OBJECTIVE: This study aimed to assess short-term effects of botulinum toxin A in ambulant adults with spastic cerebral palsy.
DESIGN: A single-centre double-blind, placebo-controlled, randomized clinical trial.
SUBJECTS: Patients were recruited through advertisements. Inclusion criteria were: spastic cerebral palsy, age 18–65 years, decreased walking, walking without aids for minimum 20 m, and no cognitive impairments.
METHODS: A total of 66 participants, mean age 37 (standard deviation 11.4) years, were enrolled and received injections of either botulinum toxin A (n = 33) or placebo (n = 33). Primary outcomes were: sagittal kinematics of ankle, knee and hip, and health-related quality of life (Short Form 36). Secondary outcomes were: visual analogue scale for muscle-stiffness/spasticity, Timed Up and Go, 6-minute walk test, and Global Scale of perceived effect.
RESULTS: No significant differences were found between the groups in the primary outcomes. In the secondary outcomes the botulinum toxin A group rated improvement in visual analogue scale muscle-stiffness/spasticity and the Global Scale of perceived effect. No serious adverse events occurred.
CONCLUSION: Botulinum toxin A injections alone gave no benefit over placebo in lower limb sagittal kinematics and Short Form 36 in ambulatory adults with cerebral palsy. However, self-reported rating of muscle-stiffness/spasticity and global effects indicated positive effects of botulinum toxin A. Further studies with specific post-injection rehabilitation and longer study period are warranted.
Key words: botulinum toxin; cerebral palsy; adult; gait; muscle spasticity.
J Rehabil Med 2011; 43: 338–347
Correspondence address: Grethe Maanum, Sunnaas Rehabilitation Hospital, Bjørnemyr, NO-1450 Nesoddtangen, Norway. E-mail: grethe.manum@sunnaas.no
Submitted June 25, 2010; accepted December 2, 2010
INTRODUCTION
As cerebral palsy (CP) is a group of permanent motor disorders, caused by a non-progressive disturbance in the immature brain (1), treatment options and research have traditionally focused on the development of optimal motor function in children with spastic CP (2–4). However, a proportion of these children can expect to experience decreasing walking ability in early adulthood (5, 6). Factors contributing to the characteristic gait strategy in spastic CP include neurological factors, such as stretch-sensitive muscle overactivity (7), reduced selective motor control, muscle weakness and impaired balance, secondary factors, such as muscle contractures, bony deformities, and compensatory tertiary mechanisms (3). Surveys have demonstrated that increasing muscle-stiffness and balance difficulties are among the major complaints in ambulant adults with spastic CP (5, 6).
Intramuscular injections of botulinum toxin A (BoNT-A) reduce the force available for muscle contractions, through peripheral reversible chemical denervation (8). Four recent publications reflect the extent of research as well as the consensus about the role of BoNT-A in treatment programmes for children with spastic CP (2–4, 8). The literature reveals that research on troublesome muscular hyperactivity in adulthood has largely concentrated on adult-onset spastic paresis (8, 9). Reviews on spastic paresis and the use of BoNT-A following acquired brain injury, such as stroke and multiple sclerosis (MS), report that BoNT-A reduces resistance to passive movements and demonstrates benefit in Global Satisfaction. However, functional benefits have mainly been demonstrated in open-label studies, and there are few randomized placebo-controlled studies of the lower limbs (8–11).
An important goal in clinical treatment programmes for ambulant children with spastic CP is to maximize gait function and minimize secondary problems such as joint contractures (3). Physiotherapy is the core approach, with a number of complementary strategies, including orthotics, tone reduction by BoNT-A, serial casting and orthopaedic surgery, sequential or in combination (2–4). In stroke survivors and patients with MS, there is also a strong consensus on using BoNT-A in an integrated approach with specific physiotherapy as the most common adjuvant therapy (9, 12).
Due to their longstanding neurological impairment, ambulant adults with spastic CP often have several problem areas, including musculoskeletal pain and fatigue (5, 6). Thus, the most effective and safe management techniques of balancing muscle tone may be different from both developing ambulant children with spastic CP and patients with adult-onset spastic paresis (2, 9, 13). The literature is scarce on both physiotherapy approaches and the use of BoNT-A in the population of high-levelled ambulant adults with spastic CP (5, 14). One randomized controlled trial (RCT), and two open-label trials have demonstrated positive results on lower limb function, self-reported effects and health-related quality of life (HRQOL) from BoNT-A (15–17). However, none of these studies was performed in adults with spastic CP only.
Recognizing that muscle hypertonia in adults with spastic CP has both fixed and modifiable components, such as spastic co-contractions, may be useful in the search for effective management programmes for the impairment from increased muscle-stiffness during walking (7–9, 13, 18). Through its effect of reducing available force to muscle contractions; BoNT-A has the potential of modulating muscle overactivity and balance muscle forces across joints.
We conducted an RCT with parallel group design to investigate the effect of BoNT-A injections on lower limb muscle overactivity in ambulant adults with CP, addressing the effect on outcome measures covering the framework of the World Health Organization (WHO)’s International Classification of Functioning, Disability and Health (ICF) (19). We hypothesized that one treatment sequence with BoNT-A injections would improve: (i) sagittal kinematics (joint angles), (ii) HRQOL, (iii) mobility, and (iv) self-reported outcomes on muscle-stiffness/spasticity and the report of overall treatment effect.
METHODS
The study was a single-centre study at Sunnaas Rehabilitation Hospital (SRH) in Norway, registered in ClinicalTrials.gov (NCT00432055), and reported according to the Consolidated Standards of Reporting Trials (CONSORT). The study protocol was approved by the Data protection supervisor, Regional Ethical Committee, and the Norwegian Medicines Agency (EudraCT number 2006-001427-19).
Subjects
Well-functioning adults with spastic CP were recruited through advertisements in newspapers and on the websites of the Norwegian CP-Association and SRH from November 2006 to August 2008. Responders were screened in a semi-structured telephone interview by the first author, regarding decreased walking ability from adolescence to current age, and, after a referral, examined at the outpatient clinic. Potential participants were responders with spastic CP (20), demonstrating hypertonia as judged with the modified Ashworth scale (MAS) (21), reporting decreased walking ability, and being able to undergo a 3-dimensional gait analysis (3DGA) (3), without devices or support. Decreased walking ability was determined based on the answers given during the telephone interview. A person was defined as having decreased walking ability when one or more of the following problems were reported: (i) reduced walking distance; (ii) increased muscle-stiffness/spasticity and pain during walking; and (iii) increasing balance difficulties (22). The eligibility criteria are presented in Table I, and participant characteristics are presented in Table II. We used the Gross Motor Function Classification System (GMFCS) (23), which categorizes gross motor function into 5 levels (level I being the highest) to classify the respondents’ functional level. MAS, a manual muscle strength test, and popliteal angle were used as indicators of muscle hypertonia, strength, and passive range of motion (ROM) in the lower limbs, respectively (22).
|
Table I. Inclusion and exclusion criteria
|
|
Inclusion criteria
|
Exclusion criteria
|
|
Diagnosis of CPa
|
Cognitive impairmentf
|
|
Spastic unilateral or bilateral CPb
|
Pregnancy or planning pregnancy
|
|
Hypertonicityc in lower-extremity muscle group(s)
|
BoNT-A treatment in the last 6 months
|
|
Age between 18 and 65 years
|
Orthopaedic surgery in the last 18 months
|
|
GMFCS levels I–III
|
Musculoskeletal pathology with no indication for BoNT-A treatmentg
|
|
Decreased walking compared with adolescenced
|
Other diseases that can affect the level of physical function (rheumatoid, neurological, psychological)
|
|
Walking without aids for minimum 20 m
|
|
Gait characterized by functional equinus and/or pathological knee extension or flexion strategye
|
New treatment the past 4 weeks which affect the musculoskeletal system (pain-killers, acupuncture, physical therapy, fitness training)
|
|
No changes in other treatments during the study period
|
|
|
aFrom medical records.
bAccording to the Surveillance of Cerebral Palsy in Europe (20) and verified by clinical investigation.
cDefined as the occurrence of a resistance as judged by the modified Ashworth scale with a score of at least “2” for 1 muscle group or “1” for several muscle groups (21, 22).
dSemi-structured telephone interview.
eVisual observation of gait during a 6-minute walk test and video gait analysis (3).
fDocumented in medical records.
gJudged by the investigator.
CP: cerebral palsy; BoNT-A: Botulinum toxin A; GMFCS: Gross Motor Function Classification System.
|
|
Table II. Characteristics of participants in the study
|
|
Category
|
Placebo
(n = 33)
|
BoNT-A
(n = 33)
|
|
Demographics
|
|
|
|
Gender, n
|
|
|
|
Men
|
16
|
14
|
|
Women
|
17
|
19
|
|
Age, years, mean (SD)
|
38.4 (12.1)
|
36.2 (10.6)
|
|
Married or cohabitant/single, n
|
17/16
|
12/21
|
|
Education (duration), n
|
|
|
|
First level (9 years)
|
7
|
6
|
|
Second level (12 years)
|
11
|
15
|
|
Third level (> 12 years)
|
15
|
12
|
|
Income, n
|
|
|
|
Paid work ≥ 20%
|
18
|
16
|
|
Disablement benefit/unemployed
|
10
|
11
|
|
Student
|
5
|
6
|
|
Diagnosis and functional level
|
|
|
|
Spastic cerebral palsy, n
|
|
|
|
Unilateral
|
14
|
16
|
|
Bilateral
|
19
|
17
|
|
GMFCS present/adolescencea, n
|
|
|
|
Level I
|
4/15
|
5/15
|
|
Level II
|
24/17
|
24/18
|
|
Level III
|
5/1
|
4/0
|
|
Clinical characteristics
|
|
|
|
Medical historyb, n
|
|
|
|
Medication
|
12
|
17
|
|
Other diagnosis
|
14
|
16
|
|
Musculoskeletal surgery lower limb
|
43
|
41
|
|
Previous BoNT-A treatment
|
2
|
2
|
|
Lower limb impairmentc
|
|
|
|
Median muscle hypertonia (IQR)
|
1.6 (0.9–2.7)
|
1.3 (0.8–1.9)
|
|
Median muscle strength (IQR)
|
4.6 (4.3–4.8)
|
4.5 (4.2–4.6)
|
|
Mean popliteal angle (SD)
|
50.9 (10.5)
|
53.5 (11.2)
|
|
Training, n
|
|
|
|
Never
|
4
|
2
|
|
Every month
|
7
|
7
|
|
Every week
|
7
|
10
|
|
Several times a week
|
15
|
14
|
|
Physiotherapy, n
|
|
|
|
Never
|
8
|
6
|
|
Every month
|
8
|
5
|
|
Every week
|
13
|
17
|
|
Several times a week
|
4
|
5
|
|
Orthotics/orthopaedic shoes, n
|
8
|
9
|
|
aPresent level classified by principal investigator (GM), adolescent level classified by GM on the basis of information from the participants (22).
bFrom medical records, several participants have more than 1 medication and/or other diagnosis and/or previous surgery procedure.
cModified Ashworth scale (0–4) for 8 different muscle groups (bilateral hip adductors, knee-extensors, knee flexors and ankle plantar flexors) calculated as the mean of the measurements; manual muscle testing (0–5) for 14 different muscle groups (bilateral hip flexors/extensors/abductors, knee flexors/extensors and ankle plantar-/dorsalflexors) calculated as the mean of the measurements; and unilateral popliteal angle for left and right side calculated as the mean of the measurements (22).
SD: standard deviation; IQR: inter-quartile range; BoNTA: botulinum toxin A; GMFCS: Gross Motor Function Classification System.
|
Procedures
Potential participants were assessed by a multidisciplinary team through a standardized clinical history-taking and a clinical examination, and visual observation of gait (3, 22, 24). Persons fulfilling the eligibility criteria (Table I) were invited to participate in the study. After obtaining written consent, baseline assessments were conducted and treatment goals defined. The principal therapeutic goal was to improve gait, thus resolving complaints such as stiffness due to muscle overactivity, pain, and reduced mobility. Intramuscular injections were administered within one week after the pre-treatment assessment. Two of the participants were assessed 3 and 6 weeks prior to therapy, respectively. The post-treatment assessment was exactly 8 weeks after the injections. All assessments were performed by the same experienced multidisciplinary team, physician (GM) and 1 of 3 physiotherapists, never reviewing the previous test values. The questionnaire used at week 8 was sent to the participants with a prepaid stamped return envelope at 16 weeks.
Drug treatment was BoNT-A; a dilution of 50 U/ml of Botox® (Purified Neurotoxin Complex, Allergan, Inc., Irvine, CA, USA). Placebo treatment was 0.9% saline. BoNT-A, and placebo syringes of 2 ml were prepared by a nurse and a pharmacist at SRH. All the injections were performed by the same physician (GM). Electromyography (EMG) and electrical stimulation were used to confirm the presence of abnormal muscle activity and to guide the injections (15, 25). Selection of target muscles was based on gait evaluation, clinical examination and electrical response (Table III). Dosing was based on the 2002 Guidelines (26); the specifications are given in Table IV. Usual drug therapies and/or physiotherapy/training were allowed, when they were unchanged throughout the study. Training was defined as subsets of physical activities planned and structured with repetitive bodily movements done to improve or maintain one or more components of physical activity and fitness (27). Side-effects and unintended effects were registered at weeks 8 and 16.
|
Table III. Target muscle identification for injectionsa
|
|
Target muscles
|
Criteria observed during gait analysisb
|
Criteria observed during clinical examinationc
|
Criteria during intervention
|
|
Medial hamstringsd and biceps femoris
|
Increased flexion of the knee during initial contact and stance phase
|
Increased muscle tone
Increased popliteal angle, but not > 90º
|
Positive response from auditive EMG and electrical stimulation of motor point
|
|
Rectus femoris
|
Decreased progression to flexion of the knee joint during initial swing phase
|
Increase muscle tone
Patella alta
|
Positive response from auditive EMG and electrical stimulation of motor point
|
|
Adductorse
|
Adduction of the hip interfering with stability during gait cycle
|
Increased muscle tone
|
Positive response from auditive EMG
|
|
Gastrocnemius
|
Increased knee flexion during stance and/or pathological plantar flexion of the ankle during gait cycle
|
Increased muscle tone
Reduced maximum ankle dorsiflexion with knee extended, but not less than –20º
|
Positive response from auditive EMG
|
|
Soleus
|
Plantar flexion of the ankle during stance phase
|
Increased muscle tone
Reduced maximum ankle dorsiflexion with knee 90º flexed but not less than –10º
|
Positive response from auditive EMG and electrical stimulation of motor point
|
|
Tibialis posterior
|
Varus/adduction/supination of the forefoot during gait cycle
|
Callus (signs of overload) on the lateral border of the foot
|
Positive response from auditive EMG and electrical stimulation of motor point
|
|
Flexor digitorumf
|
Toe flexion during stance phase
|
Callus (signs of overload) of the toes
|
Positive response from auditive EMG and electrical stimulation of motor point
|
|
aBotulinum toxin A (Botox®) 50 U/ml or placebo (0.9% saline). bVisual observation of (i) gait strategy in the 6-minute walk test, and (ii) sagittal and frontal plane video assessments from 3-dimensional gait analysis (3). cIncreased muscle tone as judged by a score on modified Ashworth Scale ≥1 (21, 22). dSemitendinosus and semimebranosus. eAdductor longus and – magnus. fLongus and brevis; brevis identified with electromyogram (EMG) only.
|
|
Table IV. Injections given in the study. All results presented as n
|
|
Muscle
|
Placebo
(n = 33)
|
BoNT-A
(n = 33)
|
Pre-specified injection dose
|
|
Rectus femoris
Participants not injecteda
Number of injected musclesb
Hamstringsc
Participants not injected
Number of injected muscles
Gastrocnemius
Participants not injected
Number of injected muscles
Soleus
Participants not injected
Number of injected muscles
Tibialis posterior
Participants not injected
Number of injected muscles
Flexor digitorumd
Participants not injected
Number of injected muscles
Total number of injected muscles
Total (ml) injected
|
25
16
20
24
7
38
23
11
23
13
31
2
104
205.5
|
27
12
20
22
6
40
22
11
23
14
32
2
101
201.0
|
1.5–2 ml (75–100 units Botox®)
2–7 ml (100–350 units Botox®)
3–4 ml (50–200 units Botox®)
2 ml (100 units Botox®)
1.5 ml (75 units Botox®)
0.5 ml–1.5 ml (25–75 units Botox®)
|
|
aNumber of participants not receiving injection into defined muscle. bNumber of muscles injected; for several of the participants bilateral treatment. cSemitendinosus 1–1.5 ml (50–75 U Botox®), semimembranosus 1–1.5 ml (50–75 U Botox®), biceps femoris 2 ml (50–100 U Botox®). dFlexor digitorum brevis 0.5 ml (25 U Botox®), flexor digitorum longus 1.5 ml (75 U Botox®) (26). Maximum dose 8 ml (400 U Botox®), 1 ml per injection site.
BoNTA: botulinum toxin A.
|
Outcomes
The outcome measures were made at weeks 0 (baseline), 8 and 16 (Fig. 1). The primary outcome measures were kinematics at predefined gait events and HRQOL. The secondary outcomes were on basic mobility and subjective reports of muscle-stiffness/spasticity and global treatment effects.
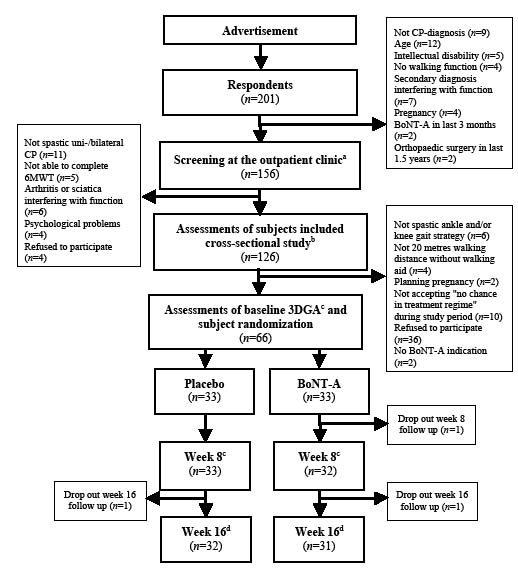
Fig. 1. Study flow of participants in the present study. aReferral from the responders’ general practitioner or neurologist. bMaanum et al. (22). c3-dimensional gait analysis (3DGA); otherwise data from cross-sectional study used as baseline data. Outcome measures at week 8 evaluation: 3DGA, Timed Up and Go, 6-minute walk test, 36-item Short Form Health Survey (SF-36), visual analogue scale (VAS) muscle-stiffness/spasticity, Global Score, and registration of adverse events. dOutcome measures at week 16 evaluation: postal questionnaire included SF-36, VAS muscle-stiffness/spasticity, Global Score, and registration of adverse events. CP: cerebral palsy; BoNTA: botulinum toxin A.
Primary outcomes
Kinematics (joint angles) was assessed by 3DGA with a 6-camera VICON 527 motion analysis system (Oxford Metrics Ltd, Oxford, UK), 15 passive reflective markers and 2 AMTI forceplates (Kistler, Winterhur, Switzerland)). Each individual walked 20 m at a comfortable speed until at least 3 successful recordings of kinematic and kinetic information were obtained. Workstation and Polygon software were used to collect and process the raw data, and Pipeline External Communication Server (PECS, Vicon, Oxford, UK) extracted the following joint angles: (i) ankle dorsiflexion at initial contact, peak ankle dorsiflexion in stance- and swing-phase (ii) knee extension at initial contact, peak knee extension in stance- and swing-phase, and (iii) hip ROM during a gait cycle. Except for 3 subjects, with marginal or inconsistent ambulation, data from 3 trials were averaged. One side was analysed in each subject; randomly chosen, right side for bilateral and affected side in unilateral CP. Based on data from a reliability study on 12 adults with spastic CP (preliminary results), a mean change of more than 5° was considered to both exceed the limits of measurement error and to represent a clinically meaningful change.
The Norwegian version of Short Form 36 (SF-36 1.2) was used to measure HRQOL (28). The SF-36 is a 36-item validated generic measure providing information about 8 domains of subjective physical and mental health, derived from the summation of item scores and transformed to a 0–100 scale, with higher numbers representing better health (29). The minimum clinical important difference (MCID) for SF-36 has not been defined in CP. However, in rheumatoid arthritis, a MCID of 5–10 points has been estimated. We chose the most conservative alternative, 10 points, in this study (30, 31).
Secondary outcomes
The Timed Up and Go test (TUG) was used to evaluate basic mobility (32). Participants were instructed to rise, walk as quickly and safely as possible for 3 m, turn around, walk back to the chair, and sit down on set cues. The fastest of 3 trials, measured in seconds with one decimal point, was used (22). A study on MS has reported changes of 21% improvement or 27% deterioration to be clinically meaningful (33).
The 6-minute walk test (6MWT) was used to measure functional walking capacity (34). Participants were instructed to walk as fast and as far as possible for 6 min in a quiet hospital corridor with a 30 m marked track. The investigator repeated set phrases every minute during the walk. The total distance walked was recorded (22). A study on adults with CP has reported changes of 40 m to be clinically meaningful (35).
A 100-mm visual analogue scale (VAS) was used to determine the participants’ experience of lower limb muscle-stiffness/spasticity during walking, the ends representing the extreme limits of muscle-stiffness/spasticity experience. A 0–10 point spasticity numeric rating scale has been demonstrated to be a valid and reliable tool in the assessment of muscle-stiffness/spasticity in persons with MS, where 30% change has been reported to be clinically meaningful (36).
A Global Scale was used to document overall self-reported therapy effects, consisting of a 3-point verbal rating scale (worse – the same – better) (16).
Sample size
An a priori power calculation was conducted according to the assumption that we could achieve a standardized mean difference at follow-up of 6º in maximum ankle dorsiflexion in stance and a standard deviation (SD) of 8º (24). A significance level of 0.05 and power at 80% resulted in a sample size of 28 in each group. Five patients were added to each group in case of drop-outs.
Randomization and allocation concealment
The Department of Biostatistics, University of Oslo, independently produced a computer-generated randomization list for trial drug allocation, with the SRH pharmacy undertaking masking, dispensing and labelling. Blocks of 10 were used to ensure fairly even-numbered treatment groups. Each eligible participant was assigned an identification number at the time of inclusion, which corresponded to the treatment allocation schedule stored by the hospital pharmacist.
Blinding
In order to conceal the identity of interventions from investigators and participants, a pharmacist and a nurse who were not otherwise involved in the study prepared the BoNT-A or saline vials. The syringes were labelled with stickers that made them appear identical. All investigators were blinded to the subjects’ group assignment during the study and the explorative statistical analyses. Participants were instructed not to discuss their treatment experience with the assessors. A statistician, who was not previously involved in the study, received the study data and decided on the statistical analyses before the randomization list was delivered.
Statistics
Descriptive statistics were generated for the sample characteristics and outcome variables at baseline and follow-up separately for the two groups. There were no missing data.
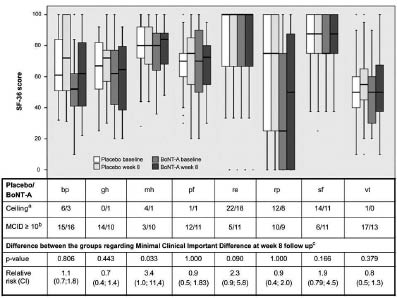
Due to the demonstrated robustness of parametric statistics for moderately skewed data (37), and because analyses with non-parametric statistics and bootstrap confidence intervals (CIs) gave similar results, a paired sample t-test was used to analyse changes in continuous variables from baseline in each group. As the residual check was found satisfactory, analysis of covariance was used to compare differences between the groups in primary and secondary outcomes at weeks 8 and 16, with the week 8 or 16 score as dependent variables and baseline score and treatment group as independent variables. The Fischer’s exact test with relative risk (RR) and risk ratio were applied to compare the Global Scale scores between the two groups. These statistical methods were also used in secondary explorative analyses of the proportions of participants achieving a clinically relevant improvement for the SF-36 scales and VAS-muscle-stiffness/spasticity. The results of the 8 scales in SF-36 were visualized by means of box-plots (Fig. 2).
Fig. 2. Short Form Health survey (SF-36) presented with its 8 domains: bodily pain (bp), general health (gh), mental health (mh), physical function (pf), role emotional (re), role physical (rp), social function (sf) and vitality (vt). aNumber of participants in each group with the maximum score of 100 at baseline. bNumber of participants improving their score from baseline to week 8 ≥ 10 (predefined minimum clinical important difference (MCID). cFischer's exact test. p < 0.05; BoNTA: botulinum toxin A; CI: confidence interval.
The results presented are based on per-protocol analysis (n = 65) at week 8 and the last results carried forward at week 16 (n = 65). All statistical tests were two-sided, and a 5% significance level was used. Ninety-five percent CIs for differences from baseline were presented in order to identify significant changes. Statistical analyses were performed using SPSS for Windows, version 15 (SPSS Inc., Chicago, IL, USA).
RESULTS
The study was undertaken between November 2006 and January 2009. Of 201 respondents 66 persons (30 men) were enrolled in the study (Fig. 1). Mean age was 37 years, age range 18–62 years. Demographics, type of CP, GMFCS, SF-36, and lower limb impairment showed no statistically significant differences from the population of n = 126 assessed for eligibility (Fig. 1). The study population was, in general, a high-functioning group with the majority having higher education, being employed or students, and classified at GMFCS level II. Two in the placebo group reported no regular physiotherapy or training. Otherwise, 64 of the participants reported varying degrees of regular physiotherapy and/or fitness training (Table II). The total doses, and muscles selected for injection, were similar between the groups (Table IV). The follow-up rate was 65 at week 8 and 63 at week 16 (Fig. 1).
Outcomes
The 95% CI of between-groups difference demonstrated no statistically significant or clinically relevant benefits of BoNT-A on the kinematic variables at week 8 (Table V). All differences in the kinematics, also at group level (week 8 vs baseline), were small with narrow CIs.
|
Table V. Primary outcomes: dynamic range of motion (kinematics) at defined gait events and 36-item Short Form Health Survey (SF-36)
|
|
|
Placebo (n = 33)
|
BoNT-A (n = 32)a
|
Between groups (n = 65)
Adjusted difference
Mean (95% CI)
|
|
|
Baseline
Mean (SD)
|
Week 8
Mean (SD)
|
Difference
Mean (95% CI)
|
Baseline
Mean (SD)
|
Week 8
Mean (SD)
|
Difference
Mean (95% CI)
|
|
Kinematics
|
|
|
Ankle initial contact
|
–6.2 (7.1)
|
–6.9 (6.6)
|
–0.7 (–2.2 to 0.8)
|
–7.2 (6.0)
|
–7.5 (5.9)
|
–0.3 (–1.5 to 1.1)
|
0.2 (–1.6 to 2.1)
|
|
Ankle peak stance
|
14.6 (4.7)
|
14.5 (4.3)
|
–0.1 (–1.2 to 1.3)
|
10.7 (4.7)
|
11.8 (5.5)
|
1.1 (–0.3 to 2.4)
|
0.2 (–1.7 to 2.0)
|
|
Ankle peak swing
|
–0.2 (4.9)
|
–0.5 (5.3)
|
–0.3 (–1.7 to 1.1)
|
–1.8 (6.2)
|
–1.8 (6.5)
|
0.0 (–1.2 to 1.3)
|
0.1 (–1.7 to 1.9)
|
|
Knee initial contact
|
17.2 (8.8)
|
17.5 (8.2)
|
0.3 (– 2.4 to 1.6)
|
17.4 (7.6)
|
16.1 (6.8)
|
–1.3 (0.0 to 2.6)*
|
–1.6 (–3.7 to 0.5)
|
|
Knee peak stance
|
7.6 (8.6)
|
7.9 (7.6)
|
0.3 (–2.0 to 1.4)
|
6.0 (8.6)
|
5.3 (7.0)
|
–0.6 (–1.0 to 2.3)
|
–1.4 (–3.3 to 0.5)
|
|
Knee peak swing
|
53.2 (8.6)
|
52.8 (8.7)
|
–0.4 (–2.2 to 1.4)
|
53.7 (6.9)
|
52.8 (8.1)
|
–0.8 (–2.0 to 0.3)
|
0.4 (–1.7 to 2.4)
|
|
Hip range of motion
|
41.1 (7.5)
|
41.7 (7.4)
|
0.6 (–1.7 to 0.5)
|
43.4 (6.8)
|
42.6 (6.8)
|
–0.8 (–1.8 to 0.2)
|
–1.2 (–2.6 to 0.2)
|
|
SF-36
|
|
|
Mental health
|
77.7 (16.8)
|
79.3 (15.9)
|
1.6 (–2.6 to 5.8)
|
74.4 (14.6)
|
78.5 (15.0)
|
4.1 (–0.9 to 9.1)
|
1.4 (–4.4 to 7.2)
|
|
Vitality
|
51.5 (22.8)
|
56.5 (22.6)
|
5.0 (–1.3 to 11.3)
|
45.2 (15.5)
|
51.2 (21.9)
|
6.1 (1.4 to 10.8)*
|
–0.27 (–7.8 to 7.7)
|
|
Bodily pain
|
64.8 (22.1)
|
72.9 (24.1)
|
8.1 (1.5 to 14.7)*
|
54.4 (24.7)
|
61.0 (23.6)
|
6.6 (0.3 to 12.8)*
|
–4.4 (–12.9 to 4.2)
|
|
General health
|
63.5 (18.9)
|
67.6 (19.2)
|
4.2 (–0.4 to 8.8)
|
59.8 (22.9)
|
60.3 (22.8)
|
0.4 (–5.8 to 6.6)
|
–4.7 (–11.8 to 2.4)
|
|
Social function
|
83.3 (17.9)
|
84.1 (20.8)
|
0.8 (–5.3 to 6.8)
|
80.1 (18.7)
|
85.6 (15.9)
|
5.5 (–0.2 to 11.2)
|
3.4 (–4.0 to 10.9)
|
|
Physical function
|
64.9 (17.8)
|
68.8 (20.0)
|
3.9 (–0.8 to 8.7)
|
67.8 (20.7)
|
69.7 (17.5)
|
1.9 (–3.4 to 7.1)
|
–1.2 (–7.6 to 5.2)
|
|
Role physical
|
54.6 (42.1)
|
63.6 (39.6)
|
9.1 (–5.1 to 23.3)
|
43.8 (39.1)
|
46.9 (40.0)
|
3.1 (–11.8 to 18.1)
|
–11.6 (–29.1 to 5.9)
|
|
Role emotional
|
77.8 (36.0)
|
79.8 (33.3)
|
2.0 (–8.2 to 12.2)
|
69.8 (39.1)
|
82.3 (29.3)
|
12.5 (–2.4 to 27.4)
|
5.7 (–8.1 to 19.5)
|
|
*p ≤ 0.05. aThe BoNT-A group had 1 drop out. Data are mean standard deviation (SD) for placebo and botulinum toxin A (BoNT-A) at baseline and week 8, and mean (95% confidence interval (CI)) from paired t-test and adjusted mean from analysis of covariance using group and baseline values as independent variables and results on week 8 as dependent variables. Adjusted mean difference = BoNT-A group vs placebo group.
|
No statistically significant differences were found between the groups in HRQOL for either of the domains at week 8 (Table V and Fig. 2) or week 16 (data not shown). Both groups demonstrated significant improvement at week 8 for bodily pain, and the BoNT-A group also showed significant improvement for vitality (Table V). Neither of the groups demonstrated any significant changes at week 16 (data not shown). Several of the SF-36 domains showed the upper limits of CIs exceeding the predefined MCID on the group level as well as between the groups (Table V). Exploratory analyses investigating the number of participants who had improved according to the predefined MCID for SF-36 showed a statistically significant difference in favour of the BoNT-A group for mental health at week 8 (Fig. 2), and for mental health and social functioning at week 16 (data not shown).
At week 8 outcomes on mobility showed no significant differences between the groups. Both groups had a statistically significant improved 6MWT distance, whereas only the BoNT-A group had a statistically significant improvement for the TUG (Table VI).
|
Table VI. Secondary outcomes: visual analogue scale muscle-stiffness/spasticity, 6-minute walk test, Timed Up and Go, and Global Score
|
|
|
Placebo (n = 33)
|
BoNT-A (n = 32)a
|
Between groups (n = 65)
|
|
Baseline
Mean (SD)
|
Week 8
Mean (SD)
|
Difference
Mean (95% CI)
|
Baseline
Mean (SD)
|
Week 8
Mean (SD)
|
Difference
Mean (95% CI)
|
Adjusted difference
Mean (95% CI)
|
|
Muscle-stiffness/spasticityb
|
45.8 (22.7)
|
40.7 (21.0)
|
–5.1 (–12.2 to 2.0)
|
41.5 (24.9)
|
28.6 (21.6)
|
–13.0 (–20.9 to –5.0)**
|
–9.6 (–18.7 to –1.2)*
|
|
6-minute walk test
|
493.4 (74.7)
|
504.4 (69.2)
|
11.0 (1.7 to 20.2)*
|
495.1 (92.1)
|
514.5 (94.8)
|
18.6 (4.2 to 33.4)*
|
7.9 (–8.3 to 24.1)
|
|
Timed Up and Go
|
7.4 (2.6)
|
7.3 (2.6)
|
–0.14 (–0.5 to 0.2)
|
7.3 (1.9)
|
6.9 (2.0)
|
–0.34 (–0.6 to –0.1)*
|
–0.21 (–0.6 to 0.2)
|
|
|
Positive treatment effect (n)
|
Positive treatment effect (n)
|
Relative risk (95% CI)
|
Risk difference (95% CI)
|
|
Global Scorec
|
9
|
19
|
2.2 (1.96 to 2.44)*
|
32% (9.3% to 54.9%)*
|
|
*p < 0.05, **p < 0.01. Data are mean (SD) for placebo and botulinum toxin A (BoNT-A) at baseline and week 8, and mean (95% CI) from paired t-test and adjusted mean from analysis of covariance using treatment group and baseline values as independent variables and results on week 8 as dependent variable.
aThe BoNT-A group had 1 drop out.
bVisual analogue scale 0–100 mm.
cThree-point verbal rating scale (worse – the same – better). The numbers regarding positive treatment effects correspond in percent to 27% and 59%, respectively. One participant in each group scored “worse”; in data analyses for the risk statistics they were both classified as “the same”.
SD: standard deviation; CI: confidence interval.
|
At week 8, both groups demonstrated reduced muscle-stiffness/spasticity (Table VI), with a statistical significant difference between the groups in favour of BoNT-A. Using 30% improvement from baseline as clinically relevant, improvement for VAS-muscle-stiffness/spasticity was observed for 16 subjects in the treatment group and 9 in the placebo group (RR=1.83 (95% CI = 0.95–3.53). At week 16 there was no significant difference between the groups (data not shown).
Improvement on the Global Scale (“better”) was reported for 19 respondents in the BoNT-A group vs 9 in the placebo group (Table VI). At week 16, no significant differences between the groups were found (data not shown).
Adverse events
There were 16 mild intervention-related adverse events, such as leg pain at the injection site(s), headache, nausea, fatigue, increased muscle-stiffness/spasticity and reduced balance, reported non-systematically distributed among 9 participants receiving BoNT-A, and 7 receiving placebo. All events were self-limiting and none required discontinuation from the study. No falls were reported. Two participants reported “worse” on the Global Scale at week 8 with “generalized pain” (placebo) and “unstable ankle” (BoNT-A) as stated reasons.
DISCUSSION
To our knowledge, this is the first RCT on the effects of BoNT-A in ambulant adults with spastic CP. We evaluated the short-term effects of BoNT-A treatment in the lower limbs, using kinematics and SF-36 as primary outcomes, and measures on mobility and self-reported muscle-stiffness/spasticity and global treatment effect as secondary outcomes. No statistically significant or clinically relevant differences were found between BoNT-A and placebo for joint angles and HRQOL. However, we found significant differences in favour of the BoNT-A group for VAS-muscle-stiffness/spasticity and the Global Scale. Furthermore, the treatment with BoNT-A was safe with no serious adverse events.
The optimal pharmacological effect of BoNT-A, and time to adapt to the potential effects on mobility and HRQOL, were decisive for examination of effects at week 8 post-treatment. Data from week 16 were used to investigate the duration of potential effects. As the difference between the intention to treat and per-protocol population was very small (Fig. 1), we decided per protocol analysis for week 8 results and the last results carried forward for week 16 results. As there is little knowledge about effective rehabilitation programmes for ambulant adults with CP (3, 14), we were concerned about introducing a treatment programme that could potentially be exhaustive and/or provoke pain (12, 13). Therefore we chose to evaluate the effects of BoNT-A alone, and not the effects of a multi-modal treatment programme, including specific physiotherapy, serial casting and orthotics, which is usual clinical practice based on consensus among the experts (2–4, 8, 9). Consequently, the present study was a compromise in terms of scientific and clinical issues.
We did not find any clinically relevant effects of BoNT-A on predefined kinematic variables from 3DGA. However, the trend of positive results in the BoNT-A group for knee extension at initial contact (Table V) demonstrates that the treatment may be effective. Comparable studies have found improved joint angles during gait in children with CP with spastic equines or flexed knee gait and stiff-knee gait due to stroke (10, 13, 38, 39). A recent RCT on post-stroke ankle spasticity demonstrated results similar to those of our study with small effects on gait strategies (11). Our results probably reflect that gait strategies in adults with spastic CP are relatively fixed with co-existing other impairments, such as contractures, muscle weakness and reduced motor control and balance. As such, reducing muscle tone alone is not enough to change joint angles during gait (3, 9, 11, 13).
On SF-36, both groups had statistically significant improvements in bodily pain at week 8, indicating a placebo effect. However, it is possible that the pain relief in the control group may have occurred due to the injection procedure and/or irritation due to the injected substance in spite of no active substance being present (40). Thus, our results may not have been a pure placebo effect. With regard to the pain-reliving effect of BoNT-A, there is increasing evidence that BoNT-A not only interferes with the release of acetylcholine at neuromuscular junctions, but also interferes with the release of excitatory neurotransmitters associated with chronic pain (41). As such, potential effects could be both reduced hypertonicity and modulation of effects involved in chronic myofascial pain. The placebo group demonstrated a trend of positive changes for the physical domains at week 8. However, this was not supported by significant differences between the groups regarding the number of participants undergoing a change from baseline to a degree defined as clinically relevant (Fig. 2). An explanation could be the recognized paradoxical scoring of scales on physical function in populations with chronic physical disabilities (42). In contrast explorative analysis on MCIDs revealed a significant difference in favour of BoNT-A on mental health and social function. Others have demonstrated similar results on the psychological dimensions, and explained this as an indirect effect of an increase in the ability to perform and enjoy everyday activities (17), whereas another study on stroke patients did not find any improvement in these domains of SF-36 (10). Thus, our results for mental health and social functioning in favour of BoNT-A should be interpreted with caution.
At the group level, the TUG showed a significant improvement in the BoNT-A group, but not in the placebo group. This might indicate that subgroups experienced a positive effect of BoNT-A on muscular hyperactivity, where a potential mechanism could be reduced co-activation patterns (7, 18, 33, 39).
In contrast to the limited evidence about BoNT-A effects on functional improvements, there is evidence about its effects on muscle tone (8–11, 38). Prior to all the injections the presence of abnormal muscle activity was confirmed by a positive response on auditive EMG (11, 15, 16, 25). Thus, our results on VAS muscle-stiffness/spasticity indicate that adults with spastic CP have impairment due to stretch-sensitive muscle overactivity that can improve with BoNT-A. This is comparable with studies on patients with MS, which have demonstrated similar results for the perceived severity of muscle-stiffness/spasticity when carrying out interventions targeted towards muscle overactivity (12, 36). Potential positive effects of BoNT-A are also confirmed by the Global Scale scores, which demonstrated a 59% treatment effect and 27% placebo effect. In contrast, there was no difference between treatment and placebo on self-reported lower limb goal attainment in two recent RCTs on post-stroke patients and children with spastic CP (11, 38). This might reflect that our study population had other primary complaints, and thus reported effects on the impairment from lower limb muscle-stiffness and pain more than on functional ability, such as gait strategy (15, 16). We recognize that these subjective outcomes may be influenced not only by change in symptoms and impairments due to muscle overactivity, but by expectations and other personal factors. To avoid the effects of methodological error, we conducted a randomized placebo-controlled study with strict attention to procedures for randomization and blinding.
Due to the advertisement procedure, we recruited more women than men. However, we assume the study population to be fairly representative of high-functioning community-living ambulant adults with spastic CP (22). The high attendance at physiotherapy and/or physical training programmes is probably explained by the inclusion criteria of “decreased walking ability” and a previous history of regular physiotherapy during childhood.
This study has several limitations. The power calculation was based on assumptions of kinematic SDs in previous publications on BoNT-A intervention in children with CP. In this study, the SDs for the chosen kinematics were all within the range of the SD used when estimating sample size. However, SF-36 demonstrated relatively wide CIs, and therefore results for the SF-36 subscales may be inconclusive. Our results for the primary outcomes may indicate a lack of instrument responsiveness (9), but alternatively may reflect the fact that no true change in the underlying “health construct” and kinematics has occurred. Efforts were made to choose appropriate outcomes covering the dimensions of the ICF (2, 9, 10, 19). 3DGA, which is regarded as the gold standard for measuring components important for gait in the dimension of body structure/functions (2–4, 24), was chosen to evaluate joint angles during gait by the use of kinematics. We should have considered the heterogeneity produced by the individualized intervention on different gait strategies when choosing strict kinematic gait events as primary outcomes. When we planned the trial we did not find any better outcome than the SF-36 version 1.2 for evaluating BoNT-A treatment on health status and participation (28–31). Although reported to be reliable and valid in a wide range of patient groups, no studies have investigated its use in CP. The subscales on role functioning and social functioning, demonstrated ceiling effects affecting their longitudinal validity (Fig. 2). We also revealed paradox scoring for physical function for several of the participants. Due to these limitations, we found it inappropriate to use the two component scores derived from the 8 scales. Fortunately, the distribution properties, as well as the results of VAS muscle-stiffness/spasticity, TUG and 6MWT in our study may indicate their usefulness as outcome measures in similar studies. Considering the intervention, the VAS might have been a more adequate primary outcome measure in this study.
BoNT-A injections at doses up to 400 U were safe and well-tolerated. Due to the decisions from regulatory authorities, m. iliopsoas was excluded as a target muscle, and the maximum dose injected was restricted to 400 U. This study was designed for one intervention with injections(s) alone and with week 8 as the primary endpoint. Consequently, it was not well fitted to resemble clinical practice, which can be viewed as a concern about the ecological validity of the study. These issues may have resulted in suboptimal effects for several of the participants (2, 4, 8, 13).
In summary, when doing injections only into several spastic muscles in adults with spastic CP and different gait strategies, this study indicates no short-term effects of BoNT-A treatment vs placebo for the primary outcomes. However, the secondary outcomes showed positive effects in favour of the BoNT-A group for self-reported muscle-stiffness/spasticity and global therapy effects, and the treatment was well-tolerated. Thus, BoNT-A may have a role in clinical management programmes for selected adults with spastic CP. Further studies with specific post-injection rehabilitation and a longer study period are warranted.
AcknowledgEments
The project was supported by the East Regional Health Administration and Sunnaas Rehabilitation Hospital (grant number 206 24 503). We are grateful to all the adults with CP who participated. We acknowledge the contribution of the Norwegian CP Association, the Sunnaas Rehabilitation Hospital, especially physiotherapist Kerstin L. Larsen, pharmacist Anette Storhaug, nurses Marit Tobiassen and Ingeborg Linnerud, and the staff at Sunnaas Gait Laboratory for their important work during the data collection phase.
Conflict of interest: Allergan Inc. produces Botox® and has an interest in demonstrating benefits of this drug. None of the authors has a financial relationship with Allergan in the form of honoraria, sponsorship to attend meetings and/or consultancy fees, or any personal financial interest in Botox®, or in any of the methods used in this research.
References