Johan Gäverth, PT, PhD1,2, Ann-Christin Eliasson, OT, PhD1, Kjell Kullander, MD3, Jörgen Borg, MD, PhD3, Påvel G. Lindberg, PT, PhD1,3,4 and Hans Forssberg, MD, PhD1
From the 1Department of Women’s and Children’s Health, Karolinska Institutet, 2Department of Physiotherapy, Karolinska University Hospital, 3Department of Clinical Sciences, Rehabilitation Medicine, Karolinska Institutet, Danderyd Hospital, Stockholm, Sweden and 4FR3636 Neurosciences, CNRS, Université Paris Descartes, Paris, France
OBJECTIVE: The NeuroFlexor objectively quantifies the neural, elastic and viscous components of passive movement resistance in wrist and finger flexor muscles. In this study we investigated the sensitivity of the NeuroFlexor to changes in spasticity induced by treatment with botulinum toxin type A (BoNT-A).
DESIGN: Prospective observational design.
SUBJECTS: A convenience sample of 22 adults with post-stroke upper limb spasticity scheduled for botulinum toxin treatment.
METHODS: BoNT-A was given according to individual treatment plans. NeuroFlexor assessments were made before treatment and 4 and 12 weeks after.
RESULTS: At group level, spasticity decreased significantly at 4 weeks (expected time of maximum effect) (p = 0.04). At 12 weeks, spasticity had rebounded and no longer differed significantly from baseline (p = 0.64), i.e. in line with the pharmacodynamics of BoNT-A. At the individual level, 7 participants showed a reduction in spasticity greater than the measurement error. The reduction was dose-dependent (r(20) = 0.66, p < 0.001), and largest in participants with the highest dose.
CONCLUSION: At the group level, the sensitivity of NeuroFlexor is good enough to detect reduction in spasticity after treatment with BoNT-A. Further work is needed to establish the sensitivity of NeuroFlexor on an individual level.
Key words: spasticity; sensitivity; outcome measure; upper extremity; botulinum toxin type A.
J Rehabil Med 2014; 46: 00–00
Correspondence address: Johan Gäverth, Karolinska Institutet, Department of Women’s and Children’s Health, Neuropediatric Research Unit, Stockholm Brain Institute and the Health Care Sciences Postgraduate School, Astrid Lindgrens Barnsjukhus Q2:07, SE-171 76 Stockholm, Sweden. E-mail: johan.gaverth@ki.se
Accepted Feb 25, 2014; Epub ahead of print May 21, 2014
Introduction
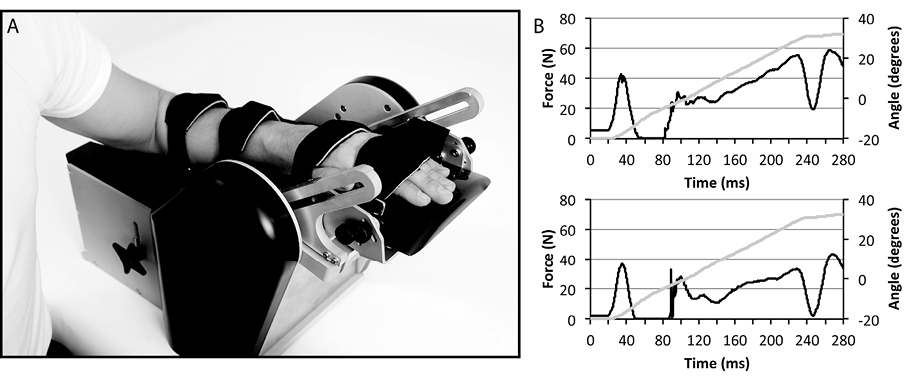
There is a lack of easy-to-use, valid, reliable and objective clinical methods to diagnose and quantify spasticity and thus to guide treatment interventions and inform the clinician about change over time (1). We therefore recently described a new method, the NeuroFlexor, to measure spasticity, intended for clinical use (2). The method comprises a mechanical device that produces a controlled passive isokinetic wrist extension, thereby stretching the wrist and finger flexor muscles (Aggero MedTech AB, Solna, Sweden) (Fig. 1). A force transducer measures the resistance during the movement, and the force is analysed using a computerized biomechanical model of the hand (2). By use of data collected at different velocities, the model allows calculation of 3 separate components that contribute to total passive muscle resistance: the neural (NC), elastic (EC), and viscous (VC) components. The NC represents spasticity according to the definition by Lance (3) “Spasticity is a motor disorder characterized by a velocity-dependent increase in tonic stretch reflexes (‘muscle tone’) with exaggerated tendon jerks, resulting from hyperexcitability of the stretch reflex” and we define the NC as the spasticity component. Evidence of the validity of the method has been presented in a previous paper (2), and in a subsequent study we have shown that the NeuroFlexor measurements have high intra-rater (test-retest) and inter-rater reliability for NC data and fair to high reliability for EC and VC data (4). However, in order for the instrument to be used to evaluate the effect of anti-spasticity interventions, knowledge of an additional psychometric property, i.e. “sensitivity to change” is required. Sensitivity to change is defined as “the ability of an instrument to measure change in a state” (5). Knowledge about sensitivity to change is a step towards establishing the responsiveness of a measure, which is similar but separate from sensitivity to change, since responsiveness takes into account the aspect of meaningfulness or clinical relevance (5). Determining what is a meaningful or clinically relevant change is, however, quite difficult in the field of spasticity management, since it is often a matter of subjective opinion or ratings of the patient, carers and healthcare professionals. It remains an elusive problem, since there are no strong correlations between reduction in spasticity and objective measures of impairments or activity limitations (6). Thus, in this study we have chosen to focus on sensitivity to change. In order to do so, we used a common clinical anti-spasticity treatment, intramuscular injections of botulinum toxin type A (BoNT-A). This is a standard treatment, well known to reduce focal spasticity (7–11). The toxin is injected into the spastic muscle, and acts by selectively and reversibly blocking the release of acetylcholine at the neuromuscular junction (12). The BoNT-A is therefore only expected to reduce the NC of the resistance to passive stretch, while not affecting the EC and VC. This treatment is therefore well suited for the aim of this study, which is to explore the sensitivity to change of the NC of the NeuroFlexor method.
Methods
Study design
The participants were examined before and after intramuscular injections of BoNT-A in the spastic wrist and finger flexor muscles. The examinations were performed on 3 occasions: at baseline (maximum 2 weeks before treatment), 4 weeks after treatment (expected time of peak effect) (13) and 12 weeks after treatment (when the effect of the treatment was assumed to have subsided significantly) (14).
Participants
A total of 22 patients scheduled for treatment with BoNT-A were recruited from an outpatient spasticity clinic at the Department of Rehabilitation Medicine, Danderyd University Hospital, Stockholm, Sweden. Inclusion criteria were: (i) stroke or traumatic brain injury > 6 months prior to inclusion, (ii) spasticity in wrist and finger flexors, with a modified Ashworth score (MAS) ≥ 1, (iii) considered suitable for treatment with BoNT-A, (iv) no fixed contractures of the wrist, and (v) ability to understand and comply with instructions. The participants’ demographic characteristics are shown in Table I. Four of the 22 participants did not participate in the assessment at 12 weeks, due to illness, problems with transportation, or finding that participation in the study was too time-consuming.
Ethical approval was obtained from the Regional Ethical Review Board in Stockholm, and informed consent was obtained from all participants according to the Declaration of Helsinki.
|
Table I. Demographic characteristics of the participants |
|
|
Variables |
Distributions n = 22 |
|
Age, years |
|
|
Mean (SD) |
50.4 (11.5) |
|
Min–max |
22–67 |
|
Gender, n (%) |
|
|
Male |
18 (82) |
|
Female |
4 (18) |
|
Paretic side, n (%) |
|
|
Right |
11 (45.5) |
|
Left |
10 (50) |
|
Bilateral |
1 (4.5) |
|
Maximal grip strength in relation to unimpaired side, %a |
|
|
Mean (SD) |
23 (18) |
|
Min–max |
0–79 |
|
Dominant side, n (%) |
|
|
Right |
19 (86) |
|
Left |
3 (14) |
|
Stroke type, n (%) |
|
|
Haemorrhagic |
12 (54.5) |
|
Ischaemic |
9 (41) |
|
Trauma |
1 (4.5) |
|
Time since stroke/trauma, months |
|
|
Mean (SD) |
50.1 (39.7) |
|
Min–max |
7–123 |
|
aOne participant excluded due to bilateral involvement (trauma). SD: standard deviation. |
|
Botulinum toxin treatment
All participants received treatment with BoNT-A in one or more of their wrist and/or finger flexors (flexor carpi radialis and ulnaris, flexor digitorum superficialis and profundus) according to their individually tailored clinical treatment plan. The physician selected the muscle(s) and the dose based on clinical experience and goal-setting. The mean BoNT-A dose (Botox®, Allergan, Irvine, CA, USA) was 111 units (95% confidence interval (CI): 87–135). The individual doses are shown in Table II. In addition, a majority of the participants received injections in other muscles in the upper extremity; the total mean dose, including units injected in wrist and finger flexor muscles, was 198 units (95% CI 155–240). No additional treatment or hand training was prescribed. No serious adverse events related to the administration of BoNT-A or measurements with the NeuroFlexor were reported during the study.
Procedure
The NeuroFlexor instrument was used to estimate the NC, EC and VC of the passive movement resistance of the wrist and finger flexor muscles (2). We followed the procedure described in our previous study (4). In brief, the participant was seated comfortably with the elbow in 90° of flexion and the hand securely fastened into the device with the wrist axis of rotation aligned with the device. The instrument performed slow and fast movements (5 and 236°/s, respectively) in a 50° range of movement with a starting angle of 20° of palmar flexion. For each participant and test occasion, one value for each of NC, EC and VC was calculated in dedicated software using averaged NeuroFlexor recordings from 9 fast and 4 slow passive movements (NeuroFlexor Scientific, Release 0.0.6, Aggero MedTech AB, Solna, Sweden).
Clinical tests
A physiotherapist (JG) performed the clinical tests according to standard procedures for each test. Wrist MAS (0–4) (15) was obtained with the participant seated with the elbow in 90° of flexion and the forearm pronated. Finger MAS was obtained with the elbow in 90° of flexion, the wrist in neutral position and the proximal and distal interphalangeal joints of fingers 2 to 5, thus allowing isolated movement in the metacarpophalangeal joint. Passive range of movement of the wrist was measured using a goniometer with the participants elbow in 90° of flexion and fingers extended (16). Maximal grip strength was measured in both hands using a digital handheld dynamometer (Baseline, Fabrication Enterprises Inc., White Planes, New York, NY, USA). Verbal encouragement was given, and the best of 3 attempts was used in the analysis. The measurement of the unimpaired hand was used to describe the relative level of impairment in the affected hand expressed as a ratio (impaired/unimpaired).
Statistical analysis
Descriptive statistics were calculated using mean and 95% CI or standard deviations (SD) for continuous data. Difference between occasions was studied using a repeated measures analysis of variance (ANOVA) for continuous data (NC, EC, VC) and a Friedman’s ANOVA for ordinal data (MAS). Post-hoc analyses were performed using the paired t-test or the Wilcoxon matched-pairs test.
The repeatability coefficient (r) from our previous reliability study (4) was used to calculate a 95% CI around the baseline value. A change after treatment resulting in measurement values outside the CI is considered a change greater than the measurement error. The 95% CI was calculated using the following formulae: lower limit = (X0 + k/ r) − k and upper limit = r(X0 + k) − k, where X0 is the observed value before treatment, r is the repeatability coefficient, and k is a constant required to correctly calculate the interval (kNC = 3, kEC = 1, kVC = 1). The repeatability coefficients for the NC, EC, and VC were 1.798, 1.897 and 1.404, respectively (4).
Associations between continuous variables were explored using Pearson’s correlation (r) and those between ordinal variables using Spearman’s rank order correlation (rs). The significance level was set at p < 0.05.
Results
Treatment-associated changes in NeuroFlexor – neural component
Four weeks after treatment there was a significant reduction in NC on a group level (t(21) = 3.199, p = 0.04). Correlation analysis showed that this reduction was dose-dependent (r(20) = 0.67, p < 0.001).
A repeated-measures ANOVA including all 3 test occasions (n = 18) showed a significant difference between the occasions (F(2, 34) = 5.078, p = 0.01 (Table III). Post-hoc analysis showed that, as for the whole group, there was a reduction between baseline and 4 weeks (t(17) = 2.402, p = 0.03) and a significant increase between 4 and 12 weeks (t(17) = −3.328, p = 0.004). There was no significant difference between baseline and 12 weeks (t(17) = 0.477, p = 0.64). When BoNT-A dose was included as a covariate in the ANOVA, the difference between occasions was no longer present (F(2, 32) = 1.395, p = 0.26), suggesting that the BoNT-A was responsible for the majority of the change in NC over time.
Individual changes in NC from baseline are shown in Table II. At the individual level, 7 of 22 participants had a reduction in NC that was greater than the expected variability; that is, greater than the measurement error (4). These individuals are shaded grey in Table II, column “diff NC at 4 weeks”. The mean dose of BoNT-A for these participants was 145 units (95% CI 107–183). For the participants with no significant change (unmarked in Table II, column “diff NC at 4 weeks”), the mean dose was 104 units (95% CI 74–135). Five participants had an increase in NC at 4 weeks; in 2 of them the increase was greater than the measurement error (#1 and #3, Table II). The BoNT-A doses in these cases were 20 and 50 units, respectively.
Treatment-associated changes in NeuroFlexor – elastic component and viscous component
There were no significant changes in EC and VC at 4 weeks (p > 0.05, Table III). The baseline levels of EC and VC for all 22 subjects are shown in Table II.
|
Table II. Individual baseline NeuroFlexor components and modified Ashworth score (MAS) and the change at 4 and 12 weeks after treatment |
||||||||||
|
Subject |
BoNT-A units |
Injected muscles (wrist and finger flexors) |
Baseline EC (N) |
Baseline VC (N) |
Baseline NC (N) |
Diff NC at 4 weeks (N) |
Baseline MAS fingers |
Diff MAS fingers at 4 weeks |
Baseline MAS wrist |
Diff MAS wrist at 4 weeks |
|
1 |
20 |
FDS = 20 |
7.1 |
0.7 |
2.8 |
7.4 |
1+ |
–1 |
1 |
–1 |
|
2 |
50 + |
FCR = 25, FCU = 25 |
5.8 |
1.7 |
18 |
–0.8 |
2 |
–2 |
2 |
–2 |
|
3 |
50 + |
FDP = 25, FDS = 25 |
–1 |
1.2 |
3.6 |
7.9 |
2 |
–2 |
2 |
–3 |
|
4 |
60 + |
FDP = 30, FDS = 30 |
12.1 |
0.7 |
10.5 |
–3.2 |
3 |
–1 |
3 |
–1 |
|
5 |
60 + |
FDP = 20, FDS = 40 |
10.9 |
–0.6 |
50 |
–10.8 |
3 |
–1 |
3 |
–1 |
|
6 |
70 |
FDP = 35, FDS = 35 |
11.5 |
0.4 |
39.4 |
–3.4 |
3 |
–1 |
3 |
–1 |
|
7 |
85 |
FDP = 35, FDS = 50 |
4.1 |
0.6 |
4.2 |
–6.7 |
1+ |
–2 |
1 |
–1 |
|
8 |
85 + |
FDP = 40, FDS = 45 |
0.8 |
0.3 |
14.1 |
4.7 |
3 |
–3 |
2 |
–2 |
|
9 |
90 |
FDS = 40, FCR = 50 |
6.4 |
0.9 |
29.5 |
–5.0 |
2 |
0 |
2 |
0 |
|
10 |
90 + |
FDP = 40, FDS = 50 |
8.5 |
–0.4 |
18.2 |
6.3 |
3 |
–3 |
3 |
–2 |
|
11 |
93 |
FDP = 25, FDS = 23, FCR = 20, FCU = 25 |
3.4 |
0 |
4.2 |
1.2 |
1 |
–1 |
0 |
0 |
|
12 |
100 + |
FCR = 50, FCU = 50 |
6.1 |
1.3 |
43.6 |
–16.8 |
3 |
–1 |
2 |
1 |
|
13 |
100 + |
FDP = 50, FDS = 50 |
14.6 |
0.6 |
32.8 |
–0.5 |
3 |
–1 |
2 |
0 |
|
14 |
100 + |
FDP = 50, FDS = 50 |
2.9 |
2.2 |
44.4 |
–32.7 |
1 |
2 |
1+ |
–1 |
|
15 |
140 + |
FDP = 30, FDS = 30, FCR = 40, FCU = 40 |
4.5 |
1 |
13.9 |
–9.8 |
2 |
–3 |
1+ |
–1 |
|
16 |
155 + |
FDP = 35, FDS = 40, FCR = 40, FCU = 40 |
6 |
1 |
21.3 |
–17.6 |
2 |
–2 |
2 |
–2 |
|
17 |
160 + |
FDP = 40, FDS = 40, FCR = 40, FCU = 40 |
5.5 |
–0.1 |
22.1 |
–2.7 |
3 |
–1 |
3 |
–1 |
|
18 |
160 + |
FDP = 40, FDS = 40, FCR = 40, FCU = 40 |
5.9 |
0.7 |
27.7 |
–19.9 |
3 |
–3 |
1 |
–1 |
|
19 |
190 |
FDP = 40, FDS = 50, FCR = 50, FCU = 50 |
7.6 |
0.5 |
68.9 |
–40.2 |
3 |
0 |
3 |
0 |
|
20 |
190 + |
FDP = 50, FDS = 50, FCR = 40, FCU = 50 |
5 |
1.6 |
30.1 |
–24.1 |
2 |
–1 |
2 |
–2 |
|
21 |
200 + |
FDP = 50, FDS = 50, FCR = 50, FCU = 50 |
4 |
1 |
45.1 |
–19.5 |
2 |
–2 |
2 |
–3 |
|
22 |
200 + |
FDP = 50, FDS = 50, FCR = 50, FCU = 50 |
9 |
0.9 |
23.3 |
–7.2 |
3 |
–1 |
3 |
–1 |
|
Participants ordered according to the number of units of botulinum toxin administered in wrist and finger flexor muscles, i.e. FCR, FCU, FDP and FDS. Grey aresa highlight individuals with a reduction in NC and wrist MAS larger than the measurement error. For NC this is based on the repeatability coefficient r = 1.798 and for MAS (0–4) it is based on the change being larger than –1 step on the ordinal scale. The sign + in column BoNT-A Units denotes participants that received injections also in muscles other than wrist and finger flexors in the upper extremity. Note that there are a few small negative baseline values in NC, EC and VC. This is related to the measurement error of the biomechanical model and explained in a previous paper (2). BoNT-A: botulinum toxin type A; FCR: flexor carpi radialis; FCU: flexor carpi ulnaris; FDP: flexor digitorum profundus; FDS: flexor digitorum superficialis; MAS: modified Ashworth score; NC: neural component; EC: elastic component; VC: viscous component; Diff: difference. |
||||||||||
|
Table III. Baseline values and the change of NeuroFlexor components 4 and 12 weeks after treatment (n = 18) |
|||||||
|
Baseline Mean (95% CI) |
Difference Baseline – 4 weeks Mean (95% CI) |
p-value |
Difference 4 weeks – 12 weeks Mean (95% CI) |
p-value |
Difference Baseline – 12 weeks Mean (95% CI) |
p-value |
|
|
NC |
25.7 (16.7 to 34.7) |
7.4 (0.9 to 14.0) |
0.028* |
–6.3 (–10.2 to –2.3) |
0.004** |
1.1 (–3.9 to 6.2) |
0.639 |
|
EC |
6.7 (4.7 to 8.7) |
0.6 (–0.9 to 2.1) |
0.408 |
–0.5 (–2.0 to 1.1) |
0.553 |
0.2 (–1.4 to 1.7) |
0.843 |
|
VC |
0.7 (0.4 to 1.0) |
0.3 (–0.0 to 0.6) |
0.051 |
0.1 (–0.2 to 0.5) |
0.372 |
0.5 (0.0 to 0.9) |
0.046* |
|
Paired t-test: *p < 0.05, **p < 0.01. NC: neural component (N); EC: elastic component (N); VC: viscous component (N); CI: confidence interval. |
|||||||
Treatment-associated changes in wrist and finger modified Ashworth score score
At the group level, there was a significant difference in MAS for the wrist between the test occasions (χ2(2, n = 18) = 18.655, p < 0.000). Post-hoc analysis showed a significant reduction between baseline and 4 weeks (Z=−3.502, p < 0.000). This reduction was still present at 12 weeks (Z = −2.581, p = 0.01), and there was no significant difference between 4 and 12 weeks (Z = −1.515, p = 0.13).
Individual MAS for the wrist and the change at 4 weeks are shown in Table II. Of the 22 participants, 17 had a change in wrist MAS of at least −1. For finger MAS this number was 19. These individuals are shaded grey in Table II. The change in wrist MAS at 4 weeks was not correlated to the dose of botulinum toxin (rs(20) = 0.09, p = 0.71), neither was the finger MAS (rs(20) = 0.07 p = 0.74.)
Discussion
The main purpose of the present study was to explore whether the NeuroFlexor is sensitive enough to monitor changes in spasticity caused by intramuscular injections of BoNT-A in wrist and finger muscles. In line with the action of botulinum toxin, the NC was reduced 4 weeks after the injections when the effect was expected to be at a maximum, and increased towards baseline level after 12 weeks, when the effect was expected to diminish (17). The passive muscle and tissue components (EC and VC) did not change over time. This was as expected, since BoNT-A prevents muscle contractions by blocking the release of acetylcholine at the neuromuscular junction and is unlikely to influence the mechanical properties of the muscle (i.e. elasticity and viscosity) (18). Hence, the method seems to be sensitive enough to monitor the treatment effect at a group level. However, when studying the outcome of the treatment at an individual level, the results were less clear-cut.
The BoNT-A injections reduced NC after 4 weeks in 17 of the 22 subjects, while an increase of NC was seen in 5 subjects. By applying the repeatability coefficient calculated in a previous study of the NeuroFlexor measurement error (4), we found that only 7 of the 17 subjects with reduced NC passed the significance level; that is, the NeuroFlexor could only identify a positive treatment response in 7 of the 22 subjects. At first glance this seems to be a poor outcome that could dismiss the method as not being sensitive enough. It should, however, be noted that 11 of the 22 had a BoNT-A dose less than 100 units (see Table II) and it is indeed questionable whether injections in the small dose range will cause a significant reduction of spasticity in the wrist flexor muscles (19). Furthermore, there is no gold standard to measure spasticity in the clinic, and the measured poor outcome may reflect the actual condition. One finding supporting this assumption is the positive correlation between the dose of BoNT-A and the reduction in NC. Table II illustrates this dose-response relationship with grey shading of the patients who had a reduction exceeding the NeuroFlexor measurement error. Six of the 7 patients with a significant reduction were among those with ≥ 100 units. This dose-effect relationship has been demonstrated previously by others using electromyography in mice (20) and in humans with cervical dystonia (21). This might imply that the NeuroFlexor is actually sensitive to the real changes induced by large doses of BoNT-A. One potential problem with this interpretation is that 3 participants were given high doses (≥ 160 units) but did not have a significant reduction in NC. However, this might have been due to the choice of muscles that were injected, or to the presence of neutralizing antibodies towards BoNT-A (although this is very rare, occurring in < 1% of cases (22)). Another problem is the increase in NC at 4 weeks in 5 of the 22 participants. The changes may reflect an absent or poor outcome of the BoNT-A injection in a similar way to those with small negative changes in NC.
Comparison between neural component and the modified Ashworth score
The results from MAS investigation showed a completely different pattern. Seventeen of the 22 participants showed a reduction in wrist MAS of at least 1 step on the scale. This might suggest that MAS would be a more sensitive measure to use when rating spasticity in the clinic. However, the validity of the MAS measurements must be questioned. There was no correlation between dose of BoNT-A and changes in MAS in this study, although such a relationship has been shown in other studies (19, 23). Large changes were seen both in participants receiving large doses and in those receiving small doses (Table II). In addition, a MAS change of only 1 step may be within the measurement error, since this is an ordinal scale with only a few steps. Indeed, the dimension of smallest real difference has not been established other than empirically (7, 24). This study indicates that a change of 1 MAS unit should not be considered significant. The reliability of MAS is only poor to moderate, and being a score that is set subjectively by the examiner it is likely to be influenced by the placebo effect. This was supported in a randomized controlled trial of BoNT-A (25) which showed that 69% (22 of 32) of the participants in the placebo group had at least 1 unit reduction in the MAS at follow-up. We therefore believe that the repeatability coefficient for the NeuroFlexor measurements is a more accurate way to determine a real change after treatment, compared with the MAS.
Study limitations
The most serious limitation of this study is that we had no gold standard spasticity measure to compare with the NeuroFlexor measurements. Such methods would have required electromyographic recordings and a more sophisticated laboratory-based set-up. In this study, we wanted to see if it was feasible to use the NeuroFlexor in a clinical setting, and it was not possible to combine this with the use of advanced equipment. Obviously MAS has serious limitations, and the lack of correlation between changes in the NC and MAS does not really tell us which one is correct. In the future, a similar study should be performed in which NeuroFlexor is compared with electrophysiological methods.
Another limitation is the small and selected sample of study patients. For safety reasons, the sample did not include the most severely affected stroke patients with obvious contractures or cognitive impairments. Before generalizing the findings of this study to the whole stroke population, other categories of patients must be tested. The drop-out at 12 weeks (n = 4) is unfortunate, but it did not severely limit the power of the statistical analysis. Finally, this was not a blinded study. Although the examiner was unaware of the BoNT-A doses until after the last examination, we cannot rule out bias in the clinical test results.
Conclusion
This study has shown that the NeuroFlexor is feasible to use in a clinical setting, and sufficiently sensitive to monitor reduced spasticity after BoNT-A at the group level. The question of whether NeuroFlexor is a valid measure to monitor spasticity in individual patients needs further study with recordings from more advanced laboratory measurements.
Acknowledgements
This work was supported by the Swedish Research Council, the Swedish Foundation for Strategic Research, the Berzelii Centre Stockholm Brain Institute, the Strategic Neuroscience Program at Karolinska Institutet, the Health Care Sciences Postgraduate School, Stiftelsen Promobilia, and the Swedish Association of Registered Physiotherapists.
We thank the staff of the Department of Rehabilitation Medicine at Danderyd University Hospital for their assistance during data collection.
Conflict of interest
The NeuroFlexor method described in this paper is patented (WO/ 2008/121067). Two of the authors (JG and PGL) are shareholders in the company Aggero MedTech AB, which manufactures the NeuroFlexor instrument.
References
