Anna Bjerkefors, PhD1–4, Jordan W. Squair, MSc1, Romeo Chua, PhD1,2, Tania Lam, PhD1,2, Zhen Chen, PhD1,2 and Mark G. Carpenter, PhD1,2
From the 1School of Kinesiology, and 2International Collaboration of Repair Discoveries, University of British Columbia, Vancouver, BC, Canada, 3Department of Neuroscience, Karolinska Institute and 4The Swedish School of Sport and Health Sciences (GIH), Stockholm, Sweden
OBJECTIVE: To use transcranial magnetic stimulation and electromyography to assess the potential for preserved function in the abdominal muscles in individuals classified with motor-complete spinal cord injury above T6.
SUBJECTS: Five individuals with spinal cord injury (C5–T3) and 5 able-bodied individuals.
METHODS: Transcranial magnetic stimulation was delivered over the abdominal region of primary motor cortex during resting and sub-maximal (or attempted) contractions. Surface electromyography was used to record motor-evoked potentials as well as maximal voluntary (or attempted) contractions in the abdominal muscles and the diaphragm.
RESULTS: Responses to transcranial magnetic stimulation in the abdominal muscles occurred in all spinal cord injury subjects. Latencies of muscle response onsets were similar in both groups; however, peak-to-peak amplitudes were smaller in the spinal cord injury group. During maximal voluntary (or attempted) contractions all spinal cord injury subjects were able to elicit electromyography activity above resting levels in more than one abdominal muscle across tasks.
CONCLUSION: Individuals with motor-complete spinal cord injury above T6 were able to activate abdominal muscles in response to transcranial magnetic stimulation and during maximal voluntary (or attempted) contractions. The activation was induced directly through corticospinal pathways, and not indirectly by stretch reflex activations of the diaphragm. Transcranial magnetic stimulation and electromyography measurements provide a useful method to assess motor preservation of abdominal muscles in persons with spinal cord injury.
Key words: corticospinal pathways; diaphragm; motor cortex; motor-evoked potentials; thoracic segments.
J Rehabil Med 2015; 47: 138–146
Correspondence address: Anna Bjerkefors, Biomechanics and Motor Control Laboratory, The Swedish School of Sport and Health Sciences (GIH), Box 5626, SE-114 86 Stockholm, Sweden. E-mail: anna.bjerkefors@gih.se
Accepted Jul 7, 2014; Epub ahead of print Dec 9, 2014
Introduction
The International Standards for Neurological Classification of Spinal Cord Injury (ISNCSCI) is the current gold standard used to classify the neurological level and completeness of a spinal cord injury (SCI) (1). Notably, motor examination does not include the trunk muscles. This makes it difficult to draw conclusions about the completeness of the neurological lesion in the thoracic segments, as clinicians rely solely on sensory examination at these levels. This severely limits the potential for accurate classification, diagnosis and effective rehabilitation of individuals with thoracic SCI, in whom even minor improvements in trunk muscle function can translate into significant increases in functional capacity and balance control (2, 3).
There is some indirect evidence for preserved function below the injury level, which has not been detected by the ISNCSCI. In a case study by Bjerkefors et al. (4), intramuscular electromyography (EMG) recordings were used to identify preserved motor function in the abdominal muscles below the injury level in a person classified as having a motor-complete (American Spinal Injury Association Impairment Scale (AIS) A) SCI at T3. Activation was elicited during voluntary efforts and in reaction to balance perturbations. This could indicate that there is some preserved abdominal muscle function despite the conclusions of the clinical examination. However, the case study could not rule out whether the activation of lower trunk muscles was induced indirectly by activation of other muscles (e.g. the upper trunk muscles), or caused by bulging of the abdomen induced by a contraction of the diaphragm (Dia).
One potential non-invasive neurophysiological method to study preserved motor tracts below the injury level is to use transcranial magnetic stimulation (TMS). TMS delivered over the primary motor cortex has been used previously for investigating corticospinal pathways in people with SCI (5–8). In the majority of these studies, the EMG responses to TMS (i.e. motor evoked potentials, MEPs) have been recorded from limb muscles innervated by cervical and lumbosacral spinal segments. In muscles innervated from thoracic spinal cord segments, MEP responses have been limited to paravertebral back muscles (6, 7), in which the responses could be explained by multi-segmental innervations of the erector spinae muscles and long muscle fibre conduction (6). Although MEPs have been evoked in the abdominal muscles of able-bodied subjects (9–16), there are no studies, to our knowledge, that have assessed the responses to TMS in the abdominal muscles of individuals with SCI. The advantage of focusing on abdominal muscles is that they receive their innervations from a limited number of segments in the caudal portion of the thoracic spinal cord (T6–T12) (17), making them an ideal candidate for testing of preserved motor function in high thoracic SCI.
The primary aim of this study was to examine possible preservation of motor function below the level of injury using TMS to elicit MEPs in abdominal muscles of individuals classified with motor-complete SCI above the T6 level, and matched able-bodied individuals, during rest and attempted voluntary sub-maximal contractions. The secondary aim of this study was to use surface EMG to measure activation of abdominal muscles during attempted maximal voluntary contractions in the same SCI and able-bodied individuals. Based on previous findings (4, 6, 7) it was hypothesized that MEPs would be elicited by TMS in the abdominal muscles of individuals classified with motor-complete SCI above T6, with similar latencies and patterns as controls. In subjects in whom MEPs are detected, it was hypothesized that abdominal muscle activity would also be detected by surface EMG during attempted voluntary maximal contractions with similar patterns as controls.
Methods
Subjects
Five individuals (2 men, 3 women; mean age 38 years (standard deviation (SD) 3.3); weight 64.4 kg (SD 5.6); height 1.73 m (SD 0.01)) with chronic motor-complete SCI volunteered for the study. The inclusion criteria for participants with SCI were: motor-complete SCI above T6-level at least one year previously, and stable neurological and medical status with no cognitive impairments. Individuals with SCI were excluded if they had: frequent experience of autonomic dysreflexia, severe spasticity, personal history of epilepsy/seizure, or disturbances of the nervous system other than the SCI. A detailed description of the participating individuals is shown in Table I. In addition, able-bodied, age-matched controls volunteered for the study (2 men, 3 women; mean age 32 years (SD 8.7); weight 67.5 kg (SD 7.7); height 1.73 m (SD 0.01)). Exclusion criteria for all participants included any of the following contra-indications for TMS: recurring or severe headaches, skull fracture or head injury including concussion, head or brain surgery, hearing problems, psychiatric impairment and/or sleep deprivation, pregnancy, heart disease, diabetes and electrodes implanted in the central or peripheral nervous system (18). All participants received oral and written information describing the study and signed written consent to voluntarily participate. The study was approved by the University of British Columbia (UBC) Clinical Research Ethics Board.
|
Table I. Detailed description of subjects with spinal cord injury and able-bodied subjects |
||||||||||||||||
|
Subject |
Agea |
Gendera |
Heighta m |
Weighta kg |
Lesion level |
Years post-injury |
Sensory score |
Total motor score |
AIS |
ZPP R/L |
Traumatic injury |
Spasticity |
Negative impact on ADL |
Medication |
||
|
Pin-prick |
Light touch |
|||||||||||||||
|
1 |
35 (46) |
M (M) |
1.83 (1.80) |
72 (70) |
C5 |
18 |
64 |
64 |
25 |
B |
– |
Yes |
Yes |
Yes |
Yes |
|
|
2 |
38 (26) |
M (M) |
1.80 (1.74) |
65 (75) |
C7 |
14 |
62 |
62 |
50 |
B |
– |
Yes |
Yes |
No |
No |
|
|
3 |
39 (24) |
F (F) |
1.80 (1.80) |
58 (74) |
C6 |
16 |
25 |
25 |
31 |
A |
C8/C8 |
Yes |
Yes |
No |
No |
|
|
4 |
35 (30) |
F (F) |
1.55 (1.57) |
60 (57) |
T3 |
21 |
44 |
42 |
50 |
A |
T4/T4 |
Yes |
Yes |
No |
No |
|
|
5 |
43 (33) |
F (F) |
1.65 (1.65) |
67 (62) |
T3 |
27 |
42 |
44 |
50 |
A |
T4/T5 |
Yes |
Yes |
No |
No |
|
|
aNumbers in brackets indicate the value for the matched able-bodied subject. Maximal values in the sensory (112) and motor (100) examination represent the least impaired function. M: male; F: female; AIS: American Spinal Injury Association Impairment Scale; A: complete impairment, i.e. no sensory or motor function in the S2–S4 segment; B: sensory incomplete, no motor function below the neurological level and includes the sacral segments (Marino et al., 19); ZPP R/L: zone of partial preservation, on the right and left side, most caudal level with any innervation (in complete injuries only); Negative impact on ADL: negative impact on activities of daily living due to spasticity; Medication: medication taken to modulate the spasticity. |
||||||||||||||||
Experimental design and procedure
Clinical classification of the spinal cord injury (Day 1). The neurological lesion level, completeness of the injury, and zone of partial preservation of the SCI was assessed by a physician/PhD experienced in using the ISNCSCI (19).
Transcranial magnetic stimulation (Day 2). Subjects lay in a supine position on a plinth with their arms folded and hips and knees bent. Subjects wore a cap marked with coordinates 1 cm apart and the location of the vertex (CZ) was identified using the international 10/20 system. Magnetic stimulation was applied over the scalp site using a MagStim 200 stimulator, Mono Pulse (The MagStim Company Ltd, Dyfed, UK) connected to a stimulating coil (figure-of-eight, outer wing diameter 10 cm). The coil was held tangentially to the scalp, approximately 45° from the mid-sagittal plane, and the central section of the coil was placed over the stimulation site. To define the location of the point of optimal excitability (POE) at the primary motor cortex, the stimulation started over the area responsible for activation of the abdominal muscles (i.e. approximately 2 cm lateral and 2 cm anterior to the vertex) (10, 12, 13). The stimulus intensity was initially set to 50% maximal stimulator output (MSO), and then increased to 70–100% MSO while the orientation of the coil and the location were slightly adjusted until the POE was localized and identifiable MEPs were recorded (14). Consistent with previous reports, the POE was easier to identify if the subject maintained a gentle sub-maximal (or attempted) contraction of the abdominal muscles during stimulation, as the MEP amplitude has been shown to increase by facilitation (20). The number of stimuli, the POE, and the % MSO were documented for all subjects. The POE on the left and right hemisphere was marked on the cap and served as a visual reference for reliable coil positioning.
Subjects lay supine with their arms crossed over their chest and the trunk secured with a strap placed over the chest to minimize movements of the upper body. Subjects were then instructed to perform sub-maximal voluntary (or attempted) contractions during trunk flexion (fingertips to the knees without moving your head) and trunk lateral bending to the contralateral side (opposite to the stimulated hemisphere) (fingertips to the contralateral knee without moving your head). To control for changes in intra-abdominal pressure, participants’ breathing pattern was matched to a metronome, with a time interval set to produce 60 beats per minute (bpm): breathe out (2 s), breathe in (2 s), breathe out (2 s) and gently contract your abdominal muscles without moving your head (2 s). The stimulation was applied during the second normal exhalation with simultaneous static sub-maximal contraction (21). The protocol was initially performed on the dominant hemisphere (i.e. the side responsible for the innervation of the dominant hand). Ten stimuli were delivered while subjects performed trunk lateral bending and trunk flexion using the same stimulation intensity as when the POE was defined. A 30 s rest was given between trials and a 2 min break between tasks. Finally, 10 stimuli were delivered at rest using the same stimulation output as above, separated by approximately 10 s. The protocol was then repeated on the non-dominant hemisphere.
Voluntary (or attempted) activation (Day 3). Subjects performed 7 different trunk muscle tasks while lying supine on a plinth: trunk flexion, trunk rotation to the left and right, trunk lateral flexion to the left and right, hollowing manoeuvre, and Valsalva manoeuvre (21). The trunk was secured to a plinth with a strap placed over the chest to minimize movements of the upper body and subjects were instructed to fold their arms. Each task was preceded by a verbal explanation by the examiner. The contraction was performed during normal exhalation, and the following instruction was given: breathe out (2 s), breathe in (2 s), breathe out (2 s), and maximally contract your abdominal muscles (2 s). The time interval was set to produce 60 bpm. Subjects practiced coordinating the trunk muscle task with the instruction to breathe until they were able to perform the task in a satisfactory manner, as judged by the examiner. Subjects performed 2 trials for each task with a 30 s rest between trials. A 2 min break was given between tasks. Only data from the trunk flexion and trunk lateral flexion tasks were analysed for the purposes of this study.
Data collection and processing
Electromyography. Muscle activity was recorded bilaterally with surface EMG from: rectus abdominis (RA), obliquus externus (OE), transversus abdominis/obliquus internus (TrA/OI), and Dia. The diaphragm was included because it has the potential to influence abdominal muscle activity indirectly through changes in intra-abdominal pressure. Prior to placing the electrodes, the skin was cleaned with alcohol and, if needed, shaved. Pairs of electrodes (10 mm diameter, Kendall, Tyco Healthcare Group LP, MA, USA) were attached with approximately 2 cm inter-electrode separation. Electrode positions were as follows: RA: approximately 3 cm lateral and 2 cm caudal to the umbilicus; OE: approximately 2 cm below the lowest point of the rib cage; TrA/OI: approximately 2 cm medial to the anterior superior iliac spine; Dia: 1 in the intercostal space between the 8th and the 7th rib in the midclavicular line, and 1 on the 7th rib with a slight lateral position (22). A ground electrode was placed over the elbow. EMG data were collected at 1,500 Hz, amplified (500 times), and band-pass filtered between 10 and 500 Hz (Telemyo 2400R, Noraxon, Scottsdale, USA), prior to digital sampling at 1,000 Hz (Power 1401, Cambridge Electronic Design, Cambridge, UK). EMG-processing was performed off-line using customized scripts developed within commercially available software (Spike2, Cambridge Electronic Design, Cambridge, UK). The TMS was triggered using a digital pulse from a CED Power 1401 and Spike2 data collection software.
Analysis
MEP onset was calculated as the time at which the MEP exceeded 2 SD above the mean baseline rectified EMG activity measured (100 ms prior to the stimulation onset) and remained beyond this threshold for at least 2 ms. MEP amplitude was calculated as the peak-to-peak amplitude of the raw EMG activity after stimulation onset. All onset times were confirmed visually by the same experimenter, and were adjusted manually in 9.6% of all trials. MEP onsets detectable in at least 5 out of 10 trials for each muscle were defined as “present” and used to calculate the mean values of individual subject.
EMG recorded during voluntary tasks was high-pass filtered at 30 Hz to remove heart rate artefacts (23) and used to calculate the root mean square (RMS) over a 500 ms time period for each muscle and task during rest and voluntary contraction. Both values were taken during the end of normal exhalation to control for changes in intra-abdominal pressure. If the mean RMS of the 2 contraction trials for a given muscle and task exceeded 2 SD above the mean resting value (calculated from all trials), the value was defined as “present” and was included in the frequency of response analysis (3). All descriptive data are presented as mean and SD.
Results
One participant (C4) completed TMS only on the right hemisphere. All other subjects completed the protocol and were free from headaches or other negative side-effects due to the stimulation. The % MSO ranged from 50 to 100 in both SCI and able-bodied subjects. These values are similar to previously reported levels for abdominal activation in able-bodied subjects (22). The total number of TMS stimuli applied to each subject varied between 71 and 109 (mean 85) and were well tolerated by both SCI and able-bodied subjects. The motor cortical representation of the abdominal muscles was approximately 2 cm lateral of the mid-line and approximately 1 cm anterior to the auricular plane for all subjects.
Motor-evoked potentials
TMS evoked MEPs in abdominal muscles and the Dia in individuals classified with motor complete SCI (AIS A and B) above T6 level and in matched able-bodied controls. A representative SCI participant (SCI 2) and their matched control (C2) are shown in Fig. 1.
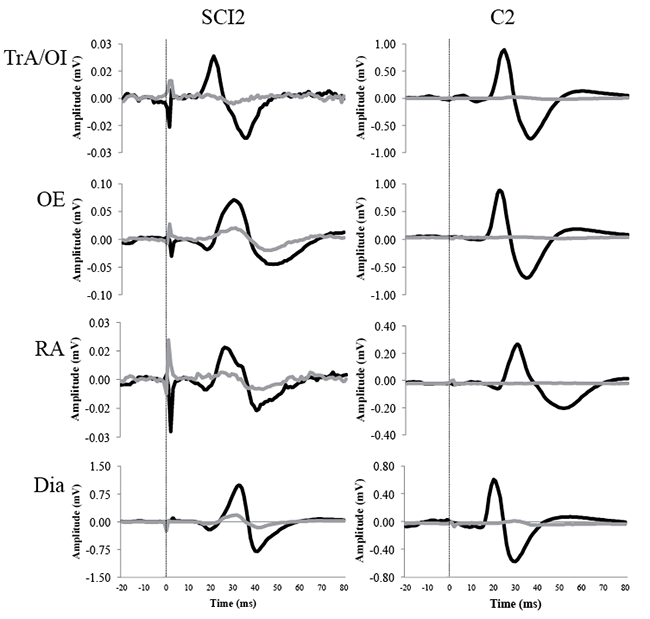
MEPs during sub-maximal contractions. TMS delivered over the right hemisphere elicited contralateral MEPs in all abdominal muscles and the Dia in all subjects (Table II). Stimulation delivered over the left hemisphere evoked MEPs on the contralateral side in at least one abdominal muscle and in the Dia in all SCI subjects, and in 4 able-bodied subjects (Table II).
Stimulation over the right hemisphere evoked MEPs on the ipsilateral side in at least 1 abdominal muscle, in all subjects, and in the Dia in 4 SCI subjects (SCI 1, 2, 3, 5) and 3 able-bodied (C1, 4, 5) subjects (Table II). Stimulation on the left hemisphere evoked MEPs on the ipsilateral side in at least 1 abdominal muscle in all subjects (Table II). In the Dia, MEPs occurred in all subjects with SCI and in 3 able-bodied subjects (C1, 2, 5) in response to left hemisphere stimulation (Table II).
On the contralateral side, mean MEP latencies in the abdominal muscles ranged from 17.2 to 21.9 ms for subjects with SCI, and from 18.0 to 20.8 ms for able-bodied subjects during sub-maximal (or attempted) contractions (Table II). In the Dia, mean onsets for the SCI and the able-bodied group varied between 16.7 and 18.1 ms, and 16.4 and 18.0 ms, respectively (Table II).
On the ipsilateral side, mean MEP latencies in the abdominal muscles ranged from 14.9 to 21.8 ms for the SCI group, and between 19.9 and 23.3 ms for the able-bodied group (Table II). Mean onsets of MEPs in the Dia varied between 17.0 and 20.5 ms for the SCI group, and between 21.9 and 24.1 ms for the able-bodied group (Table II).
MEPs during rest. MEPs were elicited less frequently during rest compared with sub-maximal contractions in individuals with SCI and able-bodied subjects. At rest, contralateral responses to TMS were elicited in at least 1 abdominal muscle on either side in 3 subjects with SCI (SCI 1, 3, 5), and in 3 able-bodied subjects (C2, 3, 5) (Table II). Contralateral Dia responses to TMS delivered over the right and the left hemisphere were observed in 4 subjects with SCI (SCI 1, 2, 3, 5) and in 3 able-bodied subjects (C2, 3, 5) (Table II). Ipsilateral responses in the abdominal muscles to TMS at rest were only observed in 1 subject with SCI (SCI 1) and 1 able-bodied subject (C5) (Table II). In the Dia, MEPs were recorded on the ipsilateral side in 2 subjects with SCI (SCI 1, 3) (Table II).
Mean onsets of contralateral MEPs to TMS at rest in the abdominal muscles ranged from 19.3 to 24.0 ms for the SCI group, and from 20.1 to 22.0 ms for the able-bodied group (Table II). In the Dia, mean onsets varied, for the SCI group between 18.8 and 19.3 ms, and for the able-bodied group between 18.5 and 20.3 ms, respectively (Table II). No group mean onsets of ipsilateral MEPs to TMS were obtained in the abdominal muscles or in the Dia for the SCI group or for the able-bodied group (Table II).
|
Table II. Onset (ms) of contralateral and ipsilateral motor-evoked potential responses (MEPs) following transcranial magnetic stimulation (TMS) delivered over the right and left hemisphere during voluntary (or attempted) sub-maximal trunk muscles tasks (bending and flexion) and in rest in people with spinal cord injury (SCI) (total number of subjects = 5) and in able-bodied people (AB) (n = 5). Responses were recorded for transversus abdominis/obliquus internus (TrA/OI), rectus abdominis (RA), obliquus externus (OE), and diaphragm (Dia). N indicates the number of subjects with MEP responses defined as “present” in at least 5 out of 10 trials. Note: One AB participant did not complete left hemisphere stimulation and therefore the total number of subjects for right-sided responses to contralateral stimulation (A) and left-sided responses to ipsilateral stimulation is n = 4 |
||||||||||
|
Muscle |
Side |
Measure |
Bending |
Flexion |
Resting |
|||||
|
SCI Mean (SD) |
AB Mean (SD) |
SCI Mean (SD) |
AB Mean (SD) |
SCI Mean (SD) |
AB Mean (SD) |
|||||
|
Contralateral responses recorded from the left and right sides |
||||||||||
|
TrA/OI |
Left |
Latency (ms) n (No.) |
19.2 (2.8) 5 (ALL) |
19.2 (2.1) 5 (ALL) |
19.5 (2.7) 5 (ALL) |
18.6 (1.6) 5 (ALL) |
21.9 1 (1) |
21.4 (0.2) 2 (3,5) |
||
|
Right |
Latency (ms) n (No.) |
21.2 (1.5) 3 (1,4,5) |
19.0 (0.7) 4 (1,2,3,5) |
21.5 (3.2) 2 (4,5) |
19.7 (1.4) 4 (1,2,3,5) |
24.0 1 (5) |
22.0 (2.3) 3 (2,3,5) |
|||
|
RA |
Left |
Latency (ms) n (No.) |
19.8 (1.1) 5 (ALL) |
20.3 (1.0) 5 (ALL) |
21.9 (1.8) 5 (ALL) |
20.2 (1.9) 5 (ALL) |
– |
20.6 1 (3) |
||
|
Right |
Latency (ms) n (No.) |
17.2 (0.2) 3 (1,2,4) |
20.8 (2.0) 4 (1,2,3,5) |
20.0 (4.3) 4 (1,2,4,5) |
20.4 (3.0) 4 (1,2,3,5) |
19.3 1 (1) |
21.8 (2.0) 2 (3,5) |
|||
|
OE |
Left |
Latency (ms) n (No.) |
18.6 (2.4) 5 (ALL) |
18.7 (1.7) 5 (ALL) |
19.0 (1.2) 5 (ALL) |
18.2 (1.8) 5 (ALL) |
23.7 (4.3) 2 (1,3) |
20.1 (0.2) 2 (3,5) |
||
|
Right |
Latency (ms) n (No.) |
19.5 (2.0) 5 (ALL) |
18.0 (1.8) 4 (1,2,3,5) |
19.1 (3.1) 5 (ALL) |
18.5 (1.9) 4 (1,2,3,5) |
19.6 (1.0) 2 (1,5) |
20.3 (1.4) 3 (2,3,5) |
|||
|
Dia |
Left |
Latency (ms) n (No.) |
16.7 (1.8) 5 (ALL) |
16.4 (1.2) 5 (ALL) |
18.1 (2.2) 5 (ALL) |
16.7 (1.4) 5 (ALL) |
19.3 (4.8) 4 (1,2,3,5) |
18.5 (0.2) 2 (3,5) |
||
|
Right |
Latency (ms) n (No.) |
17.6 (1.2) 5 (ALL) |
18.0 (1.6) 4 (1,2,3,5) |
17.1 (2.1) 4 (1,2,3,5) |
17.3 (0.7) 4 (1,2,3,5) |
18.8 (2.1) 3 (1,2,5) |
20.3 (3.7) 3 (2,3,5) |
|||
|
Ipsilateral responses recorded from the left and right sides |
||||||||||
|
TrA/OI |
Left |
Latency (ms) n (No.) |
14.9 1 (1) |
22.3 (2.4) 3 (1,2,5) |
19.5 (3.5) 3 (1,4,5) |
21.0 (0.5) 3 (1,2,5) |
14.4 1 (1) |
– |
||
|
Right |
Latency (ms) n (No.) |
20.1 1 (2) |
20.8 (2.5) 2 (3,5) |
21.8 (2.7) 2 (2,5) |
21.8 (1.5) 2 (1,5) |
– |
– |
|||
|
RA |
Left |
Latency (ms) n (No.) |
19.2 (4.4) 3 (1,4,5) |
19.9 (1.9) 4 (1,2,3,5) |
18.4 (3.9) 3 (1,2,5) |
22.5 (0.8) 4 (1,2,3,5) |
– |
20.5 1 (5) |
||
|
Right |
Latency (ms) n (No.) |
20.9 (0.6) 3 (2,4,5) |
22.0 (2.2) 5 (ALL) |
19.2 (1.5) 2 (4,5) |
23.3 (1.4) 4 (1,2,4,5) |
– |
22.8 1 (5) |
|||
|
OE |
Left |
Latency (ms) n (No.) |
17.6 (2.8) 3 (1,3,5) |
21.3 (0.7) 2 (1,5) |
18.9 (2.5) 3 (1,4,5) |
22.1 (0.5) 3 (1,2,5) |
– |
– |
||
|
Right |
Latency (ms) n (No.) |
20.9 (1.7) 5 (ALL) |
20.9 (4.0) 2 (1,5) |
21.2 (0.02) 2 (4,5) |
21.4 (2.0) 2 (1,5) |
– |
– |
|||
|
Dia |
Left |
Latency (ms) n (No.) |
18.8 (2.6) 5 (ALL) |
24.1 (0.1) 2 (1,2) |
17.0 (1.8) 4 (1,2,3,4) |
22.9 1 (5) |
20.6 1 (1) |
– |
||
|
Right |
Latency (ms) n (No.) |
20.5 (0.7) 3 (1,3,5) |
21.9 (1.7) 3 (1,4,5) |
19.2 (1.0) 2 (2,3) |
22.8 (–1.6) 2 (1,5) |
23.6 1 (3) |
– |
|||
|
No: subject number. |
||||||||||
Peak-to-peak amplitude during sub-maximal contractions and at rest. In general, MEPs evoked during rest, had smaller peak-to-peak amplitudes compared with those evoked during sub-maximal contractions (Fig. 2). Relative changes in MEP amplitudes between rest and sub-maximal contractions were smaller in SCI compared with able-bodied subjects (Fig. 2).
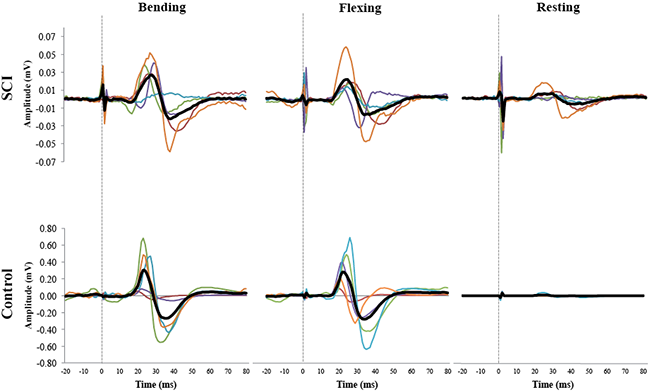
EMG activity during voluntary (or attempted) contractions
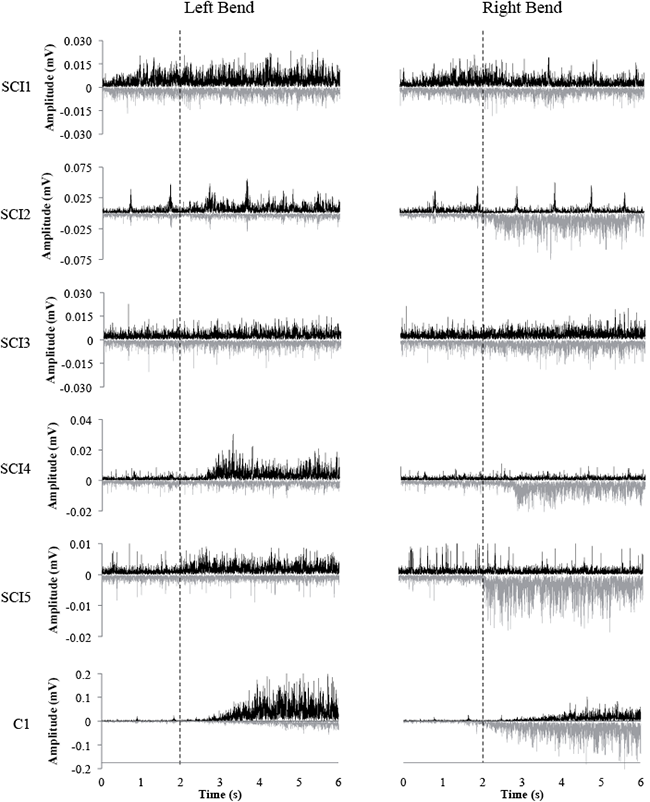
As observed in Fig. 3, individuals with SCI activated OE muscles with the same asymmetrical pattern as able-bodied, depending on the direction of lateral bending. During trunk bending to the left and right, all subjects with SCI were able to produce EMG activity above resting levels (i.e. 2 SD above mean RMS EMG value) in the ipsilateral OE muscle. Increased EMG activity was also observed, albeit less frequently, in the other ipsilateral abdominal muscles during lateral bending to the left (TrA/OI (SCI 2, 4, 5); RA (SCI 2, 4, 5)), and right (TrA/OI (SCI 2, 4); RA (SCI 2, 4, 5)). Increased EMG activity was also observed in some subjects during trunk flexion, in bilateral RA (SCI 2, 4), left TrA/OI and OE (SCI 2, 4, 5) and right TrA/OI and OE (SCI 2, 4). All able-bodied subjects were able to elicit EMG activity above resting levels in all muscles on both sides during all tasks. Furthermore, all SCI subject’s muscles that had a “present” MEP elicited by TMS on the contralateral side showed detectable EMG during voluntary contraction in the same muscle. The same was observed for muscles with “present” MEPs elicited on the ipsilateral side, with the exception of the TrA/OI muscle in 1 SCI subject (SCI 4), and the RA muscle in another SCI subject (SCI 2).
Discussion
Despite being clinically classified with motor-complete SCI above T6, all individuals with SCI were found to have some preserved muscle function in abdominal muscles in response to TMS and voluntary efforts. Activation was found in persons with complete motor and sensory loss (SCI 3, 4, 5), complete motor and incomplete sensory loss (SCI 1), as well as one individual with some partial motor activity in distal segments (SCI 2). What is most striking about these findings is that the abdominal muscles are innervated by spinal cord segments located below the level of injury in these SCI subjects, and based on their ISNCSCI, should have complete loss of motor function of all abdominal muscles. These results highlight the inaccuracy of relying on sensory function to determine motor function in the abdominal muscles of persons with SCI.
Motor-evoked potentials
This is the first study, to our knowledge, to evaluate MEPs to TMS in the abdominal muscles in people with SCI. The MEPs elicited in the abdominal muscles of SCI subjects were similar to those of the able-bodied in terms of latency, pattern and facilitation to voluntary contraction. The onset latencies of MEPs in abdominal muscles of SCI subjects were within the normal ranges observed in able-bodied reported in the current study, and in previous studies involving the TrA (16–22 ms) (13), OI (16–19 ms) (12), RA (16–19 ms) (10), OE (18–22 ms) (24), and the Dia (15–18 ms) (22, 25) muscles.
In both groups, the MEPs followed a rostrocaudal pattern of activation, with earliest onsets recorded from the Dia followed by the abdominal muscles. This rostrocaudal activation pattern has been reported previously in able-bodied (7), and reflects the somatotopic organization of motor neurones within the spinal cord, with the Dia located most rostral (C4), and the abdominal muscles more caudal (T6–T12) (17). Based on the relative differences in MEP latencies between muscles, we would argue that the observed MEP responses in abdominal muscles in both able-bodied and SCI subjects can be attributed to direct activation through descending corticospinal pathways and not via indirect pathways; for example, via stretch reflexes elicited by contraction of other muscles innervated above the lesion, or an increase in intra-abdominal pressure resulting from contraction of the Dia (25, 26). The relative mean difference in onsets observed between Dia and abdominal muscles was 2 ms in both the SCI and the able-bodied group, which is far shorter than the earliest stretch response latencies elicited by direct muscle tap of the abdominal muscles, which ranged from 15 to 22 ms (27).
SCI subjects, like able-bodied, had more frequent MEP responses to stimulation in the right compared with the left hemisphere, and had asymmetrical MEP latencies between bilateral muscles, with earlier onsets in muscles contralateral to the side of stimulation. These observations are consistent with previous reports of descending contralateral and ipsilateral corticospinal projections arising predominantly from 1 hemisphere (12, 13, 28) and earlier responses (2–5 ms) of abdominal muscles contralateral to the side of stimulation in able-bodied subjects (10, 12, 13). The differences in contralateral vs ipsilateral muscles can be attributed to the faster crossed corticospinal neurones, which descend in the lateral cortical spinal tracts (CST) (29), as opposed to slower fibres that run in uncrossed pathways in the anterior CST (30). These asymmetries persist despite the fact that trunk muscles are known to have a greater proportion of uncrossed corticospinal fibres than muscles of the limbs (31).
In the current study, both able-bodied and SCI subjects had MEPs in the abdominal muscles that were elicited predominantly when subjects were asked to produce attempted sub-maximal contractions of the abdominal muscles. In contrast, both groups had less frequent, and smaller amplitude MEPs in resting muscles. These results corroborate previous reports of facilitation of abdominal muscle MEPs with voluntary contractions in able-bodied subjects (10, 13, 32). This facilitation has been attributed to the increase in excitability of the corticospinal system with increases in the level of voluntary activity (33, 34).
Voluntary activation
All individuals with SCI were also able to voluntarily activate their abdominal muscles above resting EMG threshold during maximal trunk muscle tasks. These observations are consistent with a previous observation of voluntary activation of abdominal muscles in an individual with a motor-complete SCI (T3), recorded with indwelling electrodes (4). While previous observations were limited to abdominal muscle activity during a general Valsalva manoeuvre, the current study was able to demonstrate that the muscle activation pattern was task specific in the majority of the SCI subjects (Fig. 3). The EMG traces from the SCI group and the able-bodied subject appeared similar in activation pattern, but with lower amplitudes in the SCI group, which is most likely influenced by a lower level of activation in the descending pathways to the muscles.
Implication of classification, rehabilitation and exercising
The present findings of preserved function in motor pathways to the abdominal muscles, and the ability to activate them in individuals with SCI, clinically classified with motor-complete lesion above T6, highlights a need to revise the current ISNCSCI to include motor tests also for muscles of the trunk. While TMS has proven to be an effective means of providing more sensitive measures of motor preservation in these muscles, it is a time-consuming procedure, and may be prohibitive due to the large number of exclusion criteria, some of which are commonly associated with SCI (i.e. concussions). Therefore, other alternative methods for providing a sensitive measure of preserved function are required. Surface EMG provides one alternative, especially when considering that EMG during voluntary activation was observed in all muscles in which MEPs were elicited by contralateral TMS in the current study. While surface EMG is well suited for recording superficial muscles, the activity in deeper abdominal muscles is less accessible. Indwelling EMG provides accurate measures for deep muscles (4); however, this is also an invasive, time-consuming procedure and provides only local measures of muscle activity. A potential alternative is ultrasound measures of muscle thickness as an indirect measure of muscle activity (e.g. 35–37), although its sensitivity to very subtle levels of contraction preserved following SCI remains uncertain.
The ability to clinically identify the extent to which corticospinal inputs to abdominal muscles may be preserved is important as it may offer a potential target for rehabilitation training and exercise prescription. For example, exercise with a special kayak ergometer, which emphasizes upper-body activity and trunk postural control through complex 3D paddling movements, has been shown to have positive effects on trunk stability in sitting, upper body coordination and functional performance in people with high thoracic SCI (2, 3). It is difficult to speculate about putative neural mechanisms due to the intervention, but a possible reason for the increased postural stability and coordination could be that the training provokes an increase in neural drive in the descending corticospinal pathways to postural trunk muscles. This increased drive might, in turn, induce activation of denervated and/or atrophied musculature. Such an improvement in neural communication between the brain and muscles has been reported previously after other types of intense training periods, such as a gait training regime in people with incomplete SCI (38). When prescribing optimal exercises, the type of training task will be of importance for producing the best descending activation in available corticospinal tracts to the target muscle/muscles, for functional improvements.
Conclusion
MEP responses to TMS were elicited in the abdominal muscles in individuals with motor-complete SCI above T6, with similar latencies and patterns as in able-bodied. All subjects with SCI were also able to voluntary activate their abdominal muscles above resting EMG values during trunk muscle tasks. These findings suggest that TMS and EMG can be used to identify preserved function of corticospinal pathways to the abdominal muscles in people with SCI, and that the ISNCSCI (1, 19) at thoracic segmental level cannot, by itself, describe the condition of spinal cord tract function to the abdominal muscles.
Acknowledgements
The authors would like to thank the Swedish Centre for Sports Research (CIF), The Swedish Association for Survivors of Accident and Injury (RTP), and the Canadian Institute for Health Research (CIHR) for financial support. Special thanks are due to the participants.
References
