Charles Sèbiyo Batcho, PhD1, Gaëtan Stoquart, MD, PhD1, 2 and Jean-Louis Thonnard, PhD1,2
From the 1Institute of Neuroscience and 2Department of Physical Medicine and Rehabilitation, Cliniques Universitaires St-Luc, Université catholique de Louvain, Brussels, Belgium
OBJECTIVE: To determine whether regular brisk walking can promote functional recovery in community-dwelling stroke patients.
Patients: A total of 44 chronic stroke patients, recruited in Belgium and Benin, respectively European high-income and African low-income countries.
METHODS: This longitudinal, single-cohort, observational study with 1 intervention period and 4 time-points of assessments (2 baseline, 1 post-intervention and 1 follow-up) was structured in 3 periods: pre-intervention period (1 month), intervention period (3 months) and follow-up period (3 month). Intervention consisted of a 3 times/week group-based brisk walking programme. Primary outcome measures were ACTIVLIM-Stroke questionnaire and the 6-minute walk test (6MWT). Secondary outcome measures were the Stroke Impairment Assessment Set (SIAS), the Hospital Anxiety and Depression Scale (HADS), and the Berg Balance Scale (BBS).
RESULTS: All outcome measures were stable during the pre-intervention period (p ≥ 0.16). They all improved significantly after intervention (p ≤ 0.01), except the HADS (p = 0.058). However, during the follow-up period, SIAS (p = 0.002) and BBS (p = 0.001) decreased, while ACTIVLIM-Stroke, 6MWT and HADS showed no significant change (p ≥ 0.13).
CONCLUSION: This study suggests regular brisk walking as an effective approach to promote functional recovery in chronic stroke survivors. However, further studies are required before generalizing these results to the whole stroke population.
Key words: rehabilitation; chronic stroke; functional recovery; exercise; walking.
J Rehabil Med 2013; 45: 00–00
Correspondence address: Jean-Louis Thonnard, Institute of Neuroscience, Université catholique de Louvain, 53 Avenue Mounier, COSY-B1.53.04, BE-1200 Brussels, Belgium. E-mail: jean-louis.thonnard@uclouvain.be
Accepted Apr 24, 2013; Epub ahead of print Aug 8, 2013
Introduction
Stroke remains a leading cause of long-term disability (1). Post-stroke consequences include persistent neurological deficits and physical deconditioning that propagates disability (2) and worsens functional independence. For example, the exercise capacity of stroke patients is approximately 40% below age- and gender-adjusted norms for sedentary individuals (2–4). Consequently, stroke survivors are prone to a sedentary lifestyle that limits performance of activities of daily living (ADL) (5), and aggravates the already significant health and economic consequences of stroke (6).
Through a meta-analysis based on 11 studies, Evans and colleagues (7) found that inpatient multidisciplinary rehabilitation improved short-term survival and functional ability after stroke. However, they did not find long-term benefits, suggesting that therapy should be extended to the home, rather than being discontinued at discharge. Unfortunately, the implementation of long-term rehabilitation is problematic. In low-income countries, limited resources mean that structured rehabilitation is not offered to stroke survivors, once discharged. In high-income countries, the economic burden of stroke has increased with the application of individually tailored face-to-face long-term treatments aimed to stimulate stroke recovery after discharge. This underlines the need of cost-effective approaches that are suitable for both high- and low-income contexts, to promote functional recovery in chronic stroke patients worldwide.
Based on the fact that several studies have shown evidence of the benefits of exercise to address common post-stroke problems (8, 9), it has been advocated that regular physical activities can improve, or at least prevent from worsening, the functional status of stroke survivors. The challenge is finding appropriate physical activities that will ensure the adherence of this particular population. Indeed, previous studies found poor adherence to exercise programmes in healthy subjects and patients (10). To enhance adherence, we sought a physical activity that fits participants’ needs and is achievable at moderate intensity with minimum investment. Group-based brisk walking appeared to be a good choice. The rationale for this choice lies within 2 points. Firstly, walking is the most commonly reported activity (11), it is part of ADL, and ability to move independently plays a large role in human functional autonomy. Particularly, reduction in walking ability is a major devastating outcome of strokes (12), and its restoration is of great importance to patients and their relatives (13). For these reasons, many studies and programmes, such as the Stroke Inpatient Rehabilitation Reinforcement of Walking Speed (SIRROWS), have applied walking as a main inpatient rehabilitation tool (12, 14–16). Secondly, social support plays an important role in exercise behaviour after stroke (17). Dobkin et al. (14), through the SIRROWS project, have found that feedback about performance once a day produced gains in walking speed large enough to permit unlimited, slow community ambulation at discharge from inpatient rehabilitation. It can be hypothesized that a group-based training modality that allowed participants to benefit from the social support of other persons also experiencing a stroke condition may be efficient. Consequently, this study then tested the efficacy of group-based regular brisk walking on functional recovery in chronic stroke patients.
Participants and Methods
Study design
This study was approved by the ethics committee of the Université catholique de Louvain in Belgium and the local ethics committees of the participating caregiver centres and hospitals in Benin. Signed informed consent form was obtained from each participant. It is a 7-month longitudinal, single cohort, observational study with 4 time-points of assessments and 1 intervention period. The intervention period was preceded by a 1-month pre-intervention period and followed by a 3-month follow-up period. During the pre-intervention and the follow-up periods, participants received no particular intervention and were instructed to maintain their lifestyle, with the aim of determining their stability before intervention and to examine the long-term effects of the intervention.
Clinical Trial Registration: http://www.clinicaltrials.gov. NCT01570920.
Inclusion criteria and recruitment of participants
Inclusion criteria were: (i) having a stroke at least 6 months before inclusion; (ii) having at least minimal ambulatory capacity with supervision and/or an assistive device; (iii) having no major cognitive deficit that could prevent completion of a self-report questionnaire (score ≥ 24 of 30 on the Mini-Mental State Examination (18)). The ambulatory capacity was examined with a 10-m test that measures gait velocity (m/s) at usual individual’s pace. In this study, the score was the mean value of 3 trials each participant performed on a linear 14-m pathway, including 2-m warm-up, 10-m for time recording and 2-m cool-down.
Participants were recruited in Belgium and in Benin. They were identified from patient registers at recruitment centres. A total of 76 individuals with stroke were screened for eligibility in both countries; 32 did not participate for reasons that included not meeting inclusion criteria, being enrolled in other studies, or refusing to participate. Starting the programme were 44 patients (18 from Belgium and 26 from Benin), but 8 did not regularly participated and 2 dropped out before the fourth assessment. Finally, 34 subjects (15 from Belgium and 19 from Benin) fully completed the 7-month programme from enrolment to fourth assessment. Fig. 1 describes the recruitment process.
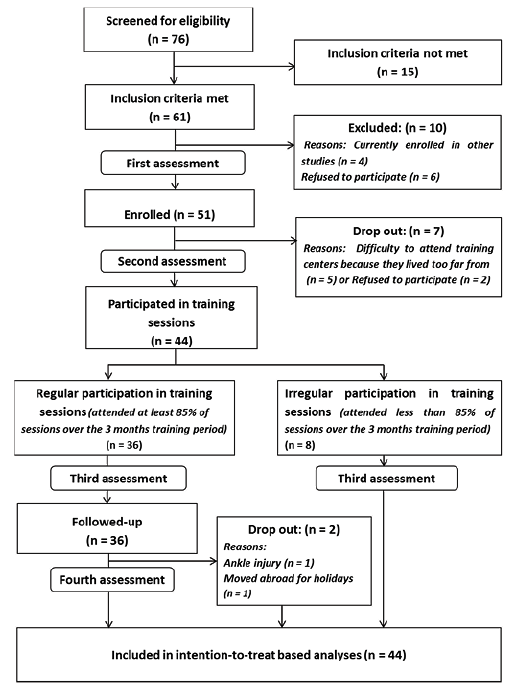
Fig. 1. Recruitment of participants.
Setting and materials
Walking training sessions were performed at sport centre facilities, providing safe and comfortable areas for group-based exercise. Participants were equipped with a heart rate monitor and pedometer during each training session to record heart rates and walking speeds. Recording participants’ performance from session to session helped to provide regular feedback and enabled the training to be individually adjusted.
Intervention
Intervention consisted of a 3 times/week group-based brisk walking programme of 3 months. Brisk walking was defined as a pace faster than normal that leaves the individual slightly breathless but still able to converse (19). Participants were instructed to walk at their fastest pace on a regular surface. Based on effort adaptation, they either walked continuously throughout the session or had intermittent walking bouts interspersed with rest periods. Training sessions were supervised by physical therapists who regularly encouraged participants to maintain a safe walking speed that was as fast as possible and gave personalized feedback after each session (mean heart rate, walked distance, etc.).
Assessment procedure and outcome measures
Each participant completed 2 baseline assessments before intervention (time 1 and time 2), 1 post-intervention assessment (time 3) and 1 follow-up assessment (time 4). These 4 assessment time-points defined 3 periods: pre-intervention period (1 month), intervention period (3 months) and follow-up period (3 month). However, the fourth assessment was not realized in irregular participants and those who dropped out during the follow-up period. Assessments included selected variables of impairment, walking performance and activity limitations. Socio-demographic data were collected during the first assessment. Primary outcome measures were the ACTIVLIM-Stroke (20) and the 6-min walk test (6MWT) (21). ACTIVLIM-Stroke is a cross-cultural Rasch-built scale that measures ability to perform activity of daily living in patients with stroke. This 20-item scale covers a large range of aspects of activity limitations, such as self-care, transfer, mobility, balance and manual ability. The 6MWT (21) is a performance test that evaluates walking endurance; it consists in recording the distance (m) a subject walked in 6 min. In our study, participants were asked to perform the 6MWT by walking as quickly as possible on flat ground a square path with a 52-m perimeter marked on the floor. They were allowed to stop and rest as necessary. Patients were informed of the time after 2, 4 and 5 min, and distance was recorded to the nearest metre. Secondary outcome measures were the Stroke Impairment Assessment Set (SIAS) (22), which evaluates the stroke-related impairment, the Berg Balance Scale (BBS) (23), which assesses balance, and the Hospital Anxiety and Depression Scale (HADS) (24), a self-reported questionnaire that assesses anxiety and depression.
Analysis
Demographics and participant performance were characterized using descriptive statistics. A one-way repeated measures analysis of variance (RM-ANOVA) was used to compare scores of ACTIVLIM-Stroke and 6MWT at times 1, 2, 3 and 4. Post-hoc analyses with Bonferroni adjustment were performed to investigate the specific evolution of scores during pre-intervention, intervention and follow-up periods. For SIAS, BBS, and HADS non-parametric statistics (Friedman test) were used to investigate overall effects. In addition, period-specific effects were analysed with pairwise comparison based on a Wilcoxon signed-rank test. For these ordinal variables, the p-values were not Bonferroni adjusted and the statistical significance have been judged according to an adjusted alpha level of 0.017 obtained as the initial alpha significance level of 0.05 divided by 3, the number of planned comparisons (25). All analyses were performed on an intention-to-treat basis, including participants who underwent at least the 3 first assessments. Any missing values at fourth assessment were imputed using the last observed values (26). Finally, the relationships between variables were analysed using Spearman’s correlation. Data were analysed with SPSS and Sigma Stat.
Results
Study participants
Belgian and Beninese participants were similar at enrolment. Indeed, comparison of baseline scores between both subsamples showed no significant differences, except for age (p = 0.001) and spontaneous gait speed (p = 0.008). The mean age and standard deviation (SD) was 64.8 years (SD 11.8) old in Belgian sub-sample vs 52.6 (SD 6.7) in Benin sub-sample. The spontaneous gait speed as measured by the 10 m walk test (10 MWT) was 0.92 m/s (0.45) in Belgian participants and 0.57 m/s (0.28) in Benin participants. Table I presents the socio-demographic characteristics of the 44 participants who started the training programme after the second assessment.
|
Table I. Sample description |
|||
|
Characteristics |
Having completed the programme (n = 34) |
Dropped out (n = 10) |
|
|
Age, years, mean (SD) |
57.97 (11.02) |
53.25 (10.53) |
|
|
Stroke delay, months, mean (SD) |
37.7 (31.7) |
26.25 (10.69) |
|
|
Weight, kg, mean (SD) |
72.6 (11.5) |
63.19 (14.99) |
|
|
Height, cm, mean (SD) |
168 (7) |
164 (9) |
|
|
Body mass index, mean (SD) |
25.6 (3.8) |
23.42 (4.72) |
|
|
10 MWT, m/s, mean (SD) |
0.73 (0.41) |
0.49 (0.34) |
|
|
Sex, n (%) |
|||
|
Female |
10 (29) |
4 (40) |
|
|
Male |
24 (71) |
6 (60) |
|
|
Social status, n (%) |
|||
|
Live alone |
4 (11.8) |
0 – |
|
|
Live in community (couple, family, others…) |
30 (88.2) |
10 (100) |
|
|
Paretic side, n (%) |
|||
|
Left |
18 (53) |
7 (70) |
|
|
Right |
16 (47) |
3 (30) |
|
|
Type of stroke, n (%) |
|||
|
Ischaemic |
20 (61,7) |
7 (70) |
|
|
Haemorrhagic |
5 (14.7) |
1 (10) |
|
|
Unknown |
8 (23.6) |
2 (20) |
|
|
Professional status, n (%) |
|||
|
Non-retired |
23 (67.6) |
6 (60) |
|
|
Retired |
11 (32.4) |
4 (40) |
|
|
10 MWT: 10-m walk test; SD: standard deviation. |
|||
Intervention effects
Table II shows means and standard deviations (SD) or medians and range of scores for each variable. Except the HADS (p = 0.058), there were significant overall effects on SIAS (p < 0.001), BBS (p < 0.001), 6MWT (p < 0.001) and ACTIVLIM-Stroke (p < 0.001). For these variables, specific effects during each of the 3 periods were examined using post-hoc analyses. Firstly, during the pre-intervention period (time 2 vs time 1), no significant change was observed (Table II), indicating that participants were stable before intervention. Secondly, between times 2 and 3 (intervention period), SIAS median score significantly changed from 54 to 64 (p < 0.001), BBS from 46 to 50 (p = 0.001). The mean 6MWT increased by 34.5 m (p < 0.001), a significant positive change of 13.4% compared with before training. Mean ADL performance significantly increased by 25.1% (p = 0.008) during training, a mean change of 0.44 logits. Activities such as getting out of a car, taking a shower, putting on socks, walking upstairs, sweeping or vacuuming, or walking more than 1 km were rated as easy or difficult, while before training they were impossible or difficult. We conducted a second analysis using an individual-based approach (27, 28). This approach accounts for standard errors (SEs) of the measurement associated with each patient’s measures and mean change between time 2 and time 3 evaluations. Based on Cohen’ guidelines (29), this analysis showed that 61.4% (n = 27) of patients reported a positive change, while 22.7% (n = 10) reported a negative change and 15.9% (n = 7) remained unchanged. Finally, most scores stabilized or decreased during the follow-up period (Table II). However, SIAS (p = 0.002) and BBS (p = 0.001) decreased slightly, but significantly. ACTIVLIM-Stroke (p = 0.97), 6MWT (p = 0.13) and HADS (p = 0.14) showed no significant differences.
|
Table II. Overall and period-specific effects of the intervention on impairment, walking performance and activity level over time |
||||||||
|
Scores over time, mean |
Overall effect (RM-ANOVA or Friedman test) |
Period-specific effects |
||||||
|
Baseline 1 (time 1) |
Baseline 2 (time 2) |
Post-intervention (time 3) |
Follow-up (time 4) |
Pre-intervention period (T1 vs T2) |
Intervention period (T2 vs T3) |
Follow-up period (T3 vs T4) |
||
|
ACTIVLIM-Stroke, logit, mean (SD) |
1.67 (1.10) |
1.75 (1.23) |
2.20 (1.22) |
2.15 (1.30) |
< 0.001 |
0.93 |
0.008 |
0.97 |
|
6MWT, m, mean (SD) |
243.6 (120.6) |
256.8 (123.5) |
291.3 (134.8) |
272.3 (130.1) |
< 0.001 |
0.42 |
< 0.001 |
0.13 |
|
SIAS, median [range] |
56.5 [37–74] |
54 [37–73] |
64 [41–75] |
61 [38–75] |
< 0.001 |
0.18 |
< 0.001 |
0.002 |
|
BBS, median [range] |
46 [25–56] |
46 [27–56] |
50 [35–56] |
49 [34–56] |
< 0.001 |
0.16 |
0.001 |
0.001 |
|
HADS, median [range] |
12 [1–35] |
12 [1–34] |
11.5 [0–21] |
9 [1–21] |
0.058 |
0.2 |
0.38 |
0.14 |
|
T1: time 1; T2: time 2; T3: time 3; T4: time 4; RM-ANOVA: one-way repeated measures analysis of variance; HADS: Hospital Anxiety and Depression Scale; BBS: Berg Balance Scale; SIAS: Stroke Impairment Assessment Set; 6MWT: 6-minute walk test; ACTIVLIM-Stroke: ACTIVLIM-Stroke questionnaire. |
||||||||
Relationship between activity level, walking performance and impairment
ACTIVLIM-Stroke correlated highly with BBS (ρ = 0.60, p = 0.01), and moderately with 6MWT (ρ = 0.53, p = 0.01) and SIAS (ρ = 0.37, p = 0.01). Correlation between ACTIVLIM-Stroke, anxiety, and depression was non-significant. This suggested that balance, walking endurance, and impairment might positively impact activity level in patients with stroke. The higher the balance and walking endurance, the higher the ADL performance. Based on the fact that BBS ≤ 42/56 had been indicated as the single best predictor of falls (30), we conducted additional analysis (Fig. 2) and the result confirmed the important role of balance in ADL performance. Indeed, patients who had a risk of fall (BBS score ≤ 42; n = 13) at baseline reported a lower ADL performance at each assessment compared with those with a BBS score above 42 (n = 21).
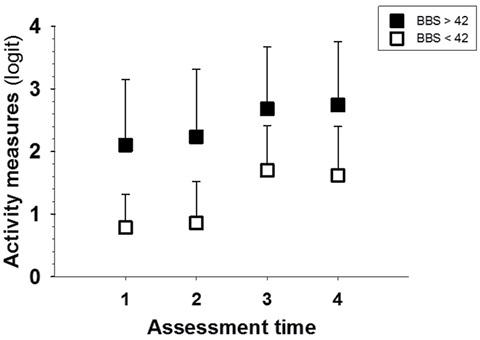
Fig. 2. Influence of balance on activity level. Participants were divided into 2 subsamples based on baseline balance level as evaluated by the Berg Balance Scale (BBS). Participants with fall risk (BBS score ≤ 42; white box; n = 13) at enrolment reported a lower activity level at each assessment than those with no risk of fall (BBS > 42; black box; n = 21). This difference was significant (p < 0.009).
Discussion
The aim of this study was to determine whether regular brisk walking improves functional recovery in chronic stroke patients. We also aimed to determine its impact on walking endurance and selected impairment variables. Our results support the hypothesis that regular brisk walking efficiently improves impairment, balance, walking endurance, and facilitates performance of ADL. Participants showed significant improvement during the intervention period, whereas they were stable during the pre-intervention period; and showed no significant positive change during the follow-up period.
These results highlighted the efficacy of group-based brisk walking in chronic stroke survivors. The absence of significant positive change during the pre-intervention and the follow-up periods indicated that interventions are necessary to stimulate functional recovery in chronic stroke patients. The 3-month walking programme helped participants to positively increase their scores in several variables of body function and activity domains. Positive change in performance of ADL is meaningful for stroke survivors’ daily life. The ACTIVLIM-Stroke provides measures with standard errors, providing an opportunity for investigation of individual changes. In clinical trials, this is essential, since change that is meaningful for groups might not be meaningful for individuals (27). This individual-based analysis indicated that most of participants (61.4%) had reported an increase in ability to perform ADL. Examining the characteristics of those who reported no positive change, it emerged that, similarly, their walking endurance as measured by the 6MWT did not significantly improve. Moreover, this group encompassed almost all non-compliant participants. This underlined the absolute necessity of regular practice of physical activities to benefit.
Our results showed that increasing walking endurance was important to reduce activity limitations, confirming previous observations (31, 32). In fact, walking ability has been identified as reflecting health and functional status in community-dwelling older people (32). Our results also confirm that balance is essential in functional recovery after stroke. Patients who had a risk of fall reported a lower activity level than those who were free of fall risk, confirming balance as an important predictor of physical activity (33). Previous studies observed that changes in balance ability correlate strongly with changes in functions, such as ambulation and ADL (34, 35). These observations highlight the importance of balance rehabilitation in stroke patients.
According to Alzahrani et al. (33), mood is associated with free-living physical activity of stroke survivors residing in the community. However, in our study, the intervention that was delivered on a group-based modality had no significant effect on mental well-being. Patients did not report a lower level of anxiety and depression after the intervention. This result did not confirm our expectation that group-based training could improve mood in chronic stroke survivors. Nevertheless, group-based programmes have a substantial economic advantage. Indeed, during the course of this programme, both in Belgium and in Benin, we provided 211 training sessions, with a mean of 5 participants (range 3–9) per session. The total required sessions in individually tailored face-to-face therapy would have exceeded 1,054, 5 times the number of sessions needed for the group-based approach. Accordingly, the total cost of professional supervision in individual-based training would have been approximately 5 times higher than for group-based training. Because stroke rehabilitation requires long-term intervention, it is essential to consider the economic cost of its management. This idea matches the increasing recommendations to promote cost-effective approaches in the long-term management of stroke (36).
Five participants were not included in the study because they lived far from the training centre, and their participation in this group-based intervention would require several hours of travelling from home to training centre. This study underlines not only the interest of regular physical activity in chronic stroke survivors, but also the challenge of designing strategies that facilitate participation in physical activity for this particular population. These strategies should consider several factors, such as the type of physical activity and the modality of implementation.
Finally, this observational study had cross-cultural aspects, occurring in Benin, a low-income sub-Saharan African country, and Belgium, a high-income European country. We observed similar effects of the brisk walking programme on both samples. The pattern of variable evolution was identical between the samples. However, during follow-up period, the 6MWT decreased by a mean of 22.3 m (p < 0.02) in the Benin sample, while the mean decrease in the Belgian sub-sample was only 12.8 m (p = 0.11). Regarding other variables, no significant difference was observed between Belgian and Benin samples. The fact that scores stabilized or decreased during follow-up period suggests that training should be continuously available for chronic stroke survivors to make sure that participants maintain an active lifestyle to improve or preserve functional recovery.
However, the methodological aspect of this study disserves discussion. It is known that clinical studies investigating the effect of a particular intervention, are stronger with a separate control group on the basis of a blinded and randomized controlled design, assuming the homogeneity of both experimental and control groups. However, given the heterogeneity of stroke survivors’ profile, it is very difficult and challenging to build experimental and control groups of post-stroke patients that are socio-demographically and clinically homogenous. Therefore, in our clinical observational study, each participant was his own control before and after the intervention. This design allowed us to isolate the impact of the intervention by controlling what happened before (pre-intervention period) and after (follow-up period). In addition, the power test calculation of our analyses confirmed the appropriateness of the present sample size. Indeed, power tests ranged from 0.78 to 0.99, at an alpha level of 0.05. It is a common knowledge that an adjustment of alpha level to a more critical cut-off, such as 0.01, enhances the level of evidence by decreasing the risk of error from the traditional 5% to 1%. In our study, the sample size calculation with alpha set at 0.01 did not require more participants, and this was not surprising given the size of observed mean differences and standard deviations, which were largely statistically significant (p-value < 0.01). Nonetheless, since this study addresses selected chronic stroke patients, the generalization of our findings to the stroke population as a whole requires further investigation.
CONCLUSION
The results of this study show that regular ground walking is a low-cost intervention that has the potential to stimulate functional recovery in chronic stroke survivors. It contributes to increasing evidence that exercise after stroke has a number of benefits, and that chronic stroke survivors may be encouraged to be active. However, further large prospective studies are required to confirm the effects of brisk walking as an effective approach to facilitate functional recovery in the chronic stroke population as a whole.
The author declare no conflicts of interest
References
