Ross Van Der Werff, PT1, Shaun O’Leary, PhD, PT1,2, Gwendolen Jull, PhD, PT1, Michael Peolsson, PhD, MSc CE, RN3, Johan Trygg, PhD, MSc CE3 and Anneli Peolsson, PhD, PT1,4
From the 1NHMRC CCRE (Spinal Pain, Injury and Health), The University of Queensland, Brisbane, 2Physiotherapy Department, Royal Brisbane and Women’s Hospital, Queensland Health, Queensland, Australia, 3Computational Life Science Cluster and Department of Chemistry, Umeå University, Umeå and 4Department of Medical and Health Sciences, Division of Physiotherapy, Faculty of Health Sciences, Linköping University, Linköping, Sweden
OBJECTIVE: To evaluate the capacity of the ultrasound-based method of speckle tracking analysis to detect changes in multilayered dorsal neck muscle activity induced by performing a lifting task.
SUBJECTS: Twenty-one healthy individuals.
DESIGN: Participants performed a loaded lifting task in 3 different postural orientations of the neck (neutral, flexed and forward head posture). Ultrasound images were recorded and speckle tracking analysis was used to quantify muscle deformation and deformation rate over 3 equal time-periods during the lifting sequence (rest, mid-lift and end-lift).
RESULTS: Significant main effects of postural orientation for the deformation measure (p < 0.05) and time for the deformation rate measure (p < 0.05) were observed in all dorsal muscles examined. Significant time by postural interactions for the deformation measure were observed in the trapezius, semispinalis cervicis and multifidus (p < 0.05) and in the semispinalis cervicis (p < 0.05) for the deformation rate measure.
CONCLUSION: Speckle tracking analysis ultrasound measurements can detect differences in multilayered muscle activity of the dorsal neck induced by postural variations during a lifting task. Findings for the deformation and the deformation rate measures suggest that they quantify a different, albeit related, mechanical event during muscle contraction in a functional task such as lifting.
Key words: ultrasound; muscle deformation; cervical muscles; speckle tracking; neck posture.
J Rehabil Med 2014; 46: 662–667
Correspondence address: Anneli Peolsson, NHMRC CCRE (Spinal Pain, Injury and Health), The University of Queensland, St Lucia, QLD 4072, Brisbane, Australia. E-mail: Anneli.Peolsson@liu.se
Accepted Feb 18, 2014; Epub ahead of print Jun 4, 2014
INTRODUCTION
Quantification of the function of complex muscle groups, such as the dorsal neck muscles, is challenging due to their multilayered structure. Traditionally, investigation of multilayered muscle groups has depended on methods such as intramuscular electromyography (EMG), but this has disadvantages due to its invasive nature. The disadvantages of intramuscular EMG may be further amplified when evaluating muscle groups, such as the dorsal neck muscles, that require a minimum of 5 fine-wire insertions for evaluation of all muscle layers. Alternative non-invasive methods, such as muscle functional magnetic resonance imaging (mfMRI), have been utilized to evaluate the function of multi-layered groups, such as the dorsal neck muscles (1–3); however, mfMRI has limitations, in that it only quantifies post-exercise metabolic events (change in water content) within the muscle (4). Thus, there is a need to develop alternative methods of non-invasively measuring the function of multilayered muscle groups.
One method of quantifying muscle function is an ultrasound (US)-based measurement called speckle tracking analysis (STA) (5). STA permits the measurement of mechanical muscle events (muscle deformation and deformation rate) that can be utilized as surrogate measures of muscle activity (6). STA has been utilized for assessment of cardiac function (7–9). It has been validated against other methods (10, 11) with good results, suggesting STA to be an accurate and valuable method to determine cardiac muscle dysfunction (11). STA has also been validated for assessment of skeleton muscle tendon (12, 13) and skeleton muscles (14) with promising results.
The aim of this study was to evaluate the capacity of STA measurements to detect differences in the behaviour of multiple dorsal neck muscle layers induced by a lifting task performed in 3 different neck postures. A previous study using mfMRI (1) has shown that altering neck posture results in changes in the differential activity of the dorsal neck muscle layers. We hypothesize that the STA measures of deformation and deformation rate will detect differences in dorsal neck muscle activity when lifting is performed in the different neck postures.
METHODS
Participants
A convenience sample of 21 right-handed healthy individuals (14 females, 7 males) (Table I) from a university population volunteered to participate in the study. Participants were recruited through advertisements at a university that included staff and students from different disciplines without professional pre-understanding of neck muscle function who expressed interest in the study. Inclusion criteria were: no incidence of neck or shoulder pain in the preceding month; no prior history of neck or shoulder disorders that required medical intervention; no signs of physical dysfunction during clinical examination of the neck. The clinical examination included relevant questions regarding their neck disorder and medical history, and a physical examination of the neck that included active range of motion, and manual examination of the cervical spine (segmental motion and pain provocation tests).
Ethical approval for the study was granted by the Institutional Medical Research Ethics Committee and was conducted in accordance with the Declaration of Helsinki. All participants received verbal and written information about the study and signed an informed consent form.
|
Table I. Characteristics of participants |
||
|
Mean (SD) |
Range |
|
|
Age, years |
25.7 (6.5) |
19–44 |
|
Height, cm |
168 (8.3) |
152–181 |
|
Weight, kg |
65 (10.9) |
50–87 |
|
Physical activitya |
3.5 (0.68) |
2–4 |
|
SD: standard deviation. aMeasured by 2 questions, on “everyday physical activity” and “exercise/sport/open-air activity” and combined into a 4-point score (where 1 = inactivity and 4 = high activity). |
||
Measurements
Ultrasound imaging and analysis. US images of the dorsal neck muscles were recorded with a 12.0 MHz linear transducer (38-mm footprint) and US Vividi (GE Healthcare, Horten, Norway) by a single sonographer who was an experienced musculoskeletal physiotherapist with over 3 years of experience in US imaging that includes in excess of 1,000 US registrations of the neck muscles. A 2-dimensional (2D) US image system was used with a high frame-rate of 50 frames/s, operating in B-mode to take recordings of the upper trapezius, splenius, semispinalis capitis, semispinalis cervicis and multifidus muscles (Fig. 1) at the level of the C4 spinous process. A standardized placement of the US transducer was ensured between participants by first positioning it in a transverse orientation at the marked C4 level on the right side to consistently identify the underlying dorsal neck muscle layers and bony landmarks. The transducer was then rotated 90° to the final longitudinal position to the right of the neck midline, as this has been found to be the optimal position to record US videos of dorsal neck muscle contractions (6).
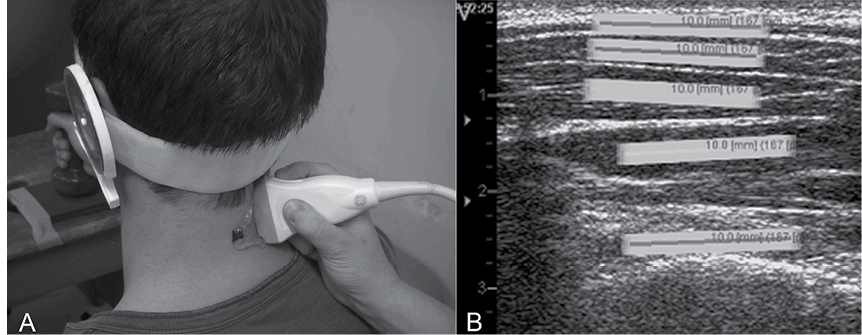
Measurement of muscle deformation and deformation rate. US of muscle results in an interference pattern of acoustic markers (speckle pattern) that can be analysed post-process utilizing the US movie sequence of images (AVI format). This process is termed speckle tracking analysis (STA). During this process a region of interest frame (ROI; 10 × 2 mm) is positioned over a standardized location within the speckle pattern of each muscle in the first frame of the video sequence. The ROI tracks its contained unique speckle pattern frame by frame through the movie sequence. As the contained speckle pattern changes length between frames with muscle activity so does the length of the ROI. This change in ROI length represents muscle deformation. The muscle deformation measure is expressed as the percentage change in the longitudinal median length of the ROI compared with that at rest (% strain). The rate of change in length of the ROI is measured as muscle deformation rate, which is deformation per time unit (expressed as % strain 1/s).
STA utilizes software containing a speckle tracking algorithm (based on a stable mathematical model (Farneback), the algorithm used was the implemented version in the open source computer vision library (OpenCV) version 2.0 (http://opencv.willowgarage.com/wiki/)) to identify and track the ROI in the movie sequence. In the speckle tracking-algorithm, the ROI is divided into 50 equidistant smaller ROIs. Displacement is calculated using Farneback algorithm for each small ROI independently. A linear least squares fit assuming a linear strain model was then solved for the displacements of the ROI end-points. We thereafter summed end-point displacements across successive frames to determine the cumulative end-point motion, which was then used to estimate the cumulative strain.
The software (5), which is currently used only for research purposes, accepts that the identical muscle region (the speckle pattern) is being tracked accurately if there is at least 80% agreement in the speckle pattern between frames (5). The calculating algorithm measures ROI deformation (the ROI moves along with the movement of the underlying speckle) in each sequential frame, comparing it with the original length recorded at rest in the initial frame. The position of the ROI for each muscle examined between participants was standardized to the mid-point of the muscle belly and orientated longitudinal to the muscle fibres.
Experimental procedure
Participants were positioned in a standardized, clinically neutral lumbo-pelvic sitting posture with no back support and feet flat on the floor (15). The participant’s right arm rested on a treatment couch to minimize any shoulder girdle muscle activity, in 70° of shoulder flexion with the elbow extended and the forearm in neutral pronation/supination. Participants held a hand weight (2.5 kg for male and 1.5 kg for female participants) that rested on a platform trigger linked to the US machine so that the moment of lift could be electronically recorded within the US video.
The lifting task was performed in the 3 different cervical postures in a randomized order between participants: neutral (Fig. 2A), flexed (Fig. 2B) and FHP (Fig. 2C). The neutral posture was achieved by guiding the participant into a clinically determined neutral orientation of the cervical spine and head. An inclinometer centred over the tragus of the left ear was set at 0° in this position so that the flexed and FHP were standardized relative to the participant’s neutral posture. The flexed posture was achieved by guiding the participant into 30° of lower cervical flexion, guided by the inclinometer. The FHP was achieved by first positioning the participant in the flexed posture, followed by facilitating upper cervical extension (maintaining lower cervical flexion) so that the participant’s line of sight was straight ahead. An investigator monitored and ensured the participant maintained the required postural alignment throughout each lifting condition.
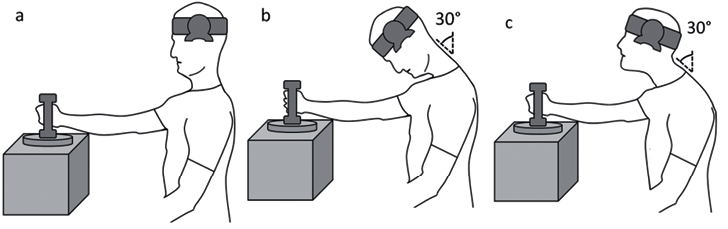
Video recordings for each of the 3 lifting conditions were commenced with the head in a neutral posture first. These initial parts of the recordings (recording started after holding the head in resting neutral position for at least 3 s) were utilized as the resting reference values for the post-process calculation of muscle deformation and deformation rate for the experimental resting posture (neutral, flexed, FHP) and for each lift condition. Following the reference recordings participants were guided into and sustained the experimental neck posture (remain in neutral or positioned in the flexed or FHP) for 2 s. They were then instructed to lift the weight off the trigger using a set count of “1, 2, 3” and hold it still below shoulder height for 3 s before lowering it back down to the supporting surface, at which point video recordings ceased (about 15-s recordings). During each trial an electronic marker was recorded within the video sequence registering the point at which the participant maintained the experimental neck posture (triggered by the investigator) as well as the point of lift as registered by the weight being lifted off the trigger.
Data management and statistical analysis
Post-process STA was utilized to calculate deformation and deformation rate values (reference resting value in the initial neutral head position subtracted) for 3 time-periods; time-point 1 – experimental rest posture, time-point 2 – mid-lift, calculated for the 0.2 s time-period before the trigger registered the weight being lifted, as we hypothesize that muscles were already active before the weight was lifted off due to a slight trigger delay, and time-point 3 – end-lift calculated for a 0.2 s time-period between 0.2 and 0.4 s following the trigger registering the lift. Values were expressed as root mean square (RMS) values for these time-periods for analysis.
Statistical analysis was performed using SPSS (version 20). Descriptive statistics (mean and standard deviation) were calculated for the RMS measurements. A repeated-measures general linear model was used to examine the main effects of condition (neutral, flexed, FHP) and time (rest, mid-lift, end-lift) for each dorsal muscle. Main effects for condition, time, and condition × time interactions were evaluated for both deformation and deformation rate. Tests for simple effects were performed post-hoc when indicated. Statistical significance was set at p ≤ 0.05.
RESULTS
Muscle deformation measurement
There was a significant main effect of condition (p < 0.005) but not time (p > 0.06), for the deformation measure recorded from all dorsal neck muscles. Significant condition by time interactions were observed for the trapezius, semispinalis cervicis and multifidus muscles (p < 0.04) (Fig. 3A–C), but not the other dorsal neck muscles (p > 0.14). Tests of simple effects revealed that muscle deformation values at rest for the flexed and FHP conditions were significantly greater than in the neutral posture for all muscles (p < 0.01) and only different at rest between the flexed and FHP condition (p < 0.01) for the trapezius muscle (higher in the flexed posture) (Table II). The only significant changes in dorsal neck muscle deformation between the rest and the end-lift time-points occurred in the neutral posture (p < 0.03) and not the flexed or FHP conditions (p > 0.07) (Table II and Fig. 3).
|
Table II. Root mean square values (RMS) with standard deviation (SD) of deformation (%) for each muscle |
|||||||||
|
Muscle |
Neutral |
Flexed |
Forward head |
||||||
|
Rest RMS (SD) |
Mid-range RMS (SD) |
End RMS (SD) |
Rest RMS (SD) |
Mid-range RMS (SD) |
End RMS (SD) |
Rest RMS (SD) |
Mid-range RMS (SD) |
End RMS (SD) |
|
|
Trapezius |
0.61 (0.47) |
0.75 (0.69) |
1.35 (1.12)* |
5.78 (3.66) |
5.30 (3.66) |
5.42 (3.58) |
3.14 (2.25) |
3.18 (2.37) |
3.32 (2.22) |
|
Splenius |
1.13 (0.91) |
1.44 (1.30) |
2.57 (2.87)* |
11.07 (8.64) |
11.10 (9.30) |
11.57 (9.84) |
7.82 (6.39) |
7.60 (6.04) |
7.46 (5.03) |
|
Semispinalis capitis |
1.14 (0.89) |
1.58 (1.03) |
2.28 (2.03)* |
10.30 (7.75) |
10.42 (7.50) |
10.41 (8.47) |
8.63 (8.67) |
8.75 (8.51) |
9.03 (8.21) |
|
Semispinalis cervicis |
1.90 (1.55) |
2.17 (1.68) |
3.13 (1.60)* |
9.60 (7.49) |
9.11 (7.41) |
8.75 (7.37) |
10.23 (6.87) |
10.14 (6.96) |
10.23 (6.84) |
|
Multifidus |
2.06 (2.63) |
2.32 (2.84) |
3.92 (4.17)* |
15.90 (15.75) |
15.56 (15.22) |
15.78 (15.44) |
11.37 (8.21) |
11.51 (8.04) |
11.28 (7.70) |
|
*Denotes significant time-effect at end of lift relative to rest (p < 0.05). SD: standard deviation; RMS: root mean square. |
|||||||||



Fig. 3. Deformation data (mean root mean square and standard deviation) for each posture condition over the 3 time-points for the muscles shown to have significant condition by time interactions. (A) Trapezius. (B) Semispinalis cervicis. (C) Multifidus. *Significant change in deformation compared with rest (p < 0.05).
Muscle deformation rate measurement
There were significant main effects of time for all dorsal muscles (p < 0.001) and significant main effects of condition for the semispinalis cervicis muscle (p = 0.02), but not the other muscles (p > 0.1). There was a significant condition by time interaction for the semispinalis cervicis muscle (p = 0.02) (Fig. 4), but not for the other dorsal neck muscles (p > 0.1).
Tests of simple effects revealed no significant differences in muscle deformation rate values at rest between the different postural conditions, with the exception of the semispinalis capitis muscle, which showed a higher deformation rate for flexed and FHP conditions, compared with the neutral posture (p < 0.03). There were significant increases in deformation rate between the rest and the end-lift time-points in all dorsal neck muscles during the lifting task in all 3 postural neck conditions (p < 0.04) (Table III and Fig. 4).
|
Table III. Root mean square values (RMS) with standard deviation (SD) of deformation rate (% deformation 1/s) for each muscle |
|||||||||
|
Muscle |
Neutral |
Flexed |
Forward head |
||||||
|
Rest RMS (SD) |
Mid-range RMS (SD) |
End RMS (SD) |
Rest RMS (SD) |
Mid-range RMS (SD) |
End RMS (SD) |
Rest RMS (SD) |
Mid-range RMS (SD) |
End RMS (SD) |
|
|
Trapezius |
0.003 (0.002) |
0.007 (0.005) |
0.019 (0.013)* |
0.005 (0.007) |
0.007 (0.008) |
0.014 (0.018)* |
0.004 (0.002) |
0.007 (0.006) |
0.021 (0.026)* |
|
Splenius |
0.005 (0.002) |
0.015 (0.014) |
0.031 (0.02)* |
0.007 (0.009) |
0.012 (0.017) |
0.018 (0.016)* |
0.005 (0.002) |
0.013 (0.017) |
0.036 (0.038)* |
|
Semispinalis capitis |
0.005 (0.002) |
0.011 (0.008) |
0.024 (0.011)* |
0.007 (0.005) |
0.011 (0.007) |
0.022 (0.023)* |
0.007 (0.004) |
0.010 (0.007) |
0.032 (0.029)* |
|
Semispinalis cervicis |
0.007 (0.003) |
0.013 (0.008) |
0.032 (0.014)* |
0.007 (0.004) |
0.011 (0.009) |
0.023 (0.017)* |
0.008 (0.003) |
0.010 (0.007) |
0.040 (0.030)* |
|
Multifidus |
0.007 (0.003) |
0.014 (0.011) |
0.031 (0.019)* |
0.007 (0.004) |
0.010 (0.008) |
0.024 (0.025)* |
0.009 (0.005) |
0.011 (0.008) |
0.026 (0.019)* |
|
*Denotes significant time effect at end of lift relative to rest (p < 0.05). |
|||||||||


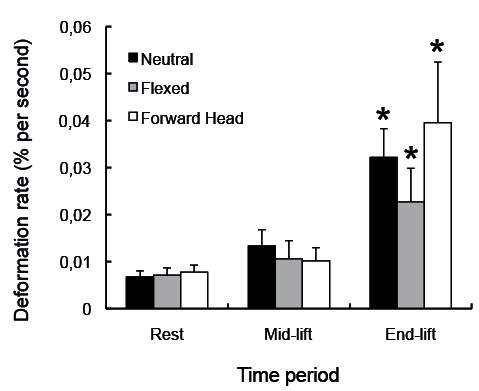
Fig. 4. Deformation rate data (mean root mean square and standard deviation) for each posture condition over the 3 time-points for the semispinalis cervicis muscles shown to have a significant condition by time interaction. *Significant change in deformation compared with rest (p < 0.05).
DISCUSSION
Detection of multilayered muscle activity
This study investigated an application of US to quantify mechanical measures (muscle deformation and deformation rate) of muscle activity in multiple muscle layers simultaneously without the need for invasive procedures such as intramuscular EMG.
The deformation measure (amplitude) was significantly different between the different postural neck conditions in all dorsal muscles and differences were largely explained by the larger deformation values for the flexed and FHP conditions at rest (in the postural position prior to the lift) compared with the Neutral posture. These findings of heightened dorsal muscle deformation in flexed and FHP postures are consistent with previous studies using EMG (15–19) and reflect the greater antigravity demands on these dorsal neck muscles when the head is held anterior to the thorax (20). The elevated muscle deformation levels at rest in the flexed and FHP conditions may also explain the lack of significant changes in deformation during the lift in these postures (deformation only increased with lift in the neutral posture) (Table II and Figs 3A–C). These findings may indicate that the dorsal neck muscles did not require as much change in mechanical muscle activity in the flexed and FHP (they were already in a state of contraction) to counteract the addition of the lifting load as was required in the neutral posture. Alternatively, the findings may indicate that the deformation measure was not sensitive enough to detect small changes in deformation when the muscles were already in a state of contraction in these flexed and FHP. While these considerations are speculative, the significant increase in deformation observed during lifting in the neutral posture compared with the flexed and FHP conditions explains the significant condition by time interactions observed for most of the dorsal neck muscles.
In contrast to the deformation measure, the deformation rate measure was not significantly different between the postural conditions (with the exception of the semispinalis cervicis muscle) (Fig. 4). Similarity between the deformation rate measures between postures at rest may indicate that these 3 static postural orientations require different amplitudes of muscle deformation to sustain the position, but by virtue of their static nature they require negligible changes in deformation rate to maintain the postures. Nevertheless, deformation rates increased significantly in all muscles during the lift for all 3 postural conditions, and this was different from muscle deformation, which only increased in the neutral posture (Table III). Potentially, deformation rate may be more sensitive to mild changes in activity, as induced by the lift in the different neck postures, whereas deformation measures are not (flexed and FHP). While these hypotheses for the observed differences in deformation and deformation rate measurements are yet to be validated, it appears that both measurements may be valuable in evaluating motor behaviour, but reflect different components of a mechanical muscle event (strain vs strain rate).
The capacity to quantify the differential activity of the multiple muscle layers of the cervical spine has potentially important clinical applications. The altered coordination between the deep and superficial muscle layers of the neck during the control of posture and motion in subjects with neck pain is well documented (1–4, 23). STA provides a potentially valuable clinical tool to quantify muscle activity non-invasively. Thus, STA may be highly beneficial to clinicians in the future when assessing muscle function and evaluating the effect of therapeutic exercise in patients with neck pain.
Clinical application
The findings of this study indicate the potential future use of these measurements in the clinical setting. Some initial evidence of a relationship between voluntary muscle contraction and deformation has been shown (14); however, further validation of the US measure is required and is a limitation of the measure in the present study. The STA method of measurement has been shown to have good test-retest reliability for the measurement of deformation/deformation rate in the cervical muscles (intraclass correlation coefficient (ICC) 0.71–0.99) (Peolsson, A., unpublished data) as well as in other structures such as the Achilles tendon (ICC 0.81–0.99) (21). Measurements utilizing US also have advantages over other methods, such as intramuscular EMG, in that they are non-invasive, and unlike surface EMG (18, 19, 22) they can record multiple muscle layers. The capacity to separately evaluate deeper muscle layers is necessary, as muscle function magnetic resonance imaging (mfMRI) studies have indicated that factors, such as mild alterations in posture during clinical tests, can significantly affect the activity of the dorsal cervical extensor muscle layers (1). Similarly, this study demonstrates apparent differences in activity between the different muscle layers in response to the different postural orientations of the neck. This new US measurement method may also have some advantages in the future over mfMRI, as deformation of muscles can be evaluated at the time of exercise event, rather than post-event activity (as is the case with mfMRI), and is a far less expensive alternative.
Methodological considerations
Speckle tracking from video sequences of ultrasonography enabled us to use a non-invasive method of real-time imaging for mechanical deformation of multiple muscle layers. Due to our post-recording method of analysis we could clearly identify margins for correct muscle labelling and ROI remained tagged, avoiding any difficulties associated with visualizing fascia between small deep muscles. The US technique, however, was limited to 2 dimensions. Any rotational motions could not be measured. In addition, muscles were in slightly different planes during recording, which might impact on the relative deformation of each muscle. The measure is unable to provide an exact representation of activity levels in entire regions of the muscle.
In the present study US imaging was recorded by an experienced sonographer following a standardized process of image acquisition and analysis. As it is acknowledged that accurate US imaging is dependent on the skill level of the sonographer, we anticipate that the findings of this study would be reproducible if performed by a sonographer of similar experience and level of skill utilizing the same standardized image acquisition and analysis procedure.
We standardized the weight and performed only one repetition of the arm lift over a very short period of time. It is possible that the results might have been different if we utilized heavier weights or higher numbers of lifting repetitions. However, we chose these load and repetition parameters as we considered it more meaningful if the measurements could detect differences in motor behaviour under these relatively gentle test conditions, which would be appropriate for a patient with neck pain. In addition, at present, longer recording times than 15 s are not possible with our US equipment due to the large file size and processing restrictions.
This study was undertaken on a healthy population sample. The findings cannot be extrapolated directly to clinical populations due to the known differences in muscle behaviour in healthy and neck pain populations (23). Future studies should examine the differences between healthy and neck pain populations to clarify further the utility of the measurements in the clinical setting.
Conclusion
This study has shown that a new non-invasive method of measuring muscle activity using US-based mechanical muscle measures can detect differences in muscle activity of the multilayered dorsal neck muscles during a lifting task in various neck postures. Further research is needed to validate this measurement method and to evaluate its use in the clinical setting.
ACKNOWLEDGEMENTS
The authors of this study declare there were no conflicts of interest. Shaun O’Leary is supported by a Health Practitioner Research Fellowship (Queensland Health and University of Queensland (NHMRC CCRE Spinal Pain, Injury and Health)). Anneli Peolsson is supported by the Swedish Medical Research Council and the Wenner-Gren Foundations.
REFERENCES
