Indre Bileviciute-Ljungar, MD, PhD1,2, Vera Häglund, MD2, Jenny Carlsson, MD3 and Anders von Heijne, MD3
From the 1Department of Clinical Sciences, Karolinska Institutet at Danderyd University Hospital, 2Department of Rehabilitation Medicine and 3Department of Radiology, Danderyd University Hospital, Stockholm, Sweden
OBJECTIVE: To increase awareness of the incidence of delayed leukoencephalopathy in rehabilitation medicine.
Subject: A 34-year-old male patient in an inpatient neurorehabilitation clinic who developed cognitive, psychological and physical deterioration 33 days after methadone intake.
METHODS: Clinical follow-up for 7 months, brain imaging with magnetic resonance imaging and computed tomography, electroencephalography, multidisciplinary team evaluation and rehabilitation, pharmacological treatment, and examination of medical records.
RESULTS: Clinical findings showed neuropsychological and motor deterioration. Brain images demonstrated that previous white matter infarctions had developed to cystic substance defects, and that abnormally high signals developed in the white matter of most cerebral lobes, with the exception of the grey matter and the cerebellum. Clinical improvement coincided with a modification in pharmacological treatment (increase in sertraline and introduction of baclofen). Brain images at 3 and 6 months after the methadone overdose showed reduced intensity of signal abnormalities and complete normalization of diffusion weighted images. Evaluation 7 months after injury estimated moderate brain injury with moderate disability and partial recovery of the patient’s capacity for previous activities of daily living.
CONCLUSION: Delayed leukoencephalopathy should be suspected in patients who deteriorate after methadone overdose. Drugs such as sertraline and baclofen may be of use in treating delayed leukoencephalopathy.
Key words: methadone-induced delayed leukoencephalopathy; sertraline; baclofen; rehabilitation outcomes; demyelination.
J Rehabil Med 2014; 46: 00–00
Correspondence address: Indre Bileviciute-Ljungar, Department of Rehabilitation Medicine, Danderyd University Hospital, Building 39, 3rd Floor, SE-182 88 Stockholm, Sweden. E-mail: indre.ljungar@ki.se
Accepted Feb 25, 2014; Epub ahead of print Jun 4, 2014
Introduction
Delayed leukoencephalopathy (DLE) was described as early as 1925, and has been studied mostly in cases of hypoxic carbon monoxide poisoning (1). Recently, there has been an increased number of case reports of methadone-induced DLE, indicating a need for medical rehabilitation professionals to be more aware of this syndrome (1–5). Most of these case reports describe radiological findings that are considered important for diagnosis (2, 5); however, few reports describe the clinical outcome, which may range from a continuous need for assistance in activities of daily living (ADL) (1) to full recovery (2). One report of a particularly severe case of methadone-induced DLE details successful pharmacological treatment using intravenous methylprednisolone, amantadine, vitamin C and vitamin E (3).
The aim of this study was to report the clinical and radiological characteristics of a patient with methadone-induced DLE, the pharmacological treatment applied, and the outcome, as evaluated using the Extended Glasgow Outcome Scale (GOSE).
Case Report
Acute phase
A 34-year-old man with previous full work capacity was found at home after an overdose of methadone tablets he had purchased on the black market. The patient had a 12-year history of depressive disorders, treated with sertraline 100 mg/daily and sporadic abuse of anxiolytic substances. Upon initial examination, his breathing was sporadic (3–4 breaths/min), circulation was remarkably decreased (10% of ejection fraction of the left ventricle measured by echocardiography) and his estimated consciousness was between 8 and 9 on the Glasgow Coma Scale. The initial methadone concentration in his blood was high (2,000 ng/ml) and benzodiazepine was detected in his urine, confirming that these drugs were the cause of the overdose. Restoration of cardio-pulmonary functions occurred approximately 18 h after the initial intake of drugs.
The patient was in intensive care for the first 8 days and remained at the department of internal medicine for the next 7 days. During this time, computed tomography (CT) of the brain (Fig. 1A) and an electroencephalogram (EEG) both appeared normal. However, his Montreal Cognitive Assessment test (MoCA) score 12 days after the overdose was 19/30, suggesting initial cognitive impairment. Thereafter, the patient was moved to the inpatient department for team-based neurorehabilitation. An MRI of the brain 23 days after the methadone overdose revealed bilateral parasagittal white matter changes cranial to the ventricles with a “string-of-beads” appearance on T2 and fluid attenuation inversion recovery (FLAIR) images. These observations were interpreted as gliosis after watershed infarctions that occurred due to hypoperfusion during the initial events (Fig. 1B).
Deterioration phase
An approximately 2-week, multi-professional, team evaluation was carried out by physicians, medical nurses, occupational therapist, physiotherapist, psychologist and social counsellor after admission to the department of rehabilitation. At the equivalent to day 31 after the overdose, summary evaluation showed that the patient continued to experience impairments in both motor skills and cognitive functions. He had difficulties with balance, coordination and walking (he used a wheeled walker), and thus required supervision in ADL. He had signs of muscular spasticity with easily evoked clonus in his legs, a positive Babinski sign, and generally lightly augmented reflexes. Neuropsychological screening showed lowered cognitive function (–2 standard deviations below the mean) in visuospatial tests, working and long-term memory tests, and an attention test. After multi-professional team evaluation an individual rehabilitation plan was proposed, comprising: (i) physical training to improve balance, co-ordination, muscle strength, endurance, ADL and hand functions; (ii) occupational therapy, individually and in a group, to improve cognition, memory, visuospatial capacity, executive function, attention, social interaction and hand functions; (iii) improvement in his socio-economic situation, since the patient had become unemployed and needed personal and community financial support; (iv) planning for further medical and psychiatric care after discharge, which also included permission to go home, training to use community transport under supervision, and assistive devices at home.
Thirty-three days after the overdose, the patient became manic and displayed exaggerated psychomotor aggressive behaviour. He became disorientated, spoke with exaggerated speech flow and responded in a confusing way. He also failed to perform an occupational task (baking a cake), which he had managed one week before, and lacked adequate insight into the situation. His motor skills suddenly became impaired, and he was not able to stand and walk with assistive help. Due to this manic behaviour, the patient’s dose of sertraline was reduced from 100 mg to 50 mg and an improvement in his semi-psychotic status was observed after 1 day. He continued to receive oxazepam as needed, zopiclone for sleep improvement and mometasone for mild skin psoriasis of the head and nails. A psychiatric consultation carried out after 3 days at the lowered dose of sertraline found no signs of psychiatric disorder. After 5 days, CT scans of the patient’s brain showed that there was no aggravated pathology; however, EEG results indicated mild abnormalities, primarily frontally, but without epileptic activity. A cognitive assessment performed 6 days after deterioration suggested extremely slow performance with a MoCA test score of 17/30 points. Consecutive routine laboratory tests regarding other medical conditions were normal (e.g. peripheral blood cells, electrolytes, liver enzymes, thyroid hormones, inflammatory parameters, glucose, vitamins, etc.). Deterioration continued for approximately 17 days with the following additional neurological impairments: increased spasticity and shaking of the left leg under physical effort, lack of visuospatial orientation on the left side and development of new symptoms not present before, such as urinary incontinence. The patient was confined to a wheelchair due to inability to walk, and required constant assistance with ADL. These complications significantly limited his rehabilitation process. After consultation with a neurologist at day 17 after deterioration, treatment with baclofen (5 mg, 3 times daily) was initiated in order to treat the increased spasticity. At the same time, the patient was mistakenly given an increased (150 mg) dose of sertraline for 3 subsequent days, after which the sertraline dose was continued at the initial dose of 100 mg. Interestingly, the patient showed remarkable improvement. Baclofen treatment was suspended after 9 days because the patient reported undesirable side-effects (muscle weakness). On day 18 following deterioration and prior to the pharmacological intervention, brain magnetic resonance images (MRI) demonstrated that the previously observed parasagittal white matter infarctions had progressed to cystic substance defects. Confluent hypersignals on FLAIR and T2 sequences in white matter in all cerebral lobes were apparent, though somewhat less pronounced in the temporal lobes (Fig. 1C). Central structures, brainstem, cerebellum and grey matter were spared and showed no abnormality. The affected areas also showed high signal intensities using diffusion-weighted imaging (DWI) due to T2 shine-through, since the apparent diffusion coefficient was normal. Based on the imaging results and clinical history, the patient was diagnosed with DLE.
By day 20 after deterioration, the patient’s cognitive ability had improved significantly and the rehabilitation process was continued according to the individual plan. The MoCA test score had increased to 21/30 and, when re-tested on day 60 after the overdose (the day of discharge to outpatient rehabilitation), it was completely normal (30/30). The patient left the inpatient department of rehabilitation with little motor impairment. He maintained moderate cognitive impairment and had an estimated GOSE score of 5, indicating lower moderate disability. At the outpatient rehabilitation the patient continued with: (i) physical training as described before during the inpatient rehabilitation; (ii) individual cognitive training with computer program CogMed QM and group training to improve memory and executive functions; (iii) individual psychological support; (iv) group education for patients with brain injuries; and (v) a return-to-work programme. Outpatient rehabilitation lasted for 3 months. At the time of discharge from outpatient rehabilitation (approximately 7 months after methadone intake), the GOSE score had improved to 6, meaning upper moderate disability with ability to perform some previous activities. Subsequent MRI analyses were performed at 3 and 6 months after the overdose and showed a slightly reduced intensity in the signal abnormalities on FLAIR images (Figs 1D and E, respectively), and complete normalization of DWI.
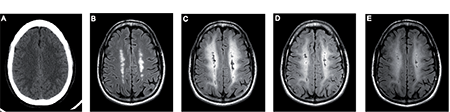
Fig. 1. Radiological images at a level directly above the ventricles. (A) Computed tomography was normal on the day of admission. (B) T2 fluid attenuation inversion recovery (FLAIR) magnetic resonance (MR) image on day 23 shows white matter changes cranial to the ventricles believed to represent deep watershed infarcts. (C) T2 FLAIR MR image 18 days after exacerbation shows extensive confluent white matter hyperintensity, consistent with a diagnosis of delayed leukoencephalopathy. (D and E) T2 FLAIR MR images from follow-up at 3 and 6 months, respectively, after the initial event show slight successive reduction in white matter hyperintensity.
Discussion
Conditions that may lead to DLE include hypoxaemia, cerebral vasodilatation and hypotension, as well as abuse of toxic substances. A combination of several circumstances most likely contributed to DLE in the present case, since the cystic changes in the white matter were mostly prominent in the area already damaged by watershed infarctions. A role of pseudodeficiency of arylsulphatase-A (ASA), an enzyme essential for myelin turnover, or delayed apoptosis of oligodendrocytes responsible for myelin production, is suggested, but not consequently found, in DLEs (1–3). Caroll et al. (6) suggested that ASA pseudodeficiency might represent an entirely incidental finding among patients with DLE, since it is a common condition with an incidence of 2%. Because of our unawareness of DLE, we did not measure the specific markers of either demyelination in cerebrospinal fluid or ASA pseudodeficiency in the peripheral leukocytes.
It is interesting to note that pharmacological modification with selective serotonin re-uptake inhibitors and gamma-aminobutyric acid (GABA) receptor agonists lead to significant clinical improvements that might be of interest in the treatment of DLE. Baclofen is a selective GABAB receptor agonist widely used for the treatment of spasticity (7). It is difficult to speculate about the effect of baclofen in this case study, since the distribution of GABAB receptors in the human brain and the precise mechanism of action are unknown (8). Experimental results from in vitro studies with human peripheral blood mononuclear cells and in vivo studies of contact dermatitis in mice suggest that baclofen has an anti-inflammatory effect (9). Serotonergic axonal injury has been suggested to be a cause of toxic leukoencephalopathies induced by 3,4-methylenedioxymethamphetamine (MDMA or “ecstasy”) (for review see 10), which may also be a cause in the present case. However, our clinical experience suggests that methadone overdose can induce a complex delayed deterioration with multiple symptoms and the development of completely new neurological and neurocognitive symptoms, and that pharmacological intervention, for example with a low dose of baclofen, might be useful in the treatment of patients with DLE.
Acknowledgement
The authors would like to thank Professor Jörgen Borg at the Department of Clinical Sciences, Karolinska Institutet, for valuable discussions of this case.
References
