Maud Stenberg, MD1, Lars-Owe Koskinen, MD, PhD2, Richard Levi, MD, PhD1 and
Britt-Marie Stålnacke, MD, PhD1
From the 1Department of Community Medicine and Rehabilitation, Rehabilitation Medicine and 2Department of
Pharmacology and Clinical Neuroscience, Division of Neurosurgery, Umeå University, Umeå, Sweden
OBJECTIVE: To assess: (i) the clinical characteristics and injury descriptors of patients with severe traumatic brain injury in Northern Sweden admitted to the single Neurotrauma Center (NC) serving this region; (ii) the care pathway of patients from injury to 3 months after discharge from the NC; and (iii) the outcomes at 3 months post-injury.
DESIGN: Population-based prospective 2-year cohort study.
Patients: Patients age 17–65 years with acute severe traumatic brain injury, lowest non-sedated Glasgow Coma Scale (GCS) score of 3–8 within 24 h post-trauma.
METHODS: Patients were treated according to an intracranial pressure-oriented protocol based on the Lund concept at the NC. They were assessed at 3 weeks after injury with Rancho Los Amigos Cognitive Scale Revised (RLAS-R), Levels of Cognitive functioning, and at 3 months with RLAS-R and Glasgow Outcome Scale Extended (GOSE).
RESULTS: A total of 37 patients were included. Hospital deaths within 3 months post-injury occurred in 5 patients. After 3 months the RLAS-R scores were significantly improved (p < 0.001). Eight patients had both “superior cognitive functioning” on the RLAS-R and “favourable outcome” on the GOSE. Thirty-four patients (92%) were directly admitted to the NC. By contrast, after discharge patients were transferred back to one of several county hospitals or to one of several local hospitals, and some had multiple transfers between different hospitals and departments.
CONCLUSION: Overall outcomes were surprisingly good in this group of severely injured patients. The routines for transferring patients with severe traumatic brain injury from a geographically large, sparsely populated region to a regional NC to receive well-monitored neurosurgical care seem to work very well. The post-acute clinical pathways are less clearly reflecting an optimized medical and rehabilitative strategy.
Key words: traumatic brain injury; outcome; demographics; critical pathways.
J Rehabil Med 2013; 45: 770–778
Correspondence address: Maud Stenberg, Department of Community Medicine and Rehabilitation, Rehabilitation Medicine, Bldg 9A, Umeå University Hospital, Umeå University, SE-901 85 Umeå, Sweden. E-mail: maud.stenberg@vll.se
Accepted April 25, 2013
INTRODUCTION
Traumatic brain injury (TBI) constitutes a major health problem globally, and is a leading cause of death and long-term disability (1). TBI is common in young adults and has a male predominance due, primarily, to high-risk behaviours most prevalent in this demographic subset of the general population (2). The specific epidemiology of TBI makes it particularly important both to prevent and optimally treat the condition, as the stakes from a lifetime perspective are unusually high. The most widely used severity classification of TBI is based on level of consciousness (LOC) at admission, e.g. as reflected in the Glasgow Coma Scale (GCS) (3). The annual incidence of all (i.e. mild to severe) instances of TBI in Sweden is estimated at 250–350/100,000 (2, 4). The subset of severe TBI (sTBI), as defined by GCS 3–8, is, however, much rarer, with incidence estimates of 4–8/100,000/year (2). However, despite its relative rarity, sTBI constitutes a major health problem, due to the major and often permanent functional impact of the injuries, and the individual suffering of patients and their families, as well as the very high societal costs. Today, in Sweden state-of-the-art medical treatment of patients with sTBI is typically conducted at specifically designated Neurotrauma Centers (NC), operating according to one or another of several proposed, standardized protocol-driven therapies, such as the Lund concept utilized in our region (5–7). This particular protocol has been evaluated in a number of outcome studies that have shown favourable results (8–10). As a benefit of improved neurosurgical care, survival rates have improved substantially; something that, at the same time, has created an increased need for rehabilitation (10). Early rehabilitation after sTBI includes assessment and treatment to improve the patient’s level of functioning (11), as well as the prevention and treatment of secondary complications. However, the available evidence has demonstrated that formalized rehabilitation interventions have beneficial effects, both early and late after sTBI. Borg et al. (12) recently reported that continued access to rehabilitation competencies from acute management after sTBI is not the standard in Sweden.
Patients with sTBI comprise a heterogeneous group with varying complexity and prognosis. It is therefore of paramount importance in each individual case to assess key clinical descriptors, pertinent circumstances related to the injury, and, perhaps most important of all, to define early prognostic indicators that could be used to triage individual need for rehabilitation and rehabilitation planning. Various scales have been developed for assessment of disability after TBI. The most commonly used outcome measure for sTBI is the Glasgow Outcome Scale (GOS) (13). This is a global and, admittedly crude, instrument that only roughly discriminates different levels of disability. A more sophisticated version of the GOS, the Glasgow Outcome Scale Extended (GOSE), has thus been developed, allowing for a more fine-tuned categorization of post-traumatic disability (14). Moreover, the so-called Rancho Los Amigos Scale Revised Levels of Cognitive Functioning (RLAS) (15) is yet another useful instrument that has been implemented in several studies to assess recovery after sTBI, and to create a knowledge base relevant to the design of novel and appropriate rehabilitation programmes.
This study was conducted in the North Health Region (NHR) in Sweden. The study is part of a larger prospective multicentre cohort study, the ProBrain study, which focuses on adults with sTBI admitted to neurosurgical departments across the country. Most research into outcome after sTBI has focused on injury severity, and few studies have considered the effect of geographical factors (16). As the NHR comprises mainly rural districts with geographically large, scarcely populated areas, with long distances between hospitals, the clinical setting in this part of the country differs substantially from the more urbanized, southern half of the country. The NHR setting is, however, representative of many other regions globally, thus motivating separate analysis. The questions are: Is it possible to obtain good or excellent outcomes in a scarcely populated, vast area? Will the logistics of transfers of patients allow for rapid admission to a single NC serving a very large region? The focus of this article is therefore on the analysis of sTBI management referred to Umeå University Hospital, being the single NC serving the northern half of the country, and thus displaying this particular logistic challenge.
The first aim of the study was to assess clinical characteristics (age, gender, education, marital status, occupation) and injury descriptors (aetiology, previous TBI, additional injuries, intoxication) of patients with sTBI in the NHR. The second aim was to track the clinical care pathways of patients from the scene of injury and up to 3 months after discharge. The third aim was to assess outcomes at 3 months post-injury.
METHODS
The study is a prospective, total population, cohort study conducted from January 2010 to December 2011. Eligible patients were consecutively included in the study, which comprised a structured initial assessment at 3 weeks and a subsequent follow-up 3 months post-injury.
The geographical area of NHR comprises almost half of the total area of the country (136,373 km2). It is divided into 4 counties and has approximately 900,000 inhabitants, comprising only 10% of the total national population. Patients sustaining sTBI in the NHR typically will first be admitted to a county hospital or a local hospital in proximity to the venue of injury for initial assessment and stabilization prior to further transportation. In accordance with the clinical protocol during the study period, all subjects with sTBI, regardless of severity, complicating illness or concomitant injuries, were admitted to the NHR NC.
Patients
Inclusion criteria for this study were: patients aged 17–64 years; with acute sTBI with lowest non-sedated GCS score of 3–8 within 24 h post-trauma. Consecutive inclusion commenced in January 2010 and ceased after December 2011. Exclusion criterion for this study was death within 3 weeks of injury.
Treatment
Patients were treated according to an intracranial pressure (ICP)-oriented protocol based on the so-called Lund concept (5–7). This modified Lund protocol has been outlined elsewhere (8, 9). In summary, an aggressive neurosurgical approach is adopted, including prompt removal of intracranial haematomas. Patients are sedated, receive continuous analgesia, are mechanically ventilated and initially nursed in a supine position with no head elevation. Midazolam is used for sedation and fentanyl for analgesia. Patients are normo-ventilated (PaCO2 4.5–5.5 kPa) and PaO2 kept ≥ 12 kPa. Normovolaemia is maintained with preferably albumin infusion and packed red blood cells. Serum albumin (≥ 40 g/l) and haemoglobin (≥ 110 g/l) levels are maintained, and a neutral to slightly negative fluid balance is achieved by using furosemide as needed. Serum levels of sodium (≥ 135 mmol/l) and blood glucose are kept within normal limits. Infusions of metoprolol and clonidine are used as needed after establishment of normovolaemia. The rationale behind this protocol is to normalize mean arterial blood pressure (MAP), to minimize fluid leakage through the capillary membrane, and to reduce stress mediated by the sympathetic nervous system. A minimum cerebral perfusion pressure (CPP) of 50 mmHg is accepted. Additional possible interventions to reduce an elevated/rising ICP (> 20 mmHg) are: low-dose barbiturates, ventriculostomy with intermittent drainage, and/or decompressive craniectomy.
Procedure
The primary hospital performed an initial computed tomography (CT) scan of the brain. (This investigation was often repeated upon arrival to the NC). Pictures were transferred electronically to the NC, where a neuroradiologist assessed the images, for this study particularly presence or absence of epidural and subdural haematomas, traumatic subarachnoidal haemorrhage, diffuse axonal injury, brain contusion(s), and impression fracture(s). The CT scans were also classified according to the Marshall classification (17). All clinical outcome data gathering was performed by one of the authors (MS) by patient assessment at 3 weeks and 3 months post-injury. Socio-demographic data and data regarding pre-morbid health were gathered by interviews of patients and/or significant others, also performed by MS. Data regarding injury characteristics and length of stay at the NC was retrieved from the medical records.
Outcomes
Outcome variables were survival/death, Glasgow Outcome Scale Extended (GOSE) (14) and Rancho Los Amigos Cognitive Scale Revised (RLAS-R) (15). The RLAS-R was used at both 3 weeks and 3 months, whereas the GOSE was used only at 3 months.
Instruments
Glasgow Coma Scale. The GCS (13) rates loss of consciousness (LOC) by assessment of the patient’s verbal, eye opening, and motor responses on a scale of 3 to 15, with higher scores indicating better responses. In the NHR the 8-point Reaction Level Scale (RLS) (18) is widely used; the inclusion criterion was RLS 8–4. Comparison of these scales has been carried out (19). Lowest level of consciousness is presented as GCS.
Rivermead Post-traumatic Amnesia Protocol (RPAP). The RPAP comprises 5 questions. It documents the time-interval from brain injury to the return of continuous memory, including periods of unconsciousness and confusion. The interviewer asks questions so that the patient will relate their first memories after the accident in a coherent chronological order. Post-traumatic amnesia (PTA) classification is divided into 4 categories: Mild < 1 h, Moderate 1–24 h, Severe 1–7 days, and Very severe > 7 days (20).
Rancho Los Amigos Cognitive Scale Revised, Levels of Cognitive functioning. The RLAS-R Levels of Cognitive functioning (15) is a medical scale with scores from 1 to 10, representing 10 states of cognitive and behavioural functioning through which patients with TBI typically progress. Higher scores indicate improved functioning. The bottom level is “No Response, Total Assistance”, and the top level is “Purposeful, Appropriate: Modified Independent”. The RLAS-R levels were dichotomized into 2 categories: inferior functioning (RLAS I–VIII) and superior functioning (RLAS-R IX–X). The scale is used for assessment of cognitive and behavioural recovery after TBI with coma. Patients are thus assessed by reaction to stimuli, ability to follow instructions, presence of confusion, behaviour with and without meaning, cooperation, attention, ability to maintain attention to the environment, verbal ability, memory, orientation and higher cognitive ability.
Glasgow Outcome Scale Extended. The GOSE (14) extends the 5 categories of the previously developed GOS (21) to 8, thereby increasing its sensitivity. The 8 categories span from “Dead” (score 1) to “Upper Good Recovery” (score 8). Results were dichotomized into “Unfavourable outcome” (GOSE 1–6), and “Favourable outcome” (GOSE 7–8).
Statistical analysis
Data were analysed with SPSS, version 19.0 for Windows. Data are reported as means. Non-parametric tests were used as the samples were small and/or not normally distributed. Thus, the Mann-Whitney U test was used for the comparison of continuous variables, and Wilcoxon’s signed-rank test for the study of paired observation variables. A χ2 test was used for the comparison of proportions.
Ethics
This study is a part of a multicentre study that was approved by the Regional Ethics Committee of Stockholm, Sweden (number 2009/1644/31/3).
RESULTS
A total of 37 patients with acute sTBI fulfilling the inclusion criteria were identified during the study period and included in the analyses. Thorough subsequent audits within the NHR to identify possible missed patients revealed an additional 6 persons, all males, who had sustained sTBI during the study period. As they were not identified within 3 weeks post-injury (as stipulated in the study protocol), they were not included in the study.
Clinical patient descriptors
Clinical patient descriptors are presented in Tables I and II. The majority were males. Mean age at injury was 41.3 years (men 40.1, females 44.0 years). Males had less education than the females. Most patients had sustained acute traumatic subdural hematomas. The lowest unsedated GCS in the first 24 h varied widely, with GCS 3 in 9 patients (24%), GCS 4–5 in 13 patients (35%) and GCS 6–8 in 15 patients (40%). Most cases were due to falls. All injury causes, except falls on the same level and “unknown”, were classified as high-energy trauma. Thirteen patients (35.1%) had additional injuries: traumatic spinal cord injury (n = 2), vertebral fracture, pelvic fracture, long-bone fracture, rib fracture ± pneumothorax, clavicular fracture, and/or intra-abdominal injuries. Two patients were pregnant. Thirty-six out of 37 patients (97%) had a post-traumatic amnesia > 7 days, thus qualifying as “very severe brain injury” according to the Rivermead PTA protocol. Mean length of stay in NC (n = 34) was 16.9 days. Although there was a trend towards longer LOS in those with additional injuries, this difference did not reach statistical significance.
|
Table I. Demographic characteristics of patients with severe traumatic brain injury in Northern Sweden 2010–2011 with lowest (non-sedated) Glasgow Coma Scale score 8–3, age 17–65 years |
|
|
Demographic characteristics |
|
|
Patients n (%) |
37 (100.0) |
|
Gender n (%) |
|
|
Male |
26 (70.3) |
|
Female |
11 (29.7) |
|
Age, years, mean (SD) [range] |
41.3 (15.2) [17–64] |
|
Male |
40.1 (15.3) [17–64] |
|
Female |
44.0 (14.9) [22–63] |
|
Education, n (%) |
|
|
< 12 years |
23 (62.2) |
|
= 12 years |
11 (29.7) |
|
> 12 years |
3 ( 8.1) |
|
Employment and livelihood when the accident occurred, n (%) |
|
|
Working (50–100%) or student (50–100%) |
24 (64.9) |
|
Unemployed (50%) |
1 ( 2.7) |
|
Sick-leave, full or part time (25–100%) |
9 (24.3) |
|
Social care |
4 (10.8) |
|
Other |
3 (8.1) |
|
Marital status n (%) |
|
|
Single person without children |
13 (35.1) |
|
One parent with children |
2 (5.4) |
|
Married, cohabitating without children |
15 (40.5) |
|
Married, cohabitating with children |
5 (13.5) |
|
Living with other |
2 (5.4) |
|
Known drug or alcohol misuse at time of injury, n (%) |
|
|
Yes |
11 (29.7) |
|
No |
26 (70.3) |
|
Previous brain injury requiring hospitalization, n (%) |
|
|
Yes |
12 (32.4) |
|
No |
24 (64.9) |
|
Missing |
1 ( 2.7) |
|
Previous brain injury (n = 14), n (%) |
|
|
Male |
9 (24.3) |
|
Female |
5 (13.5) |
|
SD: standard deviation. |
|
|
Table II. Injury characteristics of patients with severe traumatic brain injury (sTBI) in Northern Sweden 2010–2011 with lowest (non-sedated) Glasgow Coma Scale (GCS) score 8–3, age 17–65 years |
|
|
Injury characteristics |
|
|
Causes of sTBI, n (%) |
|
|
Road traffic accident, snowmobile, ATV, as a cyclist or pedestrians hit by car |
11 (29.7) |
|
Fall > 2 m |
10 (27.0) |
|
Fall, same level or unspecified |
10 (27.0) |
|
Bicycle accident |
2 ( 5.4) |
|
Horse accident |
2 ( 5.4) |
|
Skiing accident |
1 ( 2.7) |
|
Unknown |
1 ( 2.7) |
|
Lowest unsedated GCS first 24 h, n (%) |
|
|
3 |
9 (24.3) |
|
4–5 |
13 (35.2) |
|
6–8 |
15 (40.5) |
|
Lowest unsedated GCS first 24 h, median (range) |
5 (3–8) |
|
Length of stay-intensive care, median (n = 34) (range) |
16.9 (2–54) |
|
Signs of influence of alcohol and/or drugs at time of injury, n (%) |
|
|
Yes |
18 (48.6) |
|
No |
19 (51.4) |
|
Additional injury, n (%) |
|
|
Yes |
13 (35.1) |
|
No |
24 (64.9) |
|
Post-traumatic amnesia, n (%) |
|
|
Severe 1–7 days |
1 ( 2.7) |
|
Very severe > 7 days |
36 (97.3) |
|
Marshall CT classification, n (%) |
|
|
I |
1 ( 2.7) |
|
II |
15 (40.5) |
|
III |
6 (16.2) |
|
IV |
8 (21.6) |
|
V |
0 ( 0.0) |
|
VI |
7 (18.9) |
|
Main diagnosis included patients, n (%) |
|
|
S062 Diffuse brain injury |
9 (24.3) |
|
S063 Focal brain injury |
2 ( 5.4) |
|
S064 Epidural haemorrhage |
5 (13.5) |
|
S065 Traumatic subdural haemorrhage |
17 (45.9) |
|
S066 Traumatic subarachnoidal haemorrhage |
2 ( 5.4) |
|
S068 Other specified intracranial injury |
2 ( 5.4) |
|
Total |
37 (100.0) |
|
Retrospective review, additional patients found, age 18–65 years |
|
|
Male/female, n (%) |
6/0 (100/0) |
|
Age, years, mean (range) |
49.8 (31–56) |
|
Death within 3 months, n (%) |
2 (33.3) |
|
Main diagnosis: S062 Diffuse brain injury, n (%) |
1 (16.7) |
|
Main diagnosis: S064 Epidural haemorrhage, n (%) |
1 (16.7) |
|
Main diagnosis: S065 Traumatic subdural haemorrhage, n (%) |
4 (66.7) |
|
Total |
6 (100.0) |
|
Included patient and retrospective review additional patients found 2010 and 2011, n (%) |
43 |
|
ATV: all-terrain vehicle; CT: computed tomography. |
|
The age and gender distribution of the first CT scan, causes of sTBI, worst GCS within the first 24 h, previous brain injury in need of medical contact, additional injury, and TBI with signs of influence of alcohol or drugs, are shown in Table III.
|
Table III. Comparison causes of severe traumatic brain injury (sTBI), Worst Glasgow Coma Scale (GCS) in the first 24 h, Previous Brain Injury In need of medical contact, Additional injury, Traumatic Brain Injury with signs of influence of alcohol and/or drugs with age group and gender |
||||||
|
Age |
Gender |
Total n (%) |
||||
|
≤ 25 years n (%) |
26–49 years n (%) |
≥ 50 years n (%) |
Male n (%) |
Female n (%) |
||
|
Causes of sTBI |
||||||
|
Road traffic accident, snowmobile, ATV, as a cyclist or pedestrians hit by car |
6 (55) |
5 (45) |
0 (00) |
7 (64) |
4 (36) |
11 (100) |
|
Fall > 2 m |
2 (20) |
3 (30) |
5 (50) |
8 (80) |
2 (20) |
10 (100) |
|
Fall, same level or unspecified fall |
2 (20) |
1 (10) |
7 (70) |
8 (80) |
2 (20) |
10 (100) |
|
Bicycle accident |
0 (00) |
1 (50) |
1 (50) |
1 (50) |
1 (50) |
2 (100) |
|
Skiing accident |
0 (00) |
1 (100) |
0 (00) |
1 (00) |
0 (00) |
1 (100) |
|
Horse accident |
0 (00) |
2 (100) |
0 (00) |
0 (00) |
2 (100) |
2 (100) |
|
Unknown |
1 (100) |
0 (00) |
0 (00) |
1 (100) |
0 (00) |
1 (100) |
|
Total; group of age or gender |
11 (30) |
13 (35) |
13 (35) |
26 (70) |
11 (30) |
37 (100) |
|
Worst GCS in the first 24 h |
||||||
|
3 |
1 (11) |
3 (33) |
5 (56) |
6 ( 67) |
3 (33) |
9 (100) |
|
4 |
3 (50) |
1 (17) |
2 (33) |
4 (67) |
2 (33) |
6 (100) |
|
5 |
2 (29) |
4 (57) |
1 (14) |
5 (71) |
2 (29) |
7 (100) |
|
6 |
2 (22) |
3 (33) |
4 (44) |
6 (67) |
3 (33) |
9 (100) |
|
7 |
3 (75) |
0 (00) |
1 (25) |
4 (100) |
0 (00) |
4 (100) |
|
8 |
0 (00) |
2 (100) |
0 (00) |
1 (50) |
1 (50) |
2 (100) |
|
Total; group of age or gender |
11 (30) |
13 (35) |
13 (35) |
26 (70) |
11 (30) |
37 (100) |
|
Previous brain injury in need of medical contact |
||||||
|
Yes |
4 (29) |
3 (21) |
7 (50) |
9 (64) |
5 (36) |
14 (100) |
|
No |
7 (30) |
10 (44) |
6 (26) |
17 (74) |
6 (26) |
23 (100) |
|
Total; group of age or gender |
11 (30) |
13 (35) |
13 (35) |
26 (70) |
11 (30) |
37 (100) |
|
Additional injury |
||||||
|
Yes |
6 (46) |
4 (31) |
3 (23) |
8 (61) |
5 (39) |
13 (100) |
|
No |
5 (21) |
9 (38) |
10 (42) |
18 (75) |
6 (25) |
24 (100) |
|
Total; group of age or gender |
11 (30) |
13 (35) |
13 (35) |
26 (70) |
11 (30) |
37 (100) |
|
Traumatic brain injury with signs of influence of alcohol and/or drugs |
||||||
|
Yes |
6 (33) |
5 (28) |
7 (39) |
15 (83) |
3 (17) |
18 (100) |
|
No |
5 (26) |
8 (42) |
6 (32) |
11 (58) |
8 (42) |
1 (100) |
|
Total; group of age or gender |
11 (30) |
13 (35) |
13 (35) |
26 (70) |
11 (30) |
37 (100) |
|
ATV: all-terrain vehicle. |
||||||

Alcohol
Eighteen patients, 15 males and 3 females, (48.6%) were under the influence of alcohol and/or drugs (as demonstrated by clinical assessment, anamnestic information and/or blood test) at the time of injury (Tables II and III). Known current drug and/or alcohol abuse was present in 11 patients (29.7%). Significantly more intoxicated patients (n = 10) had experienced previous TBI in comparison with the non-intoxicated patients (n = 4) (p = 0.040). sTBI due to current high-energy trauma was more common among intoxicated (n = 11) than among non-intoxicated patients (p = 0.041).
Computed tomography scan
Time to first CT scan was less than 1 h for 4 patients, less than 3 h for 20 patients (55%) and less than 4 h for 30 patients (82%). The exact time elapsed from injury to CT scan was missing for 7 patients, all of whom, however, were within time limits for inclusion in the study (Table IV).
|
Table IV. Time to first computed tomography (CT) scan of the brain from accident to first hospital |
|
|
Time from accident |
n (%) |
|
0–60 min |
4 (11) |
|
61–120 min |
11 (30) |
|
121–180 min |
5 (14) |
|
181–240 min |
10 (27) |
|
241–1,320 min |
5 (14) |
|
Total |
35 (95) |
|
Time not available |
2 (5) |
|
Total |
37 (100) |
The first CT scan obtained post-injury showed that 27 patients (73%) had sustained traumatic subdural hematoma. Brain contusion(s) was found in 28 patients (76%), and 29 patients (78%) had traumatic subarachnoidal haemorrhage. Detailed results are shown in Fig 1. and Fig. 3.

Fig. 1. First computerized tomography (CT) scan of the brain compared with age and gender.

Fig. 3. First computed tomography (CT) scan of the brain compared with Glasgow Coma Scale (GCS).
Intracranial pressure
Mean ICP (mmHg) was calculated for each patient for the hour with highest ICP during the first 5 days post-trauma. Mean ICP for 31 of these patients ranged between 15 and 20 mmHg (Fig. 2). For 3 of the patients ICP was not measured, and 3 were, at that initial stage, treated abroad.
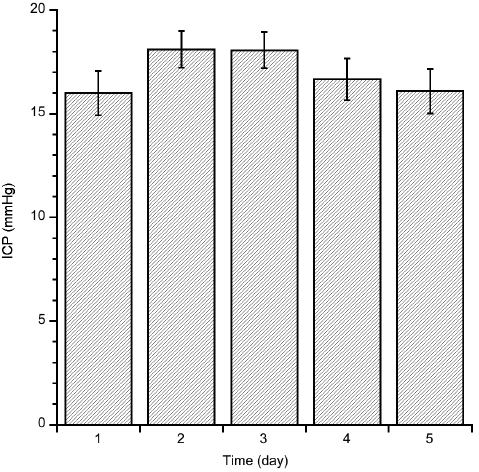
Fig. 2. Mean intracranial pressure (ICP) during the first 5 days post-trauma (n = 31).
Clinical outcomes
Death within 3 months. Hospital deaths within 3 months post-injury occurred in 5 patients (Table V). The primary diagnosis was traumatic subdural haematoma in 4 patients. They received intensive care for 21.6 days (range 19–31 days). Only one of the patients who died had significant additional injuries. Two of the patients who died were intoxicated at the time of injury. Causes of death were, respectively: post-traumatic inoperable arteriovenous fistula and aspiration pneumonia, acute and recurring tracheal bleedings, meningitis and aspiration pneumonia, pneumonia stagnation of secreation in the airways and intracranial rebleeding (after discharge to local hospital).
GOSE and RLAS-R. Table VI shows the distribution of GOSE and RLAS-R scores at 3 months post-injury. The RLAS-R scores were significantly improved from 3 weeks (5.26 ± 3.07) to 3 months (8.0 ± 2.45) (p < 0.001), and 19 patients had “superior functioning” on the RLAS-R IX–X. Eight patients had both a “superior functioning” on the RLAS-R and a “favourable outcome” on the GOSE 7–8. There were no significant differences in outcomes between patients intoxicated or not intoxicated at injury on the GOSE (4.5 ± 2.3 vs 3.9 ± 2.3, p = 0.196) and the RLAS-R (8.7 ± 1.9 vs 7.3 ± 2.7, p = 0.151).
|
Table V. Hospital deaths within 3 months after injury |
||
|
Median (range) |
n (%) |
|
|
Total |
5 (100) |
|
|
Gender |
||
|
Male |
4 (80) |
|
|
Female |
1 (20) |
|
|
Main diagnosis |
||
|
S062 Diffuse brain injury |
1 (20) |
|
|
S065 Subdural haemorrhage |
4 (80) |
|
|
Age, years |
50.8 (19–64) |
5 (100) |
|
Lowest unsedated GCS first 24 h |
||
|
3 |
3 (60) |
|
|
5 |
1 (20) |
|
|
6 |
1 (20) |
|
|
Length of stay in intensive care, days |
21.6 (9–31) |
5 (100) |
|
Length, need of sedation, days |
11 (5–20) |
4 (80) |
|
Additional injury |
||
|
No |
4 (80) |
|
|
Yes |
1 (20) |
|
|
Intraventricular blood or subarachnoid haemorrhage |
5 (100) |
|
|
GCS: Glasgow Coma Scale. |
||
|
Table VI. Lowest unsedated Glasgow Coma Scale (GCS) first 24 h, signs of alcohol and/or drugs at time of injury and Rancho Los Amigos Cognitive Scale Revised (RLAS-R) and GOSE after 3 months. RLAS-R inferior functioning (RLAS I-VIII) and superior functioning (RLAS-R IX–X), Glasgow Outcome Scale Extended (GOSE) Unfavourable outcome (GOSE 1–6), and Favourable outcome (GOSE 7–8) |
|||||
|
Lowest unsedated GCS first 24 h |
n (%) |
Traumatic brain injury n (%) |
Hospital deaths n (%) |
GOSE 1–8 after 3 months n (%) |
RLAS-R after 3 months n (%) |
|
GCS 3 |
9 (24) |
4 (22) |
3 (33) |
GOSE 1–6 = 7 (78) GOSE 7–8 = 2 (22) |
Deaths = 3 (33) RLAS II–VII = 3 (33) RLAS IX–X = 3 (33) |
|
GCS 4 |
6 (16) |
3 (17) |
0 (0) |
GOSE 3–6 = 4 ( 67) GOSE 7–8 = 2 (33) |
RLAS III–VII = 4 (67) RLAS IX–X = 2 (33) |
|
GCS 5 |
7 (19) |
4 (22) |
1 (14) |
GOSE 1–5 = 3 (43) GOSE 7–8 = 4 (57) |
Deaths = 1 (14) RLAS V = 1 (14) RLASIX–X = 5 (71) |
|
GCS 6 |
9 (24) |
4 (22) |
1 (11) |
GOSE 1–6 = 7 (78) GOSE 8 = 2 (22) |
Deaths = 1 (11) RLAS V–VIII = 4 (44) RLAS IX–X = 4 (44) |
|
GCS 7 |
4 (11) |
2 (11) |
0 (0) |
GOSE 4–6 = 2 (50) GOSE 8 = 1 (25) Missing = 1 (25) |
RLAS IX–X = 3 (75) Missing = 1(25) |
|
GSC8 |
2 (5) |
1(6) |
0 (0) |
GOSE 3–5 = 2 (100) |
RLAS X = 2(100) |
|
Total |
37 (100) |
18 (100) |
5 (14) |
GOSE 1–6 = 28 (76) GOSE 7–8 = 8 (22) Missing = 1 (3) |
Deaths = 5 (14) RLAS II–VIII = 12 (32) RLAS IX–X = 19 (51) Missing = 1 (3) |
Clinical care pathways. Most patients (92%) were admitted directly to the regional NC. After discharge, patients were typically transferred back to 1 of several county or local hospitals (Fig. 4). They were also found to commonly be transferred between different departments within a given hospital.
a
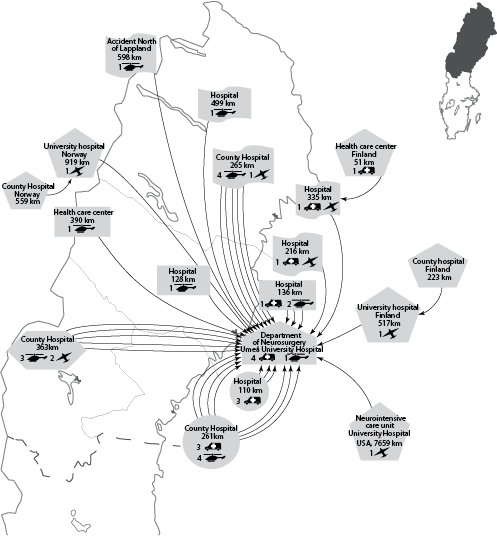
Fig. 4. (a) Clinical care pathway to the Department of Neurosurgery, Umeå University Hospital in northern Sweden (left: map of northern Sweden). (b) Clinical care pathway during 3 months after discharge (right: map of northern Sweden). Rehabilitation NHR: Rehabilitation North Health Region.
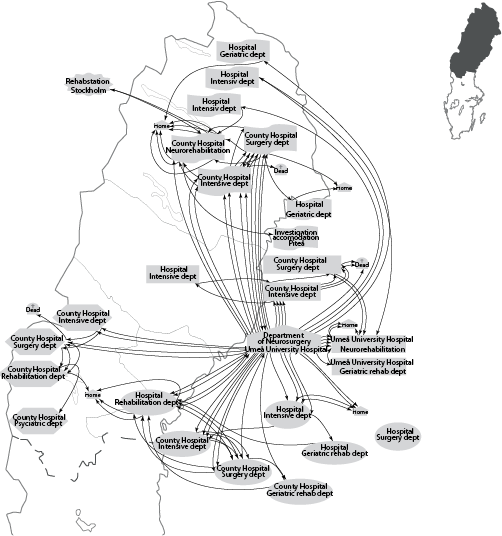
b
DISCUSSION
This study shows the clinical pathways in the NHR in Sweden. In this rural area, which covers almost half of the country, most patients nevertheless are shown to be swiftly transported directly to the regional NC. Thus, routines for pre-acute care seem to be well-established, not only in theory, but also in practice. By contrast, post-acute care after discharge from NC seemingly lacks the structured rigour of the referral and neurointensive processes. Patients are often transferred back to local hospitals at a fairly early stage. Furthermore, in many instances transfers additionally occur across departments. The medical rationale of this dispersion is not clear. There are probably several reasons behind the marked differences between acute and post-acute logistics. Although centralized and standardized treatment and rehabilitation are also likely to be needed in the post-acute stage, the individual patient’s differences and needs are factors that tend to grow in importance as the patient gradually becomes medically stabilized (22), and such aspects may have played a role in the choice of diverging pathways of the patients in the present study. Costs may be another operative factor, as each county has its own budget and has to cover the costs for patient care outside its jurisdiction. In addition, the severity of residual disability and projected prognosis is also likely to be a factor that determines the choice of post-acute clinical pathway.
Well-organized pre-hospital transportation systems for patients with sTBI have also been reported from rural regions of Norway (11). In these areas, rehabilitation in the early phases is based on close collaboration between the neurosurgical departments and rehabilitation units, but capacity problems may delay inpatient rehabilitation (12). Since similar difficulties with insufficient management routines in Sweden and Norway have been observed, researchers recently proposed a Scandinavian organization model that integrates neurointensive care and qualified rehabilitation, and ensures an effective chain of rehabilitation activities after sTBI (12). Differences in post-acute pathways after sTBI have also been demonstrated from other countries. A study from Colorado, USA (23) found that different paths reflected the outcome, and almost 25% of patients with sTBI received no rehabilitation at all. In studies that have evaluated patients with sTBI from rural and urban areas, poorer outcomes for rural residents have often been reported (16). However, with an integrated acute and post-acute network of services, similar results have been shown for rural and urban groups in Australia (24). These findings underline the importance of structured interventions in the early rehabilitation process.
In accordance with previous studies, most patients in the present study were males (4, 11, 25). Falls have commonly been reported as an injury cause typical in TBI affecting children and old persons (4, 11, 25), but were also the most frequent cause in our middle-aged patient population. Nevertheless, motor vehicle accidents are often the cause of injuries in younger persons (11, 25), especially males (4), and contributed to one-third of injuries in the present study. The findings of falls as the leading cause of injury were consistent with a previous study from our hospital (4) of all severity grades of TBI and with a recent Norwegian study of sTBI (25). In contrast, studies from the USA (23) and Australia report motor vehicle injuries as more common than falls both in rural and urban areas (24). This difference may be explained by differences in the infrastructure, traffic intensity, and transport systems in these countries in comparison with Scandinavia.
The male patients in our study had a lower education level in comparison with females, and more males than females were intoxicated at time of injury. Alcohol use at the time of injury has been shown to be a risk factor for TBI (26, 27). Although most studies have shown a relationship between alcohol and poorer outcome after TBI (27, 28), some studies have found no correlation between blood alcohol concentration and TBI outcome (29, 30). In the present study, significantly more patients who were under the influence of alcohol at time of injury had a history of previous TBI and were more often injured by high-energy trauma in comparison with the non-intoxicated patients. However, there was no significant difference between these groups on GOSE and RLAS 3 months post-injury. Based on some laboratory studies it has been argued that alcohol might have a neuroprotective effect (31, 32). To complicate an assessment of the influence of alcohol on prognosis in TBI, it is obvious that a substantial alcohol intake in itself may depress the LOC. Thus, patients intoxicated by alcohol at the time of injury may have lowered GCS scores partially or totally due to alcohol ingestion, rather than due to the severity of brain trauma, and may then be initially classified as having more severe TBI than they actually have.
For assessment of outcomes, the GOSE and RLAS scales were used. All patients improved significantly on the RLAS from 3 weeks to 3 months. At 3 months, of the 19 patients in the 2 highest RLAS categories and the 8 patients on the highest GOSE levels, 3 and 2 patients, respectively, had the lowest GCS score of 3 during the first 24 h in the acute stage. Thus, the majority of the assessed patients experienced good recovery as regards cognitive and behavioural functioning, and around one-quarter was considered as having both “superior cognitive functioning” (15) and a “favourable outcome” (14). However, it is worth noticing that even if positive results on the GOSE and the RLAS were measured, patients may still not be fully recovered at 3 months after the injury and may experience subtle deficits not covered by these instruments. Therefore, it seems reasonable to assume that some of the patients were in need of further rehabilitation interventions and follow-up.
This study has several strengths, such as a prospective design and a close collaboration as regards case identification, which resulted in most patients being included. Even the 6 patients missed for inclusion were subsequently identified, making it possible to account for this small group too. The study design and the well-established acute clinical pathways make it extremely unlikely that study results could be flawed by skewed inclusion. In addition, the extant protocols allow for referral even of those patients with seemingly very poor prognoses. Furthermore, one of the authors (MS) examined all patients, both at 3 weeks and 3 months, and ensured that data were precisely and completely documented. The number of patients in the study was rather small, but comprises the total or near-total regional population of sTBI patients injured during 2 years, and is in accordance with a recent study from northern Norway that also included older patients (25). One limitation of the study is that blood alcohol concentration (BAC) was not measured in all patients, thus decreasing the accuracy in determining the contribution of alcohol to the early clinical picture and the putative effects of alcohol on outcomes. Even with the best intentions, not all trauma patients have BAC measured at the time of admission (33).
In conclusion, the results of this study show that the routines for swiftly transferring all patients with sTBI from a geographically large, sparsely populated, rural area to a single regional NC to receive strict and well-monitored neurosurgical and neurointensive care according to a well-validated protocol seem to work well; something that is also reflected in outcome measurements, in which a high proportion of patients was found to be recovered at 3 months despite very liberal clinical inclusion criteria. In contrast, the post-acute clinical pathways are less clearly reflecting an optimized medical and rehabilitative strategy. The dispersion of patients and frequent transfers between and within a fairly large number of hospitals suggests that non-medical factors are influencing decisions, and raises doubts as to whether it is possible to maintain top-level neurorehabilitation in so many locations. Further research will examine the causes and effects of this state of affairs.
ACKNOWLEDGEMENTS
The authors would like to thank the patients and their relatives, the clinical staff of our unit, especially psychologist Anna-Lisa Nilsson. The authors also thank neuroradiologist, senior consultant Per Jonasson for assessing the CT scans and colleagues at the NC for helping us to recruit patients, colleagues in the intensive care unit and colleagues in rehabilitation department in the NHR. This study was supported by grants from Västerbotten County Council.
REFERENCES
