower extremities.
In the original study protocol, we planned to use 130% MT over the quadriceps representation of M1-UH. However, after conducting a small study involving 3 participants to test the protocol, we determined that the MT of M1-UH measured using a figure-8 coil was relatively high (i.e. 65%, 65% and 90% MO). Therefore, adoption of the original protocol could have exposed certain participants to extremely high rTMS levels, thus causing both potential safety concerns and practical problems when an intensity of > 100% MO was required. Therefore, the upper intensity limit was set at 80% MO. In addition, the averaged MT of M1-UH-leg measured using the figure-8 coil was 65.4% MO. This was considerably higher than was measured using the double-cone coil in an earlier study by Wheaton et al. (28). They demonstrated an averaged MT of M1-UH-leg at 45.7% MO. The double-cone coil, which can generate a higher electrical field intensity is expected to be superior to the figure-8 coil in the leg motor area stimulation (29). A 70-mm figure-8 coil was used in the present study because this coil is currently the most popular TMS coil worldwide. The double-cone coil was used recently in a novel procedure attempting to improve gait in patients with motor disorders (30). Therefore, additional randomized controlled trials must be conducted to evaluate the applications of this coil in patients with leg impairment.
This study has 4 limitations. First, neurophysiological measurements (e.g. MEP) were not included in the outcome measures. Without these measurements, it cannot be validated whether the leg motor area of the unaffected hemisphere rather than the affected hemisphere was selectively stimulated. Secondly, the MT, which was expected to change during the intervention period, was not measured at every rTMS session. The lack of adjustment of the stimulation intensity based on the MT changes might underestimate the treatment effects of rTMS. Thirdly, the sample size was small, with no long-term follow-up records. However, this pilot study was designed to explore the potential benefits of rTMS on mobility, and further studies with larger samples and long-term follow-up are required to confirm the results of this study. Fourthly, the PT was customized for each individual patient. Thus, the extent of similarity between the therapies administered to both groups, as well as the contribution of this factor to the present results, remains unclear.
In conclusion, this pilot study demonstrated that 1-Hz rTMS, applied in 15 daily sessions over M1-UH, yielded potential beneficial effects on the mobility and ADL independence of patients with substantial leg impairment after subacute stroke. Further research is required to confirm the results of this study and identify the long-term effects of rTMS.
ACKNOWLEDGEMENTS
This study was supported by a grant (102-wf-eva-04) from the Wan-Fang Hospital, Taipei Medical University, Taiwan.
ClinicalTrials.gov Identifier: NCT02006875.
REFERENCES
