Michiyuki Kawakami, MD1, Meigen Liu, MD, PhD1, Tomoyoshi Otsuka, MD, PhD2, Ayako Wada, MD2, Ken Uchikawa, MD, PhD3, Asako Aoki, MD1 and Yohei Otaka, MD1
From the 1Department of Rehabilitation Medicine, Keio University School of Medicine, Tokyo, 2Department of Rehabilitation Medicine, National Higashisaitama Hospital, Saitama and 3Department of Rehabilitation Medicine, Saiseikai Yokohamasi Tobu Hospital, Yokohama, Japan
OBJECTIVE: Asymmetrical skull deformity is frequently seen in children with cerebral palsy, and may contribute to postural abnormalities and deformities. The aim of this cross-sectional survey was to determine the frequency of asymmetrical skull deformity and its correlation with clinical parameters.
METHODS: A 10-item checklist for asymmetrical skull deformity, postural abnormalities, and deformities was developed, and its inter-rater reliability was tested. A total of 110 participants aged 1–18 years (mean age 9.3 years (standard deviation 4.7)) was assessed using the checklist. The frequency of asymmetrical skull deformity was analysed and related to the Gross Motor Function Classification System (GMFCS), postural abnormalities, and deformities.
RESULTS: The reliability of the checklist was satisfactory (κ > 0.8). Asymmetrical skull deformity was observed in 44 children, 24 showing right and 20 showing left flat occipital deformity. Its frequency was significantly related to GMFCS and with the patterns of asymmetrical posture and deformities (p < 0.05). Children with right flat occipital asymmetrical skull deformity showed predominantly rightward facial direction and right-side-dominant asymmetrical tonic neck reflex, left convex scoliosis, right-side-elevated pelvic obliquity, and left-sided hip dislocation. Those with left flat occipital asymmetrical skull deformity demonstrated the reverse tendency.
CONCLUSION: Asymmetrical skull deformity is frequent in cerebral palsy and closely related to asymmetrical posture and deformities. This information will be useful to manage these problems.
Key words: cerebral palsy; skull; posture; child; scoliosis
J Rehabil Med 2013; 45: 00–00
Correspondence address: Meigen Liu, Department of Rehabilitation Medicine, Keio University School of Medicine, 35 Shinanomachi, Shinjuku, Tokyo 160-8582, Japan. E-mail: meigenliukeio@mac.com
Submitted April 17, 2012; accepted August 20, 2012
INTRODUCTION
Cerebral palsy (CP) is a non-progressive lesion of the immature brain that results in impairment of movement and postural control. CP is the most common physical disability in childhood (1). Postural problems play a central role in the motor dysfunction of children with CP (2). The performance of everyday activities is noticeably influenced by such postural deficits.
Although CP is, by definition, a static encephalopathy, the associated musculoskeletal pathology is usually progressive (3–6). Hodgkinson et al. (7) reported that scoliosis was observed in 66.2% of non-ambulatory patients with CP. The degree of scoliosis was more than 60 degrees in 34.5% of patients, and two basic groups were distinguished: thoracolumbar scoliosis (41.6%) and lumbar scoliosis (41.6%). The prevalence of an oblique pelvis was 59.9%, with an important difference by side: 31.6% right oblique pelvis and 68.4% left oblique pelvis (7).
These postural deformities can result in secondary problems, such as pain, loss of ability, increased care burden, pressure ulcers, cardiovascular and respiratory problems, swallowing difficulties, and sleep disturbance, all of which are likely to have a significant effect on quality of life (8–12). Therefore, postural problems play a central role in the motor dysfunction of children with CP, and an effective method for their prevention is needed.
Clinically, asymmetrical skull deformity (ASD) is often observed in children with relatively severe CP, and may contribute to the development and aggravation of postural abnormalities and limb and spinal deformities (Fig. 1). No report, however, has analysed the frequency of ASD in children with CP and its relationship with postural abnormalities and deformities. This information would be important in the management of their postural and deformity problems. The objectives of this study were to perform a cross-sectional survey among children with CP to clarify the frequency of ASD and to analyse its relationship with clinical parameters, with emphasis on postural abnormalities and deformities.
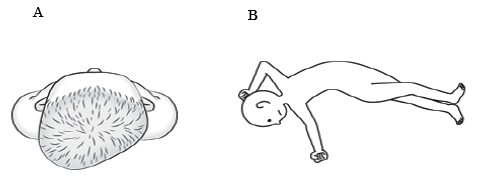
Fig. 1. A) Asymmetrical skull deformity (flattened right occiput). B)Typical postural abnormalities and limb and spinal deformities of a child with asymmetrical skull deformity. The child shows a right-side-dominant asymmetrical tonic neck reflex, left convex scoliosis, and wind-swept hip deformity.
MethodS
Development of a checklist
Based on clinical experience, a 10-item checklist for ASD, postural abnormalities, and deformities was developed (Appendix I). ASD was assessed with palpation and inspection, and it was determined which side of the occiput was flattened. The child was observed both in the supine position and in the sitting position for 3–5 min, and to which side the child’s face was directed and how long the child kept the face turned toward that direction were assessed. We also asked the primary-care givers about the predominant direction of the child’s face. The types of spinal deformities were assessed with palpation and inspection. For pelvic obliquity, the distance between the lowest portion of the rib cage and the anterior superior iliac spine was measured bilaterally. The presence or absence of hip dislocation and wind-swept deformity was assessed with plain antero-posterior X-ray imaging. With standard physical examination techniques, limb positions, muscle tone, the presence/absence and direction of asymmetrical tonic neck reflexes, and head control were assessed. Facial asymmetry was judged based on the positions of the child’s eyes and ears. The degree of head control was classified into “present” and “lowered or absent”.
The checklist was pilot-tested by two physiatrists in 13 children with CP (7 boys and 6 girls) who were hospitalized at two multiply handicapped children’s wards in National Higashisaitama Hospital. At the time of data collection, the children’s ages ranged from 4 to 7 years. To examine inter-rater reliability, kappa coefficients (13, 14) were calculated for individual items.
As for the ASD item in the checklist, its validity was assessed by comparing the clinical judgment with imaging studies. Thirteen participants were examined with head computerized tomography (CT) or magnetic resonance imaging (MRI). The slice level showing the body of the lateral ventricle was selected. The picture of the slice was scanned into a computer, and the intracranial area was calculated with Microsoft Excel 2010. This intracranial area was divided into two portions (right and left) by drawing a line between the falx cerebri and the fissura longitudinalis cerebri, and the ratio was calculated by dividing the larger area by the smaller area (Fig. 2). ASD was graded into 3 categories, i.e. “symmetrical”, “asymmetry apparent with palpation”, and “asymmetry apparent with inspection”, and compared with the right-to-left ratio calculated with the imaging studies. For statistical analyses, Tukey-Kramer multiple comparisons were used (15), setting the significance level at less than 0.05.
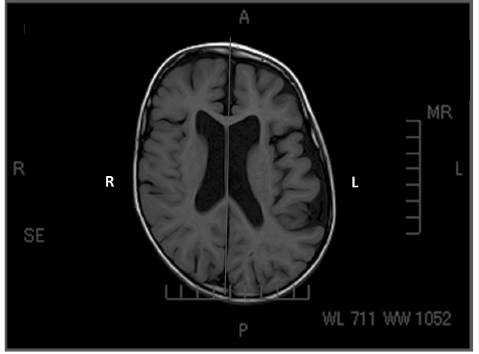
Fig. 2. This is a magnetic resonance imaging. The slice level showing the body of the lateral ventricle was selected. This intracranial area was divided into two portions (right and left) by drawing a line between the falx cerebri and the fissura longitudinalis cerebri, and the ratio was calculated by dividing the larger area by the smaller area. In this case, left-side area/right-side area is 118%.
ASD frequency and relationship with postural abnormalities and deformities
Participants were recruited from 2 day-rehabilitation programmes for handicapped children and 1 hospital for multiply handicapped children. The participants were assessed using the checklist, and the frequency of ASD and its relationship with the Gross Motor Function Classification System (GMFCS) (16, 17), postural abnormalities, and deformities were analysed. For statistical analyses, the χ2 test for independence was used (18), setting the significance level at less than 0.05.
The study was approved by the ethics committee of Keio University School of Medicine, and informed consent was obtained from the patients’ caregivers before participation in the study.
Results
Development of a checklist
For all of the items of the checklist, kappa coefficients were over 0.8 (Table 1). As to the validity of the ASD item, right-to-left ratios calculated using a CT scan or MRI slice were 102.2 % (standard deviation (SD) 2.2) for the “symmetrical” group, 107.7% (SD 6.9) for the “asymmetry apparent with palpation” group, and 117.1% (SD 3.0) for the “asymmetry apparent with inspection” group. The right-to-left ratio of the “symmetrical” group was significantly lower than those of the “asymmetry apparent with palpation” group (p < 0.05) and the “asymmetry apparent with inspection” group (p < 0.01).
|
Table I. Kappa coefficients of all the items of the checklist |
|
|
Item |
κ |
|
Asymmetrical skull deformity |
1.00 |
|
Site of deformity |
1.00 |
|
Which direction do child keep his/her face turned toward? |
1.00 |
|
How long does the child keep his/her face turned toward the direction? |
0.82 |
|
Type of spinal deformity |
1.00 |
|
Pelvic inclination: wind-swept deformity |
1.00 |
|
Pelvic inclination: hip dislocation |
1.00 |
|
Limb position: right upper extremity |
0.87 |
|
Limb position: left upper extremity |
0.86 |
|
Limb position: right lower extremity |
0.89 |
|
Limb position: left lower extremity |
0.88 |
|
ATNR |
1.00 |
|
Facial asymmetry |
0.81 |
|
Stable head and neck |
1.00 |
|
ATNR: asymmetric tonic neck reflex. |
|
Asymmetrical skull deformity frequency and its relationship with postural abnormalities and deformities
The participants were 110 children with CP (51 boys and 59 girls) aged 1–18 years (mean 9.3 years (SD 4.7) ): 96 children (87%) belonged to the spastic type, 8 (7%) to the mixed type, 2 (2%) to the athetotic type, and 4 (4%) to the ataxic type. Overall, 4% of the patients were classified as GMFCS level I, 6% as level II, 2% as level III, 15% as level IV, and 73% as level V.
ASD was observed in 44 children (20 boys and 24 girls). We found no significant gender difference (using the χ2 test for independence test, p = 0.88). Twenty-four had right and 20 left flat occipital deformity. The mean age of the ASD group was 10.0 years (SD 5.2, range 2–18), and that of the non-ASD group was 8.9 years (SD 4.7, range 1–18). The frequency of ASD was significantly related to GMFCS level (Table II). ASD was also significantly related to the patterns of asymmetrical posture and deformities (Table II). That is, children with right flat occipital ASD tended to show predominantly rightward facial direction and a right-side-dominant asymmetrical tonic neck reflex, left convex scoliosis, right-side-elevated pelvic obliquity, and right-sided hip dislocation. Those with left flat occipital ASD demonstrated the reverse tendency.
|
Table II. Frequency of asymmetrical skull deformity (ASD) and its relationship with Gross Motor Function Classification System (GMFCS), postural abnormalities and deformities. Statistical analyses were carried out with the χ2 for independence test, setting the significance level at less than 0.05 |
|||||
|
GMFCS |
|||||
|
I |
II |
III |
IV |
V |
|
|
ASD (+) |
0 |
0 |
0 |
6 |
38 |
|
ASD (-) |
4 |
7 |
2 |
11 |
42 |
|
Right-sided flattened occiput |
Left-sided flattened occiput |
Total |
|||
|
Direction of face (χ2, p < 0.01) |
|||||
|
Right |
15 |
3 |
18 |
||
|
Right/Left |
9 |
6 |
15 |
||
|
Left |
0 |
11 |
11 |
||
|
Total |
24 |
20 |
44 |
||
|
ATNR (χ2, p < 0.01) |
|||||
|
Right |
12 |
1 |
13 |
||
|
Right/Left |
1 |
2 |
3 |
||
|
Left |
0 |
5 |
5 |
||
|
None |
11 |
12 |
23 |
||
|
Total |
24 |
20 |
44 |
||
|
Scoliosis (χ2, p < 0.05) |
|
|
|||
|
Right |
6 |
8 |
14 |
||
|
Right/Left |
1 |
3 |
4 |
||
|
Left |
15 |
3 |
18 |
||
|
None |
2 |
6 |
8 |
||
|
Total |
24 |
20 |
44 |
||
|
Inclination of pelvis (χ2, p > 0.05) |
|||||
|
Right |
9 |
3 |
12 |
||
|
Right/Left |
13 |
14 |
27 |
||
|
Left |
2 |
3 |
5 |
||
|
Total |
24 |
20 |
44 |
||
|
Hip dislocation (χ2, p > 0.05) |
|||||
|
Right |
5 |
3 |
8 |
||
|
Right/Left |
5 |
2 |
7 |
||
|
Left |
1 |
1 |
2 |
||
|
None |
13 |
14 |
27 |
||
|
Total |
24 |
20 |
44 |
||
|
ATNR: asymmetric tonic neck reflex. |
|||||
Discussion
A 10-item checklist was developed to objectively and easily assess ASD, postural abnormalities, and deformities in daily clinical practice, especially in outpatient settings. For all of the checklist items, the kappa coefficients were over 0.8, indicating excellent inter-rater reliability (13, 14).
Because the diagnosis of deformational plagiocephaly is usually made on the basis of history and physical examination findings, and imaging studies are unnecessary in most situations (19–22), ASD was assessed with palpation and inspection in the present study. Its validity was suggested by comparing it with the imaging assessment.
ASD was frequent in children with CP and was closely related to asymmetrical posture and deformities. Although no study has described the frequency of ASD in children with CP, positional plagiocephaly appears to have increased in prevalence since the introduction of the “Back to Sleep” campaign, which recommended placing healthy infants on their backs to sleep (23). Its 2-year prevalence may be as high as 29.5% (24); however, prevalence appears to be age-dependent, with most cases manifesting in the first months of life (point prevalence at 6–7 weeks=16–22.1% (25); at 4 months = 19.7% (24)). By 2 years of age, the point prevalence of positional plagiocephaly may be as low as 3.3% (24).
In the present study sample, the frequency of ASD was 40%, and it was significantly higher than that in healthy infants. Its frequency was significantly related to the GMFCS level, being more frequent in severely involved children. We believe that prolonged immobility could be a contributing factor. In addition, its frequency is so high because there is a higher risk of CP with early birth, assisted delivery and infant neck problems.
Although our study indicated more frequent ASD among children with GMFCS levels 4 and 5, we cannot draw a definitive conclusion about the incidence of ASD according to GMFCS levels, because of the extremely low numbers of people in this study with GMFCS levels 1, 2 and 3. Further study with a more balanced population will be needed.
In healthy infants the Hutchinson et al. study (24) provides evidence to show that the skull shape changes naturally within the first 2 years of life. Because the frequency of ASD is significantly higher in children with CP than in healthy infants, and because children with CP tend to lie supine for a long time within the first 2 years, ASD, once it occurs, tends to persist after the first 2 years.
The diagnosis CP is seldom given during the first year of life, when the ASD develops. For infants at risk, e.g. preterm infants, infants with remaining asymmetric tonic neck reflex, prevention for ASD should be given early.
In healthy infants, plagiocephaly at birth is significantly associated with gender (boys). The preponderance of boys with plagiocephaly was explained by the suggestion of the larger male head circumference and more rapidly growing male head, together with lower flexibility of the male foetuses (26, 27). In our series, we found no significant gender difference. The reason is that boys and girls with CP spend more time in the recumbent position and abnormal posture than do healthy infants.
A recent consensus statement by an expert multidisciplinary group defined a postural management programme for children with CP as “a planned approach encompassing all activities and interventions which impact on an individual’s posture and function” (28, 29). The use of a continuous postural management programme was recommended for children with CP rated as GMFCS levels IV and V, to be used when the children were sleeping, sitting, and standing, with particular emphasis on its role in the prevention of hip dysplasia. In these studies, however, no mention was made regarding positioning related to ASD. We consider that clinicians should take into account ASD and facial direction when providing postural management for children with CP.
In the present study, children with right flat occipital ASD tended to show predominantly rightward facial direction and a right-side-dominant asymmetrical tonic neck reflex, left convex scoliosis, right-side-elevated pelvic obliquity, and right-sided hip dislocation. We assume that, at first, ASD is brought about by a predominantly one-sided facial direction during early childhood when the skull is softer, and once ASD becomes established, it aggravates the asymmetrical posture further. An asymmetrical tonic neck reflex caused by a one-sided face direction could contribute to the development and aggravation of postural abnormalities and limb and spinal deformities. Postural management focused on ASD and facial direction appears to be necessary to prevent deformity in children with CP.
Study limitations
First, the present study was conducted in only 3 institutions, and thus the results must be generalized with caution. The lopsided sample with regard to GMFCS levels is also a limitation. Additional research must be conducted in other establishments including a larger number of children. Secondly, this study was cross-sectional, and a prospective cohort study from early infancy is necessary to demonstrate the time course and causal relationships between ASD and postural abnormalities and deformities.
Despite these limitations, we believe that the present findings are useful for the better management of postural abnormalities and deformities in children with CP.
Acknowledgments
This study was partially supported by R&D Expenditures for Child Development.
References
CP: cerebral palsy; GMFCS: Gross Motor Function Classification System: asymmetric tonic neck reflex; rt: right; lt: left.
Appendix I. A 10-item checklist for asymmetric skull deformity, postural abnormalities and deformities

