Marie O. Frenkel, PhD1*, Daniela S. Herzig, MD2*, Florian Gebhard, MD, PhD2, Jan Mayer, PhD3, Clemens Becker, MD, PhD4 and Thomas Einsiedel, MD, PhD2,5
From the 1Department of Sport Psychology, Institute of Sport and Sport Science, University of Heidelberg, Heidelberg, 2Department of Traumatology, Hand-, Plastic-, and Reconstructive Surgery, Center of Surgery, Center of Musculoskeletal Research, University of Ulm, Ulm, 3German University for Prevention and Health Management, Saarbruecken, 4Clinic for Geriatric Rehabilitation, Robert-Bosch-Hospital, Stuttgart and 5Private Practice for Trauma Surgery and Orthopedics, Hechingen, Germany. *These authors contributed equally to this study and manuscript.
OBJECTIVE: To determine whether mental practice of wrist movements during forearm immobilization maintains range of motion.
DESIGN: Randomized controlled trial.
PARTICIPANTS: Eighteen healthy young men aged between 20 and 30 years were assigned to either a control or a mental practice group. Both groups were immobilized with a circular forearm cast for 3 weeks to simulate a distal radial fracture.
METHODS: The mental practice group received 1 × 60-min, followed by 3 × 30-min sessions of supervised mental practice. Consecutively, they were asked to perform 15 min/day of self-guided imagery sessions, during which they mentally exercised motion sequences of the immobilized joint. The training program followed the Mental Gait Training procedure. The control group did no training. Wrist movement was measured with a goniometer before and after immobilization.
RESULTS: Mental practice preserved dorsal extension and ulnar abduction. The sedentary control group showed due to this variables a significant decrease after cast removal. There was no significant change in palmar flexion and radial abduction in either group.
CONCLUSION: Despite the study limitations, these results suggest that mental practice may be useful in preventing loss of hand function associated with mid-term immobilization. Because of the expected clinical benefits, the low cost and simple application of the intervention, the effects of mental practice in orthopedic rehabilitation of the upper extremity warrant further study.
Key words: mental practice; rehabilitation; hand; immobilization.
J Rehabil Med 2014; 46: 225–232
Correspondence address: Marie Ottilie Frenkel, Im Neuenheimer Feld 700, 69120 Heidelberg, Germany. E-mail: marie.frenkel@issw.uni-heidelberg.de
Accepted Sep 18, 2013; Epub ahead of print Feb 10, 2014
INTRODUCTION
Many orthopedic patients are immobilized for several weeks after operations or injuries that do not allow for weight-bearing. During this recovery period immobility leads to serious complications, including reduction in range of motion and muscular atrophy. Furthermore, on a cortical level, immobilization results in central reorganization (1, 2), resulting in the loss of movement representations and inefficient central control of movement (3). In younger persons this can lead to an extended period of reduced occupational capacity, followed by an extended period of rehabilitation generally leading to full functional and social recovery. In older persons this can cause massive and sometimes irreversible functional and social problems, resulting in nursing home placement or care dependency. Immobilization of the upper limb therefore poses a highly relevant clinical problem. Regardless of how the injury is treated, whether surgically, by casting, or by relative immobilization, patients are immobilized for between 4 and 12 weeks. Exercise intervention usually does not start during the immobilization period. The goal of rehabilitation is to achieve complete and rapid recovery of range of motion, muscle strength, and function of the joint and the limb. In order to improve functional outcome, early rehabilitation could offer an advantage in the functional status or accelerate the process of recovery after immobilization (4). The treatment procedure should be proactive without stressing the bone and soft tissue. This may prevent, not only the negative physiological side-effects, but also the negative effects of central reorganization (5).
Several studies have shown that sensory input is not only the result of performed movements, but also results from simply imagining movements of the limbs (6, 7). Motor imagery is defined as an active process during which the representation of a specific motor action is internally reproduced within working memory without any overt motor output. It is also governed by the principles of central motor control (8). Images of a motor act focus on visual or kinesthetic information rather than on the other senses. Visual imagery refers to the ability to see an object or scene in your mind. It is usually static and from a third-person perspective. For example, visual imagery is accomplished when you imagine a hand with a bended wrist holding an insect between 2 fingers. Kinesthetic imagery, in contrast, requires the ability to feel, and thus to experience the somato-sensory feelings related to the movement, i.e. to perceive muscles contractions mentally. This modality is usually from the first-person perspective and involves dynamic motion (9). Motor imagery often designs a singular internal reproduction of a given motor act. If this is repeated extensively, consciously, systematically and with the intention of improving performance, it is termed mental practice (10).
Several studies have shown that mental practice follows the same movement rules and constraints as physical movements, including temporal regularities (11), programming rules such as Fitts’s law (12), kinematic constraints (11) and vegetative responses (13). Furthermore, both motor execution and mental practice seem to positively affect skill acquisition (14) and motor performance (15). These functional similarities between physically executed and kinesthetically imagined movements are explained within the mental simulation theory (16) by a partially overlap of neural substrates, more precisely of motor and sensory regions. However, the neural activation for the imagined movements seems to be less intense and less localized than when actually performing the task (17). The importance of physical experience in order to achieve an overlap between imagery and execution has been highlighted in several papers (18). This is particularly important when designing effective training protocols in rehabilitation.
Based on the neurophysiological findings and on a considerable number of experimental studies with healthy adults that demonstrate positive effects of mental practice on physical performance (19), several research groups have suggested the use of mental practice in physical rehabilitation as a simple and low-cost tool to promote motor recovery (10, 20–23). The main idea of this approach is that patients in orthopedic or neurological rehabilitation may benefit from the isomorphic neural processes described. Particularly those patients who, as the result of a fracture treated by cast, an endoprosthetic/surgical treatment operation, or paralysis after stroke, are either immobilized or still too weak to perform active physical training, might benefit from this cognitive technique.
In orthopedic rehabilitation, mental practice has slowly gained attention as a promising psychological complement to conventional exercise therapy approaches. Compared with the empirical evidence in neurological rehabilitation, fewer studies can be found. The potential of mental practice following injuries or amputation of the lower extremity has been demonstrated (24–27). In literature about orthopedic rehabilitation, however, the influence of mental practice on range of motion is hardly discussed. Although effects of mental practice on range of motion have not been shown before in an applied rehabilitation study after immobilization, an increase in flexibility has been registered in competitive athletes (28). In another study of healthy adults, mental practice coupled with proprioceptive neuromuscular facilitation resulted in a better range of motion of the hip joint than physical training used alone (29). A randomized controlled trial with patients after knee endoprosthesis showed a significant improvement in knee flexion after a 6-week mental training program (24).
In the context of immobilization of the upper extremity, a patient who underwent tendon transfer of the upper limb was treated by mental practice for elbow extension, resulting in enhanced motor recovery of elbow movement (30). A comparable study involving short-term muscle immobilization of the hand using a randomized controlled trial with healthy adults explored the effects of mental practice on the strength of the fingers (31). The results suggest that mental practice is useful in preventing the loss of strength associated with immobilization. Patients using mental practice after flexor tendon repair of a finger injury required less time for movement preparation, which can be interpreted as an improvement in the central aspects of hand function (32).
To best of our knowledge the use of mental practice during the immobilization of the wrist has not yet been described in the literature. Conducting a pilot study with healthy volunteers should be a good first step for exploring the recuperative potential of mental practice during wrist immobilization. We used a training paradigm that combined physical execution and mental rehearsal trials with observation from clinical practice so as to maximize adherence and learning effects (24, 33).
Brain imaging and behavioural studies have shown that, compared with the visual modality and a third-person perspective, the kinesthetic type of imagery from the first-person perspective share more physiological characteristics with a real executed movement (9, 34).
The main aim of this randomized prospective pilot study was to determine whether mental practice during a 3-week immobilization of healthy wrists would result in a greater preservation of the range of motion, one essential measure of hand function, compared with a non-exercising control group. Mental practice may have this physiological effect on range of motion through the impact at a cellular level on the sarcomeres or connective tissues (28).
METHODS
A total of 20 healthy male volunteers with a mean age of 25 years (standard deviation (SD) 4.29; range 20–30 years) replied to a newspaper advertisement inviting people to join the study. The participants were right-handed and lived in social circumstances that allowed the wearing of a forearm cast for 3 weeks. Other inclusion criteria were the absence of a prior injury of the left hand or arm. Exclusion criteria were a history of a neurological disease or any untreated or unstable medical condition. Two people were rejected as a result of these criteria (Fig. 1).
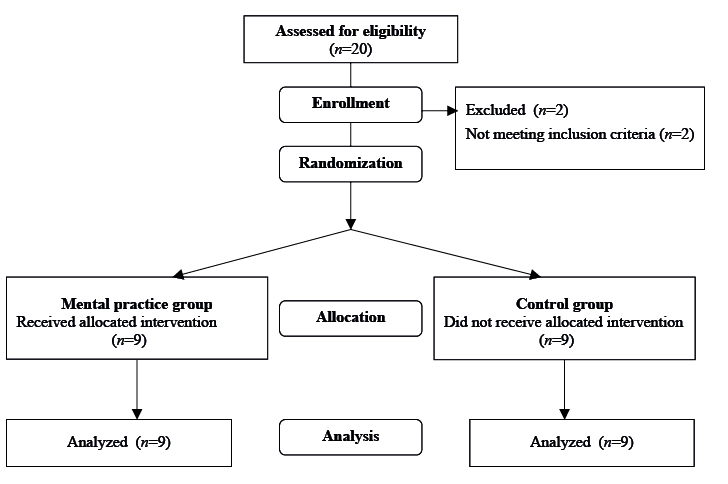
The participants were paid 150 €. In the mental practice group the compensation was paid after the follow-up assessment and with the delivery of a fully completed training-diary. This booklet was applied to augment compliance of the participants and to control whether they practiced regularly at home. Inside the diary we stated that effects through mental practice only by exercising regularly and we asked participants to keep the diary accurately. Participants noted the date, time and duration of their individual training. The study consisted of 2 test procedures: pre-intervention (baseline) and post-intervention testing. After baseline assessment subjects were randomly assigned to either the control group or the mental practice group. Due to resource problems the unblinded assessment and the randomization process were performed by TE/DSH.
The training procedure was carried out by 2 physiotherapists (BG/JT) who were not involved in any other part of the study. After the baseline assessment both groups underwent fitting of a circular forearm cast for immobilization of their non-dominant (left) radiocarpal joint for 3 weeks.
All participants gave their written informed consent. The procedures were approved by the ethics committee of the University of Ulm (No. 275/2004).
In each assessment session a clinical evaluation was applied to the participants. Range of motion of the radiocarpal joint was measured in 4 clinical parameters with a goniometer (35): dorsal extension, palmar flexion, radial and ulnar abduction of the wrist joint.
The participants in the mental practice group underwent guided training in the hospital as well as unsupervised training at home. Both procedures followed a specific protocol. Concerning the supervised training they were progressively instructed to practice the wrist-movements mentally during a 60-min session. The same tasks were re-discussed, performed and checked by the physiotherapists in 3 more guided sessions for 30 min over the next 3 weeks. All participants in the mental practice group were asked to practice individually at least 15 min every day. Each participant maintained the training diary mentioned above in order to record the regularity of this independent training.
The applied training program followed the standardized method of Mental Gait Training (25, 36). The general procedure (Fig. 2) consists of 6 levels: 1: movement; 2: description; 3: instruction; 4: practice (4a: contralateral physical/4b: mental); 5: movement representation; and 6: execution of the movement.
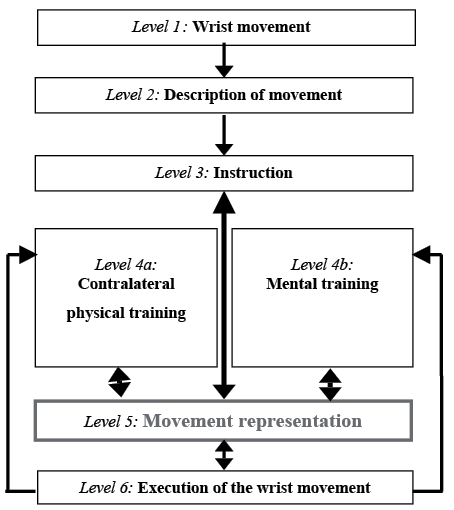
Activated inner concepts of the chosen movement (level 1), in this case the wrist movement, were important requirements of subsequently imaging the movement task. Therefore the movement had to be described first on an objective level (level 2). The physiotherapist explained the biomechanical details of the trained movement by showing his own hand as a model. In order to achieve greater learning and retention results, the trainees verbalized the motor task, and later recorded the task in written form (37). This was also a good means of intervention as it helped avoiding mistakes and enhanced precision. Based on the characterization of the movement, instructions (level 3) were worked out. The so-called “nodal points” (38), which are the key steps in the course of the inner map, were developed and emphasized by individual commands. Mental practice (level 4b) supplemented by contralateral physical training (level 4a) differentiated and stabilized the representations of the movements (level 5). Observation of the non-immobilized extremity (contralateral physical training) provided useful additional feedback (23) concerning, for example, posture or muscle tension in the trainees. The representation controls and actuates the execution of the motor task (level 6), because it functions as a reference pattern or blueprint of the actual performance. Therefore, functional representation formed a substantial factor of the quality of the movement: it supports the (re-)learning and optimizing of the performance of the movement.
To apply level 4a and b of the Mental Gait Training we used the following method (Fig. 3).
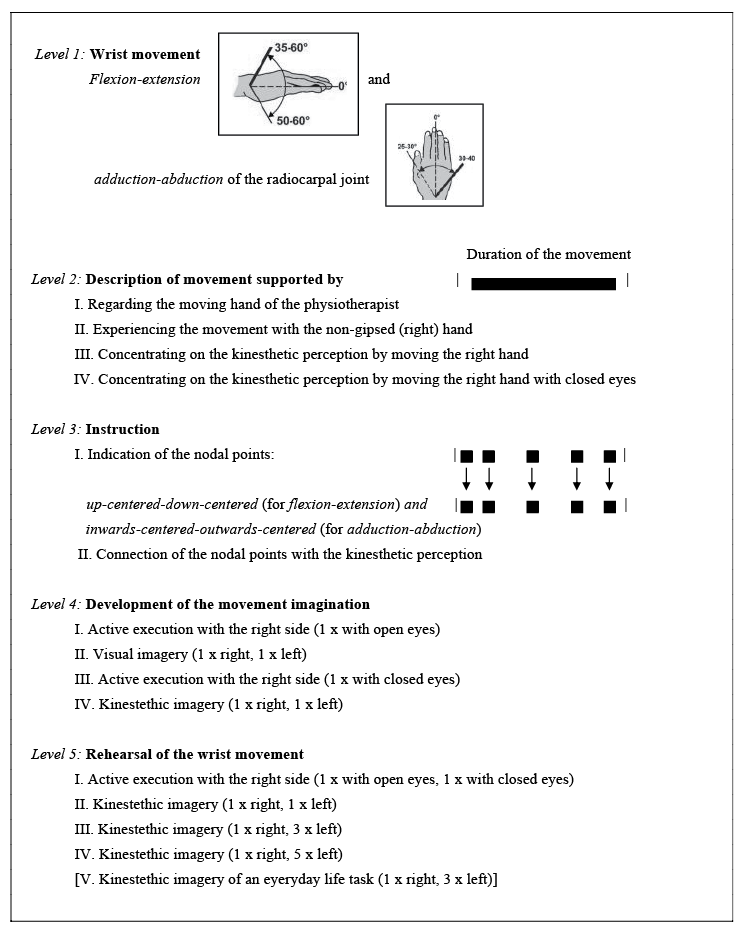
The trained wrist movements were flexion-extension and adduction-abduction of the radiocarpal joint (level 1). The “nodal points” were up-centred-down-centred and inwards-centred-outwards-centred (level 3). In order to develop the differentiated movement imagination (level 4) participants observed, but also experienced, the movements with their right (non-immobilized) hand and were asked to pay attention to their kinesthetic perceptions during the movement. To enforce the kinesthetic perception they duplicated the movement with closed eyes.
After having developed this individually customized internal concept of the movement, movement imagination was rehearsed systematically (level 5) following 3 stages: (i) active execution with the right side (1 × open and 1 × closed eyes; level 5.I); (ii) variations of the mental practice with the right and left side (first: 1 × right, 1 × left; second: 1 × right, 3 × left, 1 × right, 5 × left; level 5.II–5.IV); (iii) final memory consolidation through mental practice of an everyday life task (1 × right, 3 × left; level 5.V). While the first and second step always referred to the standardized flexion-extension and adduction-abduction movement, only the third step included an individually customized mental task. Most participants chose knocking on a door, stirring in a pot or the sign “come here”. This customized task has a closer resemblance to their real life and should elevate their motivation. Subjects were instructed to use an internal kinesthetic type of imagery from the first-person perspective. We ensured that the participants used them by letting them fix the movement description in written form and by subsequently asking regularly about details during realization of the training. Daily repeated rehearsal of the level 5 steps I–V over the period of 3 weeks stabilized the memory trace after initial acquisition of the movement. Overall, 90% of the runs were executed mentally and 10% physically. Detailed guidelines for practitioners are set out in our article “Mental practice in orthopedic rehabilitation: where, what, and how?”(39).
Participants in the control group performed no training during the intervention period.
Statistical analysis
For statistical analysis, SPSS 11.5 was used. Differences in baseline assessment were tested by a t-test. A 2-way analysis of variance (ANOVA) for repeated measures was performed to establish the effect of imagery training on clinical parameters (range of motion of the radiocarpal joint). The values established at post-assessment contrasted with the mean values of the corresponding variables during baseline (conventional limits: p ≤ 0.05, p ≤ 0.01, p ≤ 0.001; 2-sided test). The effect size of mental practice treatment on all parameters was evaluated by using a partial η2 derived from the statistical analysis.
Due to the lack of prior research studies a priori power analysis was not conducted.
RESULTS
None of the participants dropped out of the study. The results are based on all 18 persons admitted.
The participants attended all prescribed supervised training sessions. The analysis of the variable duration of the individual training documented in the training-diary proved the same amount of practice for every participant (Mper day = 15.9; SD 0.852; min = 14; max = 17). The training program was feasible and safe.
Demographic variables of the participants subdivided per intervention group (nmental practice group = 9; ncontrol group = 9) were similar.
Comparison of the baseline assessment of the mental practice group with the control group revealed no significant differences for any of the measured variables (Table I). The mean scores of both groups showed similar baseline values for dorsal extension, palmar flexion, radial and ulnar abduction of the wrist joint. The t-test revealed no statistically significant differences between groups at the pre-intervention measurement.
|
Table I. Baseline assessment |
|||
|
Range of motion of the wrist joint |
Control group (n = 9) Mean (SD) |
Mental practice group (n = 9) Mean (SD) |
p-value |
|
Dorsal extension, º |
73.89 (8.28) |
75.00 (7.07) |
0.78 |
|
Palmar flexion, º |
64.67 (10.59) |
68.33 (12.50) |
0.51 |
|
Radial abduction, º |
28.89 (14.74) |
28.89 (18.33) |
1.00 |
|
Ulnar abduction, º |
40.78 (9.18) |
38.89 (10.24) |
0.69 |
|
SD: standard deviation. |
|||
The expected effects of the immobilization were demonstrated in all participants (Table II). Range of motion of the radiocarpal joint deteriorated in both groups at the follow-up measurement in all 4 dependent variables. However, differences between both measurements were clearly smaller in the mental practice group (1.33; 4.89; 3.44; 3.00) compared with the control group (8.78; 10.11; 7.11; 12.00). These differences could be an indication of the effects of mental practice intervention.
|
Table II. Means and standard deviations (SDs) of control group and mental practice group regarding clinical parameters before (pre-) and after (post-) intervention: results of the analysis of variance for repeated measures |
|||||||||||
|
Range of motion of the wrist joint |
Control group (n = 9) Mean (SD) |
Mental practice group (n = 9) Mean (SD) |
Time |
Interaction |
|||||||
|
Pre- |
Post- |
Pre- |
Post- |
F (1,16) |
η2p |
|
F (1,16) |
η2p |
|||
|
Dorsal extension, º |
73.89 (8.28) |
65.11 (11.26) |
75.00 (7.07) |
73.67 (7.23) |
61.569*** |
0.794 |
33.375*** |
0.676 |
|||
|
Palmar flexion, º |
64.67 (10.59) |
54.56 (13.15) |
68.33 (12.50) |
63.44 (12.42) |
26.308*** |
0.622 |
3.189 |
0.166 |
|||
|
Radial abduction, º |
28.89 (14.74) |
21.78 (13.41) |
28.89 (18.33) |
25.44 (12.12) |
5.159*** |
0.244 |
0.622 |
0.037 |
|||
|
Ulnar abduction, º |
40.78 (9.18) |
28.78 (11.57) |
38.89 (10.24) |
35.89 (8.70) |
21.600*** |
0.574 |
7.776* |
0.327 |
|||
|
*p ≤ 0.05, **p ≤ 0.01, ***p ≤ 0.001. |
|||||||||||
The effect sizes differed between the different movements. The mental practice group demonstrated significantly less limitation of range of motion of the radiocarpal joint concerning the variables dorsal extension and ulnar abduction: factor time (F1,16 = 61.569, p = 0.000, η2 = 0.794), respectively (F1,16 = 21.600, p = 0.000, η2 = 0.574) and interaction time × group (F1,16 = 33.375, p = 0.000, η2 = 0.676), respectively (F1,16 = 7.776, p = 0.013, η2 = 0.327).
With respect to the palmar flexion and radial abduction, a significant difference between the imagery group and the control group was found only in the factor time (F1,16 = 26.308, p = 0.000, η2 = 0.622) and (F1,16 = 5.159, p = 0.037, η2 = 0.244), respectively, but no significant differences for the interaction time × group (F1,16 = 3.189, p = 0.093, η2 = 0.166) and (F1,1 = 0.622, p = 0.442, η2 = 0.037), respectively.
DISCUSSION
The main outcome of the present pilot study is that healthy participants in the mental practice group showed a significant treatment effect in the range of motion of the radiocarpal joint concerning the variables dorsal extension and ulnar abduction. The members of the control group had greater losses in dorsal extension and ulnar abduction compared with the members of the mental practice group after removal of the immobilizing cast. The intervention was feasible and safe and reported adherence was good. The intervention should therefore be considered for a safety and feasibility trial for mental practice in forearm fractures leading to immobilization.
The intervention did not reach all anticipated goals. We could not demonstrate a significant difference in palmar flexion and radial abduction; we could only detect an improvement by trend. By performing the wrist-movements the ability of moving the hand to the top (dorsal extension) is greater than to the bottom (palmar flexion) and turning the hand to the outside (ulnar abduction) is easier than to the inside (radial abduction). Data on the descriptive statistics level (Table II) was consistent with this observation: for the variables palmar flexion and radial abduction variation is smaller. It is therefore more difficult to obtain significant results here. Whether this was due to a lack of power, or whether it was related to the exercise regime, remains undetermined. The sample size was too small to examine the effects on ADL function or other International Classification of Functioning, Disability and Health-relevant domains, which would be the primary end-points in a rehabilitation trial.
Taken together, our data indicates that range of motion of the radiocarpal joint can be positively influenced by mental practice. The optimization of range of motion in the mental practice group may be ascribed to the regular use of the neuronal structures, in the strict sense of the internal representation of the hand movement sequence. Immobilization in the control group could be explained, on a neuronal level, by the loss of movement representations, which may have led to temporary loss of memory of the hand function (40) and inefficient central control of movement. In the mental practice group, in contrast, differentiation and stabilization of the internal representation of the movement took place. This may have inhibited the usual process of loss of mobility through immobilization. Hence, continuous input from a limb in the form of repeated mental performance of a movement appears to be a central requirement for preventing the impairment of central reorganization and central control.
Because mental practice simulates movement, it is not surprising that the neural motor network is activated while imaging motor actions. The prefrontal cortex and its connections to the basal ganglia play a crucial role in mental practice for maintaining dynamic motor representations in working memory (16). After flexor tendon repair and immobilization, patients showed activity of the basal ganglia measured by positron emission tomography while they executed finger flexion movements (40). However, activity in the basal ganglia fails directly after the immobilization period. Continuing stimulation of the basal ganglia by mental practice should prevent the neuronal decline from occurring during immobilization (32), which may also have a clinically important effect on motion.
Effects of mental practice on range of motion have also been shown for the lower extremity. The use of the mental practice of stretching by competitive athletes (synchronized swimmers) enhanced ankle, hamstrings and adductor flexibility (28). In another study with healthy adults, mental practice coupled with proprioceptive neuromuscular facilitation resulted in a better range of motion of the hip joint than was achieved by physical training alone (29). A randomized controlled trial of our group (24) with 26 patients after knee endoprosthesis demonstrated a significant improvement of knee flexion after a 6-week mental training program supplemented with mirror visual feedback (41). The incorporation of a mental practice-only condition into the experimental design still needs to confirm a positive effect attributable to mental practice alone.
The effects of mental practice on range of motion of the upper extremity have been reported only in a randomized controlled trials during immobilization after flexor tendon repair (32). A 6-week mental practice program showed no significant influence on the active motion of the fingers. This might have been due to differences in the assessment procedure and the content of the intervention. The intervention focused on the flexion of the fingers and the wrist. The measurement criteria were the mean total motion per hand, calculated by the range of motion of all joints of 4 fingers. Moseley & Barnett (42) propose not using single and simple motor tasks, but using a variety of mental movements including activities of daily living (ADL).
This study has several limitations that must be considered. As mentioned above, the participants were living in social circumstances that allowed the wearing of a forearm cast for 3 weeks. Although this recruitment could have led to bias in the results, it was indispensable for practical reasons. The possibility that participants contracted their muscles during mental practice sessions, or even consciously as isometric contractions, cannot be fully eliminated. An exclusion of this confounder would have only been possible by permanently applying an electromyogram measurement. From a methodological point of view this would not have been possible. The fact that the casts applied to both groups were identical is likely to limit the impact of any non-specific muscular contraction that occurred during immobilization.
Another potential confounder is a contralateral effect. Participants in the mental practice condition were initially asked to execute the movement with the right, non-immobilized wrist (contralateral physical training; Fig. 2). Subsequently, they systematically rehearsed alternating contralateral physical training (with open and closed eyes) with mental practice of the left hand. Ninety percent of the runs were executed mentally and 10% physically. This procedure aimed to support the development of movement imagination, to stabilize a memory trace after the initial acquisition of the wrist movement, and to differentiate the motor representations. Data in the rehabilitation context report better adherence and learning effects by combining physical execution and mental rehearsal trials (34).
Furthermore, it was difficult to control the participants’ compliance in the mental practice group. This may have led to an underestimation of the effects of mental practice. We tried to overcome this problem by asking each participant to keep a paper training-diary of the regularity of this independent training.
As we used a quasi-experimental design with an “untreated” control group in this pilot study, we cannot attribute the positive effects found only to the imagination trials of the training program. As Mental Gait Training also integrates phases of movements with the non-affected limb, this might have influenced the results obtained. Therefore, further studies should use an active control group, sharing more commonalities with the mental practice group.
The observations reported above make it impossible to draw the firm conclusion that mental practice reduces the loss of range of motion of the radiocarpal joint resulting from forearm immobilization. Despite the limitations of the present study we consider the use of mental practice to optimize the range of motion after immobilization useful.
Compared with the empirical evidence for orthopedic rehabilitation, the additional benefit of mental practice as an adjunct to physiotherapy or occupational therapy in neurological rehabilitation has been discussed and evaluated for a multitude of diseases (22, 24). Current evidence provided by systematic review articles (21) reporting medium to high effect sizes (EScorrected) 0.5–0.8 (43), support the efficacy of mental practice after stroke. From the orthopedic point of view examination of this neurological domain would be valuable because of the comorbidity, particularly of elderly patients, in orthopedic rehabilitation.
To our knowledge, the present study is the first attempt to evaluate the effect of mental practice on range of motion after immobilization of the wrist of healthy persons. This paradigm was developed to imitate a distal radial fracture. Wrist fractures in older persons have a high occurrence rate coupled with a poor outcome. Therefore, further research into the role of mental practice in this patient group is justified.
Aging of the population will lead to increasing numbers of osteoporotic fractures of the wrist, but also of the humerus, spine, or hip (44, 45). Einsiedel et al. (46) demonstrated that the risk of losing autonomy after a wrist fracture is almost as high as after a hip fracture. This is partly related to upper extremity dysfunctions with ADL, such as dressing, washing and eating. Post-fracture strategies tackling the problems mentioned above, might reduce negative outcomes and considerably improve quality of life. So far, no proactive strategies have been published that counteract the immobilization problems. Because of the expected clinical benefits of preventing the loss of hand function that occurs during immobilization, and because of the low cost and simple application of the intervention, further research into the effects of mental practice in orthopedic rehabilitation is warranted.
Conclusions about the efficacy and the effectiveness of mental practice during immobilization should be drawn integrating 3 different levels of measures. First, one should focus on the motor performance. The assessment of hand function should include functional measures and kinematic analysis of movements. Imaging and quantification of forearm muscles will allow a better understanding of the association of muscle atrophy and hand function. Secondly, it will be interesting to focus on psychological variables, such as age-adjusted imagery ability, capacity of working memory, and the individual representation of movements. Finally, future work should assess central processes by using functional and structural neuroimaging. Indicators of central control processes, such as preparation time of movements, might be useful in interpreting the findings.
Given the incidence of wrist fractures and the demographic megatrends it is important to determine whether mental practice is effective in improving outcome in older persons. This aspect should be studied first in pilot studies with the main target groups and then in controlled, follow-up designed trials with adequate power.
AcknowledgEmentS
The authors would like to thank Mrs Bettina Gündel (BG) and Mrs Jessica Tenten (JT) for their valuable support as physiotherapists and Mr Theodore Temple (TT) for English-language proofreading.
REFERENCES
