Lars Werhagen, MD, PhD and Kristian Borg, MD, PhD
From the Department of Clinical Sciences, Division of Rehabilitation Medicine, Karolinska Institutet
Danderyds Hospital, Stockholm, Sweden
Lars Werhagen, MD, PhD and Kristian Borg, MD, PhD
From the Department of Clinical Sciences, Division of Rehabilitation Medicine, Karolinska Institutet
Danderyds Hospital, Stockholm, Sweden
OBJECTIVE: Pain is a common symptom that affects quality of life in patients with post-polio syndrome. An increase in cytokine in the cerebrospinal fluid suggests that inflammation is pathophysiologically important in post-polio syndrome. Intravenous immunoglobulin might therefore be a therapeutic option. The aim of this study was to analyse the effect of intravenous immunoglobulin treatment on pain in post-polio syndrome.
METHODS: An uncontrolled clinical study. Patients with post-polio syndrome and pain (n = 45) underwent a neurological examination and were interviewed about pain before and 6 months after treatment with intravenous immunoglobulin. Pain intensity was measured on a visual analogue scale. The pain was classified according to the International Association for the Study of Pain criteria as neuropathic when it occurred in an area with decreased sensibility, or nociceptive when signs of inflammation and/or painful joints movements were present.
RESULTS: After treatment 31/45 (69%) patients were improved, with a mean visual analogue scale decrease from 53 to 42 (p = 0.001). Eighteen patients (40%) had a decrease of 20 or more points on the visual analogue scale. The effect of treatment did not differ regarding age, gender and severity of disability. CONCLUSION: Two-thirds of 45 patients with post-polio syndrome and pain reported a decrease on the visual analogue scale for pain after treatment with intravenous immunoglobulin, and 40% reported a decrease of 20 or more points on the visual analogue scale.
Key words: postpolio syndrome; pain; immunoglobulin treatment; nocieptive pain; neuropathic pain
J Rehabil Med 2011; 00: 00–00
Correspondence address: Lars Werhagen, Department of Clinical Sciences, Karolinska Institutet at Danderyd Hospital, SE-182 88 Stockholm, Sweden. E-mail: lars.werhagen@ki.se
Submitted June 21, 2011; accepted August 9, 2011
INTRODUCTION
Post-polio syndrome (PPS), i.e. an increase in symptoms or new symptoms, is often found in individuals who have recovered from paralytic poliomyelitis (1, 2). PPS occurs at least 15 years after a period of stability, and the symptoms include muscular weakness, muscular and general fatigue, as well as pain (1–4). Neurophysiological studies have shown an ongoing uncompensated denervation process (5, 6). Pain is one of the most common complaints in patients with PPS; it affects quality of life and interferes with daily life (7–12). Earlier studies have shown that pain was present most commonly in the joints of the lower extremities followed by the lower back (7, 11). The pain has been described as having a moderate to high intensity (12, 13). Women with PPS are more likely than men to report pain (8, 11). Greater initial motor unit involvement and lower extremity weakness may frequently be additional factors for developing joint pain (9). Werhagen & Borg (14) have described that when neuropathic pain occurred in PPS a concomitant neurological disease was the underlying cause (14).
In a small randomized controlled study (RCT) performed by Farbu et al. (15) showed significant effect of intravenous immunoglobulin (IVIG) on pain, but not on fatigue and muscle strength 3 months after treatment.
In the studies by Kaponides et al. (16) and Gonzalez et al. (17) IVIG had an effect on pain.
The aim of the present study was to further evaluate the effect of IVIG treatment on PPS patients with pain. The effect was evaluated with particular focus on age at the time of examination, gender, age at the time of primary polio infection, and motor index.
METHODS
This study was performed at the post-polio out-patient clinic of the Department of Rehabilitation Medicine, Danderyds University Hospital Stockholm, Sweden. PPS patients referred for treatment with IVIG were examined at the department before inclusion in the study. PPS was diagnosed according to the criteria given by March of Dimes (18).
The included patients received a total of 90 g IVIG (Xepol R, Grifols, Barcelona, Spain) over 3 consecutive days.
Examination before treatment and at follow-up 6 months after treatment included a general and a thorough neurological examination, including sensory testing to map sensory disturbances. Evaluation of motor index was performed. Joint movements were examined as well as a palpation of soft tissue.
The pain was evaluated by one of the authors (LW), and classified according to the International Association for the Study of Pain (IASP). Patients were interviewed about the distribution of their painful area as well as the quality and the temporal profile of the pain before treatment and at follow-up 6 months later. Pain was classified as neuropathic if present in an area with sensory disturbances to pin-prick and light touch and without relation to joint movements and/or infection. Pain was classified as nociceptive when aching in an area with signs of inflammation and painful joint movements was described.
Statistical analysis
Groups and subgroups are presented as absolute numbers and percentages. Categorical variables were compared using a χ2 test. Visual analogue scale (VAS) values before and after treatment were compared using the Wilcoxon signed-rank test. p < 0.05 was considered statistically significant.
Ethics
The study was approved by the Stockholm regional ethics committee.
RESULTS
Forty-five patients with PPS and pain were included in the study. One patient experienced only neuropathic pain, and a combination of nociceptive and neuropathic pain was found in 5 patients. The remaining 39 patients experienced nociceptive pain. Included were 16 males (36%) and 29 females (64%). The mean age for the included patients was 61 years (range 36–88 years). Males were slightly younger than women (58 (range 36–88) and 61 (range 48–80) years of age, respectively). Two-thirds of the included patients, 30 out of 45 (67%), had had their initial polio infection before the age of 6 years. VAS before treatment did not vary with age at the time of examination.
Patients with pain in more than one part of the body had a higher VAS than those who had pain in only one part of the body.
An effect on pain after treatment with immunoglobulin was seen in 31 of the 45 patients (69%).
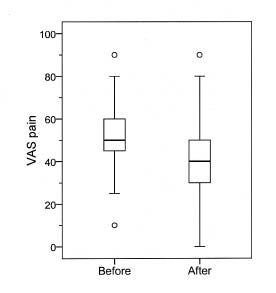
Mean VAS before and 6 months after treatment was 53 (10–90) and 42 (0–90), respectively. This difference reached statistical significance (p = 001) (Figs. 1 and 2). No difference was seen on the effect of IVIG treatment on pain between men and women. Only small differences were seen between patients who had had their acute polio-infection before the age of 6 years and those who had had their infection after the age of 6 years. Small differences were also seen between subgroups of patients after treatment.
Fig. 1. Visual analogue scale (VAS) pain in the 45 patients with post-polio syndrome before and after treatment with 90 g intravenous immunoglobulin.
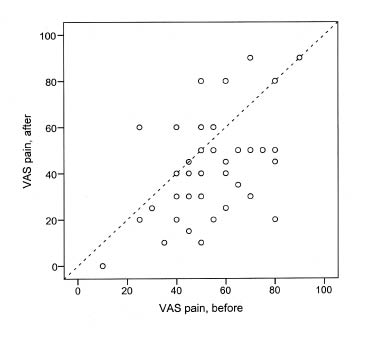
Fig. 2. Box-plot for visual analogue scale (VAS) pain in the 45 patients with post-polio syndrome before and after treatment with 90 g intravenous immunoglobulin, showing the median value and 25th to 75th percentile within the box.
An improvement of more than 20 on the VAS scale was seen in 18 patients (40%) (6 men and 12 women). The mean age was 59 years (range 36–73 years).
Six patients (13%) had no change in VAS before and after treatment and 8 patients (18%) had higher VAS after treatment than before treatment. Those with more pain after treatment were older, with a mean age of 71 years (range 60–88), and 14/18 patients (77%) had their initial polio infection before the age of 6 years. Four of the 5 patients with higher VAS after treatment experienced neuropathic pain in combination with nociceptive pain. The only patient with pure neuropathic pain had no effect of the treatment.
DISCUSSION
The main finding of the present study was a decrease in pain in patients with PPS who were treated with IVIG. Two-thirds of the patients included in the present study experienced a positive effect of the treatment on pain. The results of the present study thus corroborate the findings of earlier studies (15–17). In the studies by Farbu et al. (15) and Kaponides et al. (16) IVIG treatment had an effect on pain. In the study by Gonzalez et al. (17) a better quality of life, better muscle strength and increased physical activity were found. Pain was not a primary endpoint for the study, but when evaluating IVIG treatment in PPS patients with reported pain an effect was found (23). The present study adds a clinical perspective with a more qualitative dimension. The study is, however, a clinical one without a control group, and this has several limitations. The results are therefore difficult to interpret. Perhaps the positive expectations might per se diminish the pain. With a control group it is possible to evaluate a psychological effect of the treatment.
In order to classify and evaluate pain in an adequate way it is essential to interview the patient and to perform a neurological examination including testing of sensory functions.
In the present study the same physician examined and evaluated the patient both before inclusion and at follow-up, which makes it easier to interpret the answer and evaluate the analgesic effect. For the intensity of pain the VAS scale was used, which is simple to perform and easy to evaluate.
Regarding the effect on pain in the present study, there was no difference between males and females, i.e. the effect was not gender-specific and might point to a primary effect for example on the inflammatory reaction. For more information on the background to the clinical effect further studies with IVIG treatment should be followed by determination of cytokines in the cerebrospinal fluid before and after treatment.
The findings of the present study are, however, of greater clinical value, since the study performed by Gonzalez et al. (17) was based on selected patients with inclusion criteria of age and ability to walk, as well as a paresis that would be between 25% and 75% of expected strength. In this study, however, there were no criteria of age, ability to walk or level of paresis. Thus, the present group of patients is more representative for the PPS patients seen at a regular post-polio out-patient clinic.
It was speculated by Gonzalez et al. (17) that the cytokines could affect the motor neurone itself on the basis of a neurodegenerative effect, or affect the neurotransduction-transmission at various levels of the neuromuscular junction. However, Gonzalez et al. did not exclude the possibility that the effect on pain could lead to better motor performance. In the present study there was a difference in pain reduction in patients with different levels of paresis, which may be an argument for a role of pain reduction also in the effect of IVIG on motor function. In future studies it would be of value to separate patients with and without pain when studying the effect of IVIG on motor function.
One also has to consider that pain is a complex symptom and can be caused by many factors, for example damage to the nervous system, arthrosis, other concomitant disorders, overuse of muscles and psychiatric factors such as depression. In the present study the age of patients was high, other disorders were present, and one may expect overuse of muscles and life situations with severe disabilities. Thus, it is likely that other factors may, at least partly, explain the pain.
We conclude that two-thirds of 45 patients with PPS and pain reported a decrease in the VAS score for pain after IVIG treatment. The effect was better in younger patients, patients with more pronounced paresis, and in patients who had their acute polio infection before the age of 10 years.
ACKNOWLEDGEMENTS
BMA Lisbet Broman and Reg Nurse Gullevi Bramert are acknowledged for skilful technical assistance. The study was funded by the research foundations of Karolinska Institutet, Stockholms läns landsting (ALF) and the Gustaf V 80-year Research Foundation.
REFERENCES
