Grethe Maanum, MD1,2, Reidun Jahnsen, PT, PhD1,3, Johan K. Stanghelle, MD, PhD1,2, Leiv Sandvik, PhD4,5, Kerstin L. Larsen, PT, MSc1 and Anne Keller, MD, PhD6
From the 1Sunnaas Rehabilitation Hospital, Nesoddtangen, 2Faculty of Medicine, University of Oslo, 3Oslo University Hospital, Rikshospitalet, 4Centre for Clinical Research, Oslo University Hospital, Ullevaal, 5Faculty of Dentistry, University of Oslo and 6Oslo University Hospital, Ullevaal, Oslo, Norway
OBJECTIVE: To investigate face and construct validity of the Gait Deviation Index (GDI) in adults with spastic cerebral palsy. The International Classification of Functioning, Disability and Health (ICF) was used as a framework, defining gait and walking as the manner or style of walking (“body function”), and the execution of gait (“activity”), respectively.
DESIGN: A cross-sectional study.
METHODS: PARTICIPANTS: 66 adults with spastic cerebral palsy, mean age 37 years, and previously collected data on 50 healthy adults (reference population). Variables: GDI from three-dimensional gait analysis, Gross Motor Function Classification System (GMFCS), 6-min walk test (6MWT), Timed Up and Go (TUG), and Physiological Cost Index (PCI).
RESULTS: Mean GDI was 74.3 in adults with cerebral palsy, and 101.1 in the reference population. A significant difference in GDI was found between the reference population and GMFCS level I (p < 0.001), between I and II (p < 0.001), but not between II and III (p = 0.633). The associations between GDI and 6MWT, TUG and PCI were r = 0.30, r = –0.30, and r = –0.56, respectively.
CONCLUSION: GDI demonstrated similar distributional properties as those reported in children with cerebral palsy, suggesting satisfactory face validity. Low correlations between GDI and 6MWT/TUG reflect that gait and functional walking/mobility are different constructs, implicating the importance of selecting outcomes in all ICF domains when evaluating walking ability in adults with spastic cerebral palsy.
Key words: adult; cerebral palsy; gait; Gait Deviation Index.
J Rehabil Med 2012; 00: 00–00
Correspondence address: Grethe Maanum, Sunnaas Rehabilitation Hospital, Bjørnemyr, NO-1450 Nesoddtangen, Norway. E-mail: grethe.manum@sunnaas.no
Submitted June 5, 2011; accepted November 9, 2011
INTRODUCTION
Cerebral palsy (CP) is defined as a motor dysfunction resulting from a non-progressive brain lesion occurring early in an individual’s development (1). Studies show that ambulant adults with CP frequently experience premature walking difficulties (2–4). Gait assessment tools valid in both children and adults with CP would be useful for long-term follow-up in clinical practice and research.
Three-dimensional gait analysis (3DGA) is regarded as the criterion investigative instrument in both research and clinical service for gait impairments in children with CP (5, 6). It provides objective information about joint motions (kinematics), time-distance variables (spatiotemporal data), and joint moments and powers (kinetics). Despite its objectivity, when selecting kinematics from 3DGA as an outcome, it is a challenge to choose one single parameter due to the type and amount of data (7).
A recently developed measure, the Gait Deviation Index (GDI), is based on 3DGA kinematics from the pelvis and hip in 3 planes, from the knee and ankle in the sagittal plane, and from foot progression, providing a total of 459 gait data points (7). By the use of singular value decomposition 15 “gait features” are extracted. Applied to a control group these “gait features” define an averaged, non-pathological gait. The absolute distance between a subject exhibiting gait pathology and the control group is then calculated, providing a measure with good statistical properties from which the extent of gait pathology can be determined (7, 8). Two publications on healthy children and children with CP have demonstrated good face validity, showing that healthy children had a GDI score of approximately 100 with a standard deviation (SD) of 10, while the study populations of ambulant children with CP had a mean score of 72 (SD 10) and 77 (SD 13), respectively (7, 9). To our knowledge, there are no publications on the GDI in adults with CP.
This study aimed to examine the face and construct validity of the GDI for adults with spastic CP. Face validity was studied by investigating whether the GDI had similar distributional properties to those previously demonstrated in child populations (7, 9). Construct validity was studied by investigating the association between the GDI and activity measures for functional mobility and walking capacity, such as the Timed Up and Go test (TUG) and 6-min walk test (6MWT), and energy cost during walking, as measured with the Physiological Cost Index (PCI) (10–12). The International Classification of Functioning, Disability and Health (ICF) (13) was used as a framework. Gait and walking were defined as the manner or style of walking (“body function”), and the execution of gait (“activity”), respectively.
METHODS
Participants
A sample of 66 ambulant adults (n = 30 men) with spastic uni- or bi-lateral CP (14), and no intellectual impairment (normal schooling) were included in this cross-sectional study. The details about recruitment strategy and study population have been reported previously (15). Thirty participants had unilateral and 36 had bilateral spastic CP, classified at GMFCS level I (n = 9), II (n = 48) or III (n = 9). Age range was 18–65 years (mean age 37 years, SD 11.4). In addition, our previously collected data on 50 healthy adults tested at the motion analysis laboratory were used as the reference population (16). The study was approved by the data protection supervisor and the Regional Ethical Committee.
Variables
Data were used from the baseline assessment in the previously reported randomized controlled trial on adults with spastic CP (15). Kinematic data were collected using the Vicon Motion System (Vicon, Oxford, UK) with 6 infrared cameras (MX13, 100 Hz), 2 AMTI OR6-7 force plates (Kistler, Winterhur, Switzerland), and 15 reflective markers positioned on the participants according to the Vicon Plug-In-Gait model. All participants performed their walk without aid, barefoot, and at a self-selected comfortable speed. One researcher (KLL) undertook all the data processing using the Workstation and Polygon software (Vicon Motion Systems). The strides across the force plates with the most similar speeds from 3 gait-trials were selected for further analyses.
To calculate the GDI, the chosen gait-trials were exported from Polygon into an Excel template designed to correctly format the data and then further imported into an Access database developed for the purpose of performing the final GDI calculation, as described by its authors (7). At first, the GDI scores for our reference population were calculated relative to the control data by Schwartz & Rozumalski (7). Following this, the GDI scores for the adult CP participants were calculated using our own reference data. One randomly chosen gait trial with individually calculated right and left GDI scores was used to obtain the descriptive GDI scores for the reference population (n = 100 limbs) and the participants with CP (n = 132 limbs). For subsequent analyses, the GDI scores of the left and right leg were averaged and described as the mean GDI (mGDI) (9); for the participants with CP, the mean of 3 left and 3 right gait trials were used.
GMFCS classification was based on observation as well as information from the participants (15, 17). The GMFCS is so far only published for 5 age bands covering childhood and youth (17, 18). However, the validity and reliability of using the GMFCS in adults with CP have been investigated in 3 studies (19–21).
In TUG, the participants were instructed to rise, walk as quickly and safely as possible for 3 m, turn around, walk back to the chair, and sit down. The fastest of 3 attempts was used, its time measured in seconds down to one decimal (10, 15). For 6MWT, the participants were instructed to walk as fast and as long as possible for 6 min in a hospital corridor with a 30-m marked track. The investigator repeated set phrases every minute during the walk, and the total distance walked was recorded. These tests were performed with the participants’s usual shoes and assisting devices if required (11, 15).
PCI was calculated by the following formula: PCI = (heart rate when walking – resting heart rate)/walking speed (12), using data obtained from the test procedures of 6MWT. Before the 6MWT, the participants were put on a Polar heart rate monitor (Polar Electro Oy, Kempele, Finland), and sat quiet and relaxed for at least 5 min or until their heart rate reached a steady state (resting heart rate). The resting heart rate was attained when heart rate readings taken 1 min apart were within 5 beats of one another. The last value was chosen and controlled by a 15-s palpation of the radial pulse. Immediately after 6MWT was completed, and while the participants was still standing, the Polar monitor was used to record the maximum heart rate. Despite the recognized limitations of this equation, the correlation between heart rate measurement and the volume of oxygen uptake has shown high agreement in children with CP (22, 23).
Statistical analyses
The GDI distribution for the reference population and the group with CP was investigated by descriptive statistics. A one-way analysis of variance (ANOVA) with Tukey’s post hoc test was used to determine significant differences in mGDI scores between the reference population and different GMFCS levels. Pearson’s or Spearman’s correlation (r) was used to estimate the association between mGDI and TUG, 6MWT, and PCI. The magnitude of the r was evaluated as little (0–0.25), low (0.26–0.49), moderate (0.50–0.69), high (0.70–0.89) and very high (0.90–1.0) (24). All statistical tests were two-sided, and a 5% significance level was used. Statistical analyses were performed using SPSS for Windows, versions 15 and 18 (SPSS Inc., Chicago, IL, USA).
RESULTS
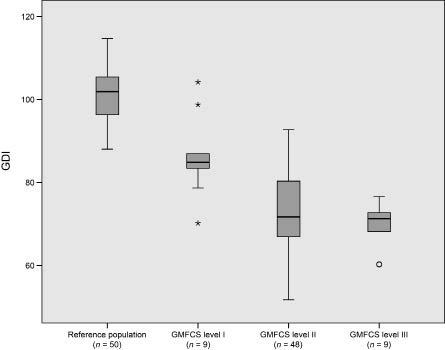
The GDI mean 101.1 (SD 8.8) for the reference population, and the lower GDI scores in adults with CP (mean 74.3, SD 11.6), were similar to previous publications on the GDI’s distributional properties (7, 9). The distributions of mGDI scores for the reference population and the adults with CP at different GMFCS levels are illustrated in Fig. 1, showing statistically significant differences between the reference population and the groups with CP (p > 0.001). There were statistically significant differences in mGDI between participants at GMFCS level I and level II (mean difference 13.5, 95% confidence interval (CI) 5.7–21.2), and also between level I and level III (mean difference 17.0, 95% CI 7.0–27.1). However, there were no significant differences in mGDI between participants at level II and level III (mean difference 3.5, 95% CI –4.2–11.3). The associations between mGDI and functional walking capacity (6MWT), and basic mobility (TUG) were low (6MWT: r 0.30, 95% CI 0.09–0.49; TUG: r –0.30, 95% CI –0.52 to –0.05). A moderate association was found between mGDI and PCI (r –0.56; 95% CI –0.69, –0.38).
Fig. 1. Median and interquartile range of mean right and left Gait Deviation Index scores (mGDI) are presented in the boxes for the reference population and for the cerebral palsy population by GMFCS levels. The whiskers are the lines extending from the top and bottom of the box, representing the minimum and maximum values when they are within 1.5 times above or below the interquartile range. Values outside this range are plotted as outlying or extreme values. GMFCS: Gross Motor Functional Classification System.
DISCUSSION
The face and construct validity of the GDI was investigated in adults with spastic CP. The GDI demonstrated similar results in distributional properties (mean (SD) and between GMFCS-levels) as those reported in studies on healthy children and ambulant children with CP (7, 9). Thus, the GDI appears to discriminate between degrees of gait pathology indicating good face validity both in healthy adults and adults with spastic CP. Low associations between the GDI and the results of 6MWT and TUG suggest that gait and functional walking capacity/mobility are two different constructs, which implicate the importance of selecting outcomes in both the ICF’s “body functions” and “activity” domains when evaluating walking ability in adults with spastic CP.
Except for one short report on amputees (25), no publications have reported associations between the GDI and functional tests similar to daily activities. However, two studies in children with CP have investigated the associations between the GDI and functional walking ability or gross motor function derived from questionnaire scorings, clinical assessments, and standardized observational measurements (7, 9). Finding that the GDI distinguished between these instruments’ different levels, they suggested that the GDI is related to functional walking ability and gross motor function. These results probably reflect that the gait strategy of children with CP is closely related to their performance of gross motor activities in daily life. Our results, which show only a low association between the GDI and 6MWT, and also between the GDI and TUG, probably reflect that other issues beyond a deviating gait strategy may be important for functional walking capacity or mobility in adults with CP (2–4).
We found that gait impairment, as estimated by the GDI, was less associated with basic mobility (TUG) and walking distance (6MWT) than the increased heart rate required for walking (PCI). This finding, of a higher association between two measures in the domain of “body functions”, seems reasonable. The reason that only a moderate association was found is possibly explained by the fact that an increased heart rate during walking does not discriminate between increased energy cost due to gait impairment or low aerobic capacity (12). The implications of these findings are the importance of adding functional tests that cover the “activity” domain, if an index in the domain of “body functions” is selected as an outcome. Also, when evaluating walking ability in adults with CP, our results support the inclusion of a relevant clinical exercise test.
The assumption of a more complex link between the level of gait impairment and walking ability in adults, compared with children, may also be supported by our finding that the GDI did not discriminate between GMFCS level II and III. This is in contrast to other studies, showing that such gait indexes could discriminate between GMFCS level II and III in children with CP (9, 26). The capacity vs performance (i.e. what a person can do vs what he or she does in daily life) may makes GMFCS level III more heterogeneous in adults than in children with CP, because of the element of choice with regard to their use of assistive devices (3, 18–21, 27).
The limitations of the present study include the use of heart rate to estimate energy cost. Furthermore, the participants used their usual shoes and orthotics when performing the 6MWT and TUG, while they did the 3DGA barefoot.
In conclusion, the GDI distinguished different levels of gait impairments in adults with CP and demonstrated its potential to be used as an outcome for research purposes. Future research needs to determine its reliability and responsiveness. Our results support the use of both technical (3DGA) and functional (6MWT/TUG) tests in clinical practice and rehabilitation research, where the topic is increasing walking difficulties in adults with spastic CP. In clinical practice, the Movement Analysis Profile, which uses the same 3DGA kinematics as the GDI, but presents them as nine distinct kinematic variables, may be a more appropriate outcome due to a higher level of details (8, 28).
ACKNOWLEDGEMENTS
This study was supported by the East Regional Health Administration and Sunnaas Rehabilitation Hospital (grant number 206 24 503). The authors are grateful to all the adults with cerebral palsy who participated, and would like to acknowledge A. Opheim, PT, MSc, for practical work with the Access database developed to calculate the GDI.
REFERENCES