Tiina Rekand, MD, PhD1 and Marit Grønning, MD, PhD1,2
From the 1Department of Neurology, Haukeland University Hospital and 2Institute of Clinical Medicine, University of Bergen, Bergen, Norway
Tiina Rekand, MD, PhD1 and Marit Grønning, MD, PhD1,2
From the 1Department of Neurology, Haukeland University Hospital and 2Institute of Clinical Medicine, University of Bergen, Bergen, Norway
BACKGROUND: Spasticity is a frequent disabling symptom in patients with multiple sclerosis, which contributes to functional deterioration.
OBJECTIVE: To evaluate the long-term effect of intrathecal baclofen therapy in multiple sclerosis-related spasticity and to evaluate the side-effects of long-term therapy, and the doses of baclofen required.
METHODS: Fourteen patients with multiple sclerosis were followed up clinically for a mean of 62 months (range 19–137 months). Clinical evaluation was made using individual goals, modified Ashworth scale, and Kurtzke Expanded Disability Status Scale.
RESULTS: Spasticity, measured with the modified Ashworth score, decreased in all patients by a mean of 1 point. The score on the Expanded Disability Status Scale improved in 2 cases. Prior to implantation, 10 patients (72%) reported severe pain. After implantation 3 improved and 7 became pain-free. The daily doses needed for treatment were highly individual. The effect of intrathecal baclofen on spasticity lasted observation time. One patient experienced progressive cognitive impairment as a side-effect of baclofen.
CONCLUSION: Intrathecal baclofen is well-tolerated and the effect lasts for up to 12 years. A thorough continuous clinical assessment is required because the differentiation between symptoms of multiple sclerosis progression and side-effects of baclofen may be difficult. Intrathecal baclofen should be considered as an option for long-term treatment of patients with advanced spasticity. Pain control can also be achieved by optimized intrathecal baclofen treatment.
Key words: multiple sclerosis; spasticity; pain; rehabilitation; baclofen; ITB.
J Rehabil Med 2011; 43: 511–514
Correspondence address: Tiina Rekand, Department of Neurology, Haukeland University Hospital, NO-5021 Bergen, Norway. E-mail: tiina.rekand@helse-bergen.no
Submitted July 1, 2010; accepted March 17, 2011
Introduction
Management of spasticity is frequently a challenge in neurology, which has both positive and negative effects on patients. Severe spasticity, causing increased loss of function or pain, should be treated. There are several options for management of spasticity. Oral medications, such as baclofen or tizanidine, may cause considerable side-effects and have only partial effects on disabling spasticity. Therefore, intrathecal baclofen (ITB), delivered via a programmable implanted pump, may be an option for management of more severe conditions (1).
ITB was introduced at the end of the 1980s (2). The advantage of ITB is that the drug is delivered directly to the intraspinal space and can therefore have a direct effect on the neuronal receptors of the spinal cord. Systemic side-effects can usually be avoided by a stepwise increase in the dose. The optimal doses are evaluated in relation to the response of each individual. Spastic paraparesis is the primary goal of ITB treatment. Previous data has demonstrated beneficial a long-term effect of ITB treatment among patients with supraspinal and spinal spasticity (3–7). However, observational data for long-term use of an ITB pump in patients with progressive neurological diseases is limited.
More than 30% of patients with multiple sclerosis (MS) report spasticity that interferes with activities of daily living (8). It has been suggested that patients with spasticity related to MS do not need high doses of baclofen (9). It is not known whether this is also the case in long-term therapy. However, the development of tolerance to therapy has been reported (10). At our clinic the first ITB was implanted in 1998. The aim of this study was to identify the long-term outcome, complications and challenges related to ITB treatment of patients with MS.
Material and Methods
The Spasticity Management Clinic at Haukeland University Hospital deals with adult neurological patients with advanced spasticity. Sixteen of our patients have MS. The 14 MS patients who had been treated with ITB for at least 12 months by 1 January 2010 and followed up at the clinic were included in this study. The admitted patients had disabling spasticity that did not respond sufficiently to physiotherapy and oral medication. Patient files were reviewed before and after ITB implantation. Demographic data, symptoms, findings, complications (e.g. infections, complications of therapy, and pain), and the objectives of the patients with respect to ITB treatment, were recorded. All patients had a preoperative test bolus baclofen of 25, 50 or 100 µg intrathecally and the effect on spasticity was evaluated by a physiotherapist and a neurologist 4 h later, as described by Azouli et al. (11). The initial bolus dose was 50 µg. New tests were performed with a 75 or 100 µg dose if the effect on spasticity after the initial dose was not convincing. A dose of 25 µg was used if the patient experienced worsening of function after 50 µg. Patients with at least 2 points decrease in the modified Ashworth score after the test dose were accepted for treatment with ITB. The effect of ITB on spasticity was evaluated in relation to the modified Ashworth scale, Kurtzke Expanded Disability Status Scale (EDSS) and individual goals. Mobility was defined as improvement of transfer from chair to bed or improved sitting position. Information about pain was collected by the initial interview and routinely recorded in the patient files. Pain was categorized as interfering or not interfering with activities of daily living. A reduction in pain was defined as total relief of pain or change to a level not influence daily activities.
Functional level was assessed with the EDSS score, where 0 describes normal functional level and 10 death due to MS (12). An EDSS score of 7 indicates wheelchair dependence. All but one patient had an EDSS score of 7 or more. Neurological findings and spasticity were evaluated prior to and after ITB implantation. The modified Ashworth scale (13) was used for evaluation of spasticity, whereby a score of 0 characterizes normal tone and 5 indicates that the affected part is rigid in flexion or extension. All patients were evaluated individually and goals for improvement of function were established (Table I).
After implanting the pump at the thoracic level (usually Th5-Th6, Synchromed EL or Synchromed II, Medtronic Inc; Minnesota, MN, USA) at the Department of Neurosurgery, the patients were transferred to the Spasticity Management Clinic. The patients stayed at the clinic for one week to evaluate the effect on spasticity and adjust the dosage. Patients were subsequently followed up regularly by the authors for clinical evaluation and refill of the pump. The patients were followed up for a mean 62 months. Indications for changing dosage (spasticity still interfering with treatment goal or overdosage with intrathecal baclofen resulting in an increase in muscle weakness and deteriorating daily activities) were recorded continuously in the patient files. All patients had physiotherapy and oral medication prior to the implantation. After ITB implantation they continued with physiotherapy, but oral baclofen was discontinued.
Results
The individual indications for treatment and the demographic characteristics of patients are presented in Table I. All patients achieved individual goals for treatment. In 6 cases, optimal dosage regarding individual treatment goals was achieved during the first year after implantation, but in 8 cases there was a need for adjusting the dosage throughout the observation period.
|
Table I. Demographic characteristics, indications for, and duration of, intrathecal baclofen (ITB) treatment |
||||
|
Patient number |
Age at ITB (years) |
Sex |
Indication for ITB |
Duration of ITB (months) |
|
1 |
47 |
F |
Improve mobility and prevention of pressure ulcers |
19a |
|
2 |
41 |
M |
Improve mobility |
19 |
|
3 |
62 |
M |
Improve mobility and pain control |
41a |
|
4 |
60 |
M |
Improve mobility and pain control |
27 |
|
5 |
51 |
F |
Improve mobility and pain control |
98 |
|
6 |
38 |
F |
Improve mobility and pain control |
123 |
|
7 |
61 |
F |
Improve mobility and pain control |
137 |
|
8 |
63 |
F |
Improve mobility and pain control |
75 |
|
9 |
32 |
F |
Improve mobility and bladder catheterization |
51 |
|
10 |
44 |
F |
Improve mobility, bladder catheterization and pain control |
54 |
|
11 |
52 |
F |
Improve mobility and pain control |
19 |
|
12 |
26 |
F |
Improve mobility and pain control |
52 |
|
13 |
51 |
F |
Improve mobility and pain control |
132 |
|
14 |
60 |
F |
Improve mobility |
19 |
|
aDeceased. F: female; M: male. |
||||
The mean age at definite MS diagnosis was 37 years (age range 20–57 years), and at ITB implantation 48 years. First signs of spasticity developed a mean of 7 years (range 1–11 years) before the implantation. Mean spasticity measured by the modified Ashworth score in the lower extremities prior to implantation was 3.9 (range 3.5–5). The ITB doses needed were highly individual and did not depend on duration of MS, duration of spasticity, or duration of ITB treatment (Table II, Fig. 1). In the total group of MS patients the modified Ashworth score improved bya a mean of 1 point after ITB implantation (baseline vs 1 year after implantation). Seven patients have been treated with ITB for more than 5 years.
|
Table II. Dosage of intrathecal baclofen (ITB) and duration of multiple sclerosis (MS), spasticity and treatment with ITB |
|
|
ITB dose (µg/day) Mean (range) |
|
|
All MS patients |
173 (55–500) |
|
Duration of MS diagnosis Less than 10 years (n = 4) 11–20 years (n = 6) More than 20 years (n = 4) |
267 (87–500) 143 (77–255) 144 (55–250) |
|
Duration of diagnosed spasticity Less than 10 years (n = 6) More than 10 years (n = 8) |
214 (80–500) 154 (55–265) |
|
Duration of ITB Less than 5 years (n = 7) More than 5 years (n = 7) |
197 (80–500) 155 (55–255) |
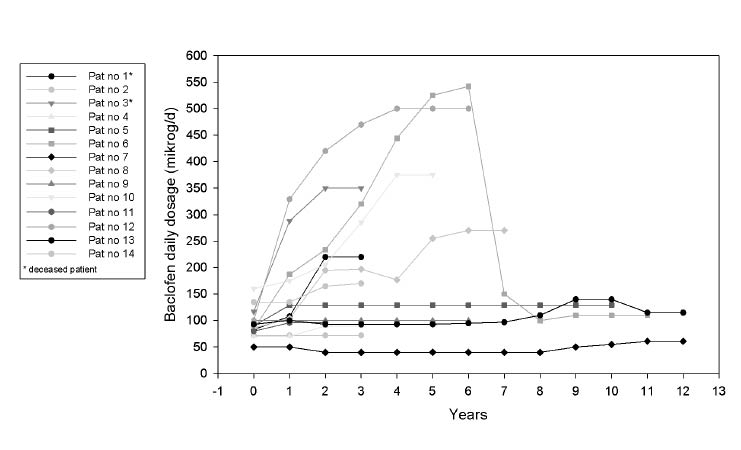
Fig. 1. Baclofen dosage with time since implantation of the pump.
The mean EDSS score was 7.5 (range 6.5–8.5). The EDSS score changed from 7.5 to 7 points in 2 patients (patients two and 8; Table I) after the initiation of ITB treatment, assessed 1 year after ITB implantation. In other cases the EDSS remained unchanged.
Ten patients (72%) reported disabling pain prior to implantation and 3 patients (36%) had considerable problems with activities of daily living (two patients had difficulties with bladder catheterization and one with pressure ulcers due to spasticity).
Seven of the 10 patients with pain prior to implantation became pain-free and 3 patients reported less pain on maximum tolerated doses. In two cases pain was the main cause of functional loss. In both cases, functional status improved considerably after implantation. The effect of ITB on pain lasted for the duration of the observation period.
Two patients were treated with ITB and botulinum toxin simultaneously. One patient experienced worsening of cognitive functions. This was initially considered as a natural course of MS, but was found to be mainly a side-effect of baclofen. Other complications of ITB were not observed in our patients. Two patients died as a result of progression of MS.
Case report
A 48-year-old woman presented with secondary progressive course of MS for 17 years. Seven years after the diagnosis she had developed severe spasticity in all extremities. In the lower extremities the spasticity was evaluated as a score of 4 on the modified Ashworth scale. She became wheelchair-dependent and the ITB was implanted. During the first 6 years after implantation the dosage had been changed from less than 100 µg daily to 542 µg daily because of increasing spasticity (mean Ashworth score 4) in all extremities. Four years after implantation, the daily dose exceeded 450 µg and she reported daytime drowsiness and became less responsive at the same time. The cognitive symptoms were considered to be related to progression of MS. Seven years after implantation, her first ITB was replaced with a new model. The daily dose was lowered postoperatively to 100 µg, which at that time was sufficient for treating the spasticity in her lower limbs. The spasticity in her upper extremities was then treated successfully with Botulinum toxin. Her daytime drowsiness disappeared and her cognitive function normalized.
Discussion
All our patients had advanced generalized spasticity, particularly in the lower extremities, and were treated successfully with ITB over time. The treatment was well tolerated and the patients remained sensitive to ITB during the observation period. There were no pump-related technical problems in these patients and only a few complications were observed. In 6 cases, the ITB dose remained stable for up to 12 years after the optimal dose was identified. Identification of optimal high doses (higher than 150 µg daily) was more challenging and time-consuming than for low stable doses. Optimal high doses were usually identified within the first 36 months. The patients needed highly individualized doses, some required low and some high daily doses immediately after implantation, which suggests that MS-related spasticity might be of both spinal and cerebral origin (14). The development of tolerance and the need for increasing doses of baclofen over the years has been described previously (10).
Symptoms of MS progression and side-effects of baclofen may have similar clinical expression. In the case described here, the cognitive changes were thought to be a sign of disease progression. However, reduction in the dose improved the patient’s cognitive function. Careful and critical screening of side-effects is necessary after an increase in daily doses of baclofen. In addition, interaction with other medications should be considered as a source of cognitive problems. Spasticity in all extremities may be treated with ITB for the lower extremities and with Botulinum toxin A for the upper extremities. This combination allows optimized treatment for all extremities, a lower dose of ITB, and thereby reduced risk of cerebral side-effects.
Seventy-two percent of our patients reported pain prior to, and all our patients experienced less pain after, the ITB. Two patients achieved improved functional status through pain control after initiation of ITB treatment. Pain as a disabling complication related to spasticity in MS patients has been reported previously (15). The treatment of spasticity was therefore expected to alleviate the pain, as has also been shown for other groups of patients (16). Baclofen might also have a direct effect on pain because of its influence on the gamma-aminobutyric acid (GABA) system involved in processing of pain stimuli (17). Neuropathic pain in MS patients may also respond to continuous ITB treatment (18).
All of our patients had improved mobility as a treatment goal. Considerable improvement requires optimized and effective anti-spastic treatment (19). Two patients achieved lower EDSS scores after treatment due to improved mobility. The beneficial effect of ITB for maintenance of ambulatory function has been demonstrated previously in patients with other diagnoses (5). As expected, the EDSS score did not change in all but one patient, as this is a very approximate measure of function. Therefore, individual treatment goals should be established prior to implantation of the pump, preferably by the use of validated scales, which allow for an adequate evaluation at follow-up. Improvement in functional status should be reflected in the scales used for evaluation. General function scales, such as the Functional Independence Measure, Rivermead Motor Assessment and the Fugl-Meyer score, are probably not sensitive enough to measure individual changes in function after ITB treatment and reduced spasticity.
The limitation of this study is that it is retrospective. However, the patients were followed up by the authors for the whole period. The patients were divided into two subgroups according to length of treatment. Patients with less than 12 months’ ITB treatment were excluded.
In conclusion, ITB is an option for treatment of advanced generalized spasticity, particularly in the lower extremities. Our data shows that the effect of treatment is present for up to 12 years, and that the sensitivity of patients with MS to ITB is highly individual. During follow-up it is important to evaluate any increasing or new symptoms and determine whether they are side-effects of baclofen or indicate a progression of MS.
Acknowledgements
The authors thank Dr Ellen Merete Hagen for technical help regarding the figure.
References
