OBJECTIVE: To assess the need for intervention on cardiovascular disease risks in persons with paraplegia according to: (i) two multifactorial risk models; and (ii) these models in combination with the additional risk of overweight/obesity.
DESIGN: Cross-sectional.
SUBJECTS: A total of 134 out of 153 persons, comprising more than 80% of a regional prevalence population with traumatic
paraplegia (American Spinal Injury Association Impairment Scale A–C) of minimum one year duration.
METHODS: Participants were screened for cardiovascular
disease risk using two multifactorial risk models: the Systematic Coronary Risk Evaluation and the Framingham Risk Equation. Risk factors included were: age, gender, systolic blood pressure, antihypertensive medication, smoking, total cholesterol, high-density lipoprotein cholesterol, and total cholesterol/total cholesterol ratio. In addition, overweight/obesity was assessed by body mass index.
RESULTS: Twenty-seven percent to 36% of the cohort was
eligible for cardiovascular disease risk intervention, depending on the risk model used. When overweight/obesity (spinal cord injury adjusted cut-score body mass index ≥ 22) was also considered, over 80% of the participants qualified for intervention.
CONCLUSION: Almost one-third of persons with paraplegia were eligible for cardiovascular disease risk intervention according to authoritative assessment tools. The number in need of intervention was dramatically increased when overweight/obesity as a cardiovascular disease risk was
considered.
Key words: cardiovascular disease; risk scores; ageing; paraplegia; overweight; obesity; spinal cord injury.
J Rehabil Med 2011; 43: 237–242
Correspondence address: Kerstin Wahman, Department of Neurobiology, Care Sciences, and Society, Division of Neurorehabilitation, Karolinska Institutet, SE-141 83 Huddinge, Sweden. E-mail: kerstin.wahman@rehabstation.se
Submitted February 18, 2010; accepted October 29, 2010
Introduction
After decades of work focused on improving protocols for acute care and primary rehabilitation of persons with spinal cord injury (SCI), attention is now increasingly directed toward issues of ageing. Increased lifespan has, by default, increased the risk for all-cause cardiovascular disease (CVD) in the SCI population (1). Notwithstanding these reports, it is unclear whether the SCI population has a uniquely increased risk for CVD or whether they have now been “normalized” to the CVD risk of the general population. A meta-analysis published in 2008 supports the latter belief (2). Other researchers suggest truly increased risks of CVD and increased prevalence of CVD risk factors after SCI (3–5) and earlier onset of CVD following SCI (6).
A high prevalence of individual CVD risk factors has been demonstrated in persons with SCI. The most common of these are dyslipidaemia with decreased levels of high-density lipoprotein cholesterol (HDL-C) (7, 8), impaired fasting glucose (IFG), and increased prevalence of diabetes mellitus (DM) (9), and hypertension has been reported to be increased in persons with paraplegia, but not in those with tetraplegia (10). Furthermore, abdominal obesity, increased whole body fat, and decreased lean body mass have been reported (11). Recently, inflammatory markers correlating with an increased CVD risk have been reported to be more prevalent in persons with SCI (8). In addition, detrimental lifestyle factors, such as physical inactivity (12), a diet rich in total and saturated fat (13), tobacco use (14), and increased social stress (15), aggravate the CVD risk profile, as does frequent clustering of risk factors (16). CVD risk factors are known to act synergistically, thus clustering will disproportionately increase the overall CVD hazard.
Multifactorial risk estimation models are commonly used to predict CVD risk in the general population and in patient groups (17, 18). However, clinicians should also consider population-specific factors, which may not be incorporated in the models. Examples of such factors include physical deconditioning due to immobilization, genetic risk factors for CVD, overweight/obesity, and population-specific dyslipidaemias (17). Such additional factors may increase the true risk of future disease.
To date, an SCI-specific CVD prediction model has yet to be developed. Thus, studies on SCI populations use models validated on the general population. Different models for estimation of total CVD risk in persons with SCI have yielded conflicting results. When using a CVD risk score that included diastolic blood pressure (DBP), total cholesterol (TC), number of cigarettes smoked per day, and gender, Krum et al. (19) reported a “comparable” risk in disabled and non-disabled persons. When using the Framingham Risk Equation (FRE), Cardus et al. (20) observed that an SCI cohort had a risk “similar” to that of deconditioned persons without disability. The so-called cardiometabolic syndrome, which encompasses a cluster of risk factors, has both been reported as being more prevalent among persons with SCI (21, 8) and as being as common in the general population (22).
In order to further clarify whether persons with SCI should be considered a high-risk CVD group, we have conducted a series of studies examining a regional population of persons with paraplegia. In one study, we found a high prevalence of individual CVD risk factors, including dyslipidaemia, hypertension, and overweight/obesity and frequent clustering of CVD risk factors (16). In a further analysis, we reported a significantly increased risk of myocardial infarction, hypertension, dyslipidaemia, and DM in the paraplegia group, compared with a reference population of persons living in the same region (23).
The aim of the present study was to assess in persons with chronic “wheelchair-dependent” paraplegia the need for intervention (i.e. therapeutic lifestyle intervention and/or medication) on CVD risks according to the Systematic Coronary Risk Evaluation (SCORE), FRE, and by assessment of body mass index (BMI) as an indicator of overweight/obesity.
Materials and Methods
Participants
Wheelchair-dependent men and women aged 18 years and older with traumatic paraplegia (American Spinal Injury Association Impairment Scale (AIS) grade A, B or C) for at least one year were included. ”Wheelchair-dependency” was operationally defined for the purpose of this study as ”exclusive or predominant ambulation by wheelchair” (as opposed to walking). All individuals were living in the greater Stockholm area and were enrolled in the Spinalis SCI outpatient programme, which provides follow-up for approximately 95% of the regional SCI population. A total of 153 persons fulfilling inclusion criteria were asked to participate in the study as they consecutively became due for annual check-ups at the centre. Sixteen subjects did not attend due to hospitalization or illness at home during this time or due to our inability to establish contact by letter or telephone. From the 153 enrolled patients, 137 participated in a routine medical check-up. From this group, 135 persons (104 males and 31 females) agreed to participate in this study in accordance with approval granted by the human ethics committee at the Karolinska Institute, Stockholm. One subject was excluded due to lack of blood tests. The final study sample represented over 80% of the total regional population of persons with traumatic paraplegia.
Participant interview and examination
Participant information was collected by individually-administered structured interviews. The AIS was determined by a specialist physician according to standards. After 30 min of rest, blood pressure was recorded on the left arm by a sphygmomanometer. Body weight (kg) was measured on a calibrated scale, while body height was self-reported. Blood samples were taken under fasting conditions to determine glucose concentrations and a lipid profile including TC and HDL-C. The ratio of TC/HDL-C was calculated. DM/IFG was defined as increased fasting glucose levels ≥ 6.1 mmol/l (110 mg/dl) and/or ongoing medication for DM.
Multifactorial risk models
Two multifactorial risk models were used to rate the need for intervention: the SCORE (17) and the FRE (18).
Systemic Coronary Risk Evaluation. SCORE was developed from a dataset of 12 European countries incorporating risk analysis in 205,178 persons and 7,934 cardiovascular-related deaths. SCORE comprises both paper-based risk charts and a web-based application (“Heart Score”). SCORE was used to assess the absolute 10-year probability for experiencing a fatal cardiovascular event (17). SCORE includes twoprotocols: one based on TC and the other based on TC/HDL-C ratio. Risk factors included in SCORE are: age, gender, systolic blood pressure (SBP), smoking, and either TC or TC/HDL-C ratio.
Risk dichotomization according to SCORE is: < 5% is classified as “low” risk, while ≥ 5% is classified as an “increased” risk, the latter with a need for therapeutic lifestyle changes (TLC) and/or medication. Persons with CVD and/or DM are defaulted to the high-risk group according to the assessment protocol. Heart Score was used when calculating risk with TC as the lipid risk criterion, whereas SCORE paper-based charts were used when including the TC/HDL-C ratio. Assessments were conducted for participants within the recommended SCORE age range of 40–65 years and for the total study group.
Framingham Risk Equation. The FRE is based on the Framingham study, which started in 1948 with 5,209 adults included from the town Framingham, Massachusetts, USA. The study is ongoing and now includes the third generation (4,095 participants), i.e. the children of the second-generation (5,124 persons).
FRE was used to calculate 10-year risk for so called “hard” coronary heart disease (i.e. myocardial infarction and/or coronary death) (18). Risk factors included are: age, gender, TC, HDL-C, smoking, SBP, and use of blood pressure (BP) medication.
Criteria for risk classification in FRE are: < 10% classified as “low” risk requiring no treatment; 10–20% classified as “intermediate” risk, indicating need for TLC; and > 20% classified as “high” risk, indicating need for both TLC and medication. According to the protocol, persons with manifest CVD and/or DM are placed in the high-risk group by default.
Body Mass Index
BMI was used to assess overweight/obesity. It is computed as body weight (in kg) divided by the square of body height (in m). The SCI population has no specific definition of overweight/obesity. Therefore, 3 alternative BMI criterion scores were used to define overweight: ≥ 25, which is used in the general population (22), and ≥ 23 and ≥ 22, which both have been suggested for use in the SCI population (24, 25).
Need for CVD intervention based on CVD risk models and BMI. In this analysis, the risk factor overweight/obesity was added to the results from the two risk models. Participants with low CVD risk according to SCORE and FRE were thus stratified by the following BMI categories, i.e. < 22 and ≥ 22; < 23 and ≥ 23; and < 25 and ≥ 25.
Data analysis
Data were analysed using SPSS 15.0 for Windows software. Frequency and descriptive statistics were calculated. The total CVD risk based on results from the SCORE and FRE models and age, gender, time since injury, level of injury (T1–T6, T7–T12, and L1–L4), and BMI were analysed. Age, gender (reference category: female coded “1”), and two levels of injury (reference category: T1–T6 coded as “1” and T7–T12 coded as “1”) were utilized as independent variables for each regression equation for the FRE and SCORE dependent variables. With an independent variable (level of injury) that has 3 levels, only two dummy variables are needed (n levels minus 1 dummy variable), as the default regression equation would be the case where the two dummy variables were equal to zero. In this case, L1–L4 is the default case, since in any situation it will be equal to “0” in the equation. The entry method for the 4 independent variables was utilized to assess the relative importance of each variable in the equation. Time since injury was not used as a predictor in the regression, due to its high correlation with age (r = 0.48, p < 0.01) and thus multicolinearity in the model. The criterion for statistical significance was set at α < 0.05.
Final linear regression model with Systemic Coronary Risk Evaluation as the dependent variable. Table III depicts the unstandardized coefficient (B) and its standard error, the standardized coefficient (β), and the t statistic and significance level of the unstandardized coefficient. The overall model was statistically significant (F = 49.5 [4,129], p < 0.001) and explained 59% of the variance (adjusted R2) in the dependent variable.
Final linear regression model with Framingham Risk Equation as the dependent variable. Table IV depicts the unstandardized coefficient (B) and its standard error, the standardized coefficient (β), and the t statistic and significance level of the unstandardized coefficient. The overall model was significant (F = 29.6 [4,129], p < 0.001) and explained 46% of the variance (adjusted R2) in the dependent variable.
Results
Core clinical characteristics of the study group are summarized in Table I. Nearly 80% of the study population were men, approximately 40% had an injury level at T7–T12, and 80% were classified as AIS-A. The mean age was 48 years and duration of injury was 18.5 years. Table II shows the descriptive statistics of the outcome variables and the two risk equations.
| Table I. Characteristics of the study sample; n = 134 |
| Patients, n (%) |
| Male | 104 (78) | |
| Female | 30 (22) | |
| Age, years, mean (SD) [range] | 47.8 (13.8) [18–79] |
| Years of injury, mean (SD) [range] | 18.5 (12.4) [1–48] |
| Level of injury, n (%) |
| T1–T6 | 44 (33) | |
| T7–T12 | 66 (49) | |
| L1–L4 | 24 (18) | |
| AIS – level, n (%) | | |
| A | 108 (80) | |
| B | 13 (10) | |
| C | 13 (10) | |
| SD: standard deviation; AIS: American Spinal Injury Association Impairment Scale. |
| Table II. Descriptive statistics of the variables included in the study |
| | n | Mean (SD) | Min–Max |
| Glucose (mg/dl) | 133a | 93.44 (24.81) | 57.60–250.20 |
| Total cholesterol | 134a | 187.23 (38.14) | 98–304 |
| LDL cholesterol | 133a | 118.44 (34.41) | 39–211 |
| HDL cholesterol | 134a | 45.43 (13.93) | 12–117 |
| Total cholesterol: HDL cholesterol ratio | 134a | 4.51 (1.84) | 1.77–17.00 |
| BMI | 135 | 24.38 (3.98) | 15.50–36.50 |
| Framingham score | 134a | 8.30 (9.61) | 0.9–30.1 |
| SCORE | 134a | 1.95 (2.62) | 0–12 |
| aIndicates missing data due to errors of blood sample analysis. SD: standard deviation; LDL: low-density lipoprotein; HDL: high-density lipoprotein; BMI: body mass index; SCORE: Systematic Coronary Risk Evaluation. |
Systematic Coronary Risk Evaluation
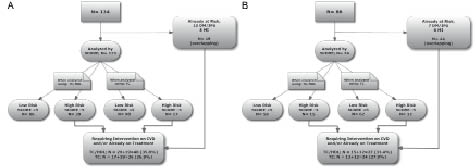
The need for intervention for the study population as a whole according to SCORE, analysed both by TC and TC/HDL-C ratio, respectively, is shown in Fig. 1. Approximately 15% of the subjects were directly classified as at-risk according to European guidelines on CVD and were thus defaulted to the high-risk group. Of the remaining 115 subjects assessed by SCORE (TC), 15% received a high-risk classification. Thus, slightly less than 30% required intervention according to protocol when using TC. When analysis was restricted to those within the standard age range (40–65 years), approximately 30% required intervention according to the TC protocol. When the TC/HDL-C ratio was used, more than 35% had an increased CVD risk requiring intervention according to protocol. When the analysis was restricted to the standard age range of 40–65 years, more than 30% were classified as requiring intervention according to the TC/HDL-C ratio protocol (Fig. 1B).
Fig. 1. Study participants ((A) ages 18–79 years and (B) ages 40–65) using the Systematic Coronary Risk Evaluation risk model. DM: diabetes mellius; IFG: impaired fasting glucose; TC: total colesterol; HDL: high density lipoprotein; CVD: cardiovascular disease; MI: Myocardial infarction.
Framingham Risk Equation
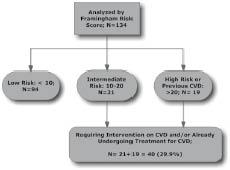
In accordance with FRE protocol guidelines, which in this case are similar to those of SCORE, approximately 15% with established CVD and/or IFG/DM defaulted into the high-risk group. An additional 15% were identified as having intermediate risk (10–20%).
Thus, according to FRE, approximately 30% were classified as requiring intervention (Fig. 2).
Fig. 2. Study participants (ages 18–79 years) using the Framingham risk model. CVD: cardiovascular disease,
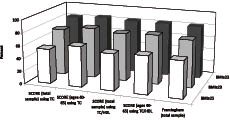
Overweight/obesity in combination with SCORE and FRE. In Fig. 3, we report the total number of subjects who qualified for intervention based on the 2 risk models and the 3 selected BMI levels. When using BMI level ≥ 22 kg/m2 as a criterion for overweight, 82–85% of the sample required intervention, depending on the prediction model. At BMI levels ≥ 23 and ≥ 25 kg/m2, 73–79% and 54–63% of study participants, respectively, qualified for treatment intervention, again depending on the prediction model.
Fig. 3. Participants qualifying for intervention after incorporating multi-level body mass index (BMI) risks in those assessed at low risk. SCORE: Systemic Coronary Risk Evaluation; TC: total colesterol; HDL: high-density lipoprotein.
Final linear regression model with SCORE and FRE as the dependent variable. Increasing age (the strongest predictor, β = 0.74) was associated with a higher SCORE risk, whereas being female and having a lesion level of T1–T6 or of T7–T12 was associated with a lower SCORE risk (Table III). Increasing age (the strongest predictor, β = 0.64) was associated with a higher FRE, whereas being female and having an injury at T1–T6 was associated with a lower FRE. The T7–T12 level of injury was a non-significant predictor in the equation (Table IV).
| Table III. Linear regression model for significant independent variables with the Systemic Coronary Risk Evaluation value as the dependent variable in persons with paraplegia |
| | Unstandardized coefficients | Standardized coefficients |
| B | SE | Beta | t | p-value |
| Age | 0.14 | 0.01 | 0.74 | 13.39 | < 0.001 |
| Gender, female reference | –1.32 | 0.35 | –0.21 | –3.79 | < 0.001 |
| Level of injury,
T1–T6 reference | –1.43 | 0.43 | –0.26 | –3.34 | 0.001 |
| Level of injury, T7–T12 reference | –1.12 | 0.40 | –0.22 | –2.78 | 0.006 |
| F (4,129) = 49.5, p <0.001, adjusted R2 = 0.59. SE: standard error. |
| Table IV. Linear regression model for significant independent variables with the Framingham score as the dependent variable in persons with paraplegia |
| | Unstandardized coefficients | Standardized coefficients |
| B | SE | Beta | t | p-value |
| Age | 0.45 | 0.045 | 0.640 | 10.03 | < 0.001 |
| Gender, female reference | –4.88 | 1.47 | –0.21 | –3.31 | 0.001 |
| Level of injury, T1–T6 reference | –4.86 | 1.80 | –0.24 | –2.70 | 0.008 |
| Level of injury, T7–T12 reference | –1.75 | 1.70 | –0.09 | –1.03 | 0.307 |
| F (4,129) = 29.6, p < 0.001, adjusted R2 = 0.46. SE: standard error. |
Discussion
The key finding of this study is that according to the protocols for SCORE and FRE approximately one-third of the study population required intervention for CVD. When subjects were classified into the low-risk groups according to SCORE and FRE, respectively, and were additionally assessed at BMI ≥ 22, the number of subjects in need of intervention increased to 82–85%. As expected, risk increased with age and male gender. A thoracic lesion level correlated with a lower risk in the SCORE model, while for FRE this held true for T1–T6.
SCORE was included in our risk assessment strategy, as it is recommended by European guidelines on CVD prevention (17) and it is based on European population data. FRE was included in our analysis since it has been used in most CVD risk assessment studies (18). Furthermore, FRE has been used previously in risk assessment for SCI populations, while SCORE has not. When assessing CVD risk in an all-level, all-degree-of-completeness SCI population, many factors may influence the result, such as varying degrees of immobilization and autonomous system impairment. In order to increase homogeneity, the category of “wheelchair-dependent” paraplegia was selected. It is possible that the results of this study may not be representative for other SCI subcategories.
Finnie et al. (26) found a CVD risk profile similar to that found in the present study by applying FRE in a SCI population in Canada. They argued that FRE might in fact underestimate the CVD risk in this patient group and suggested the addition of inflammatory risk markers in the assessment model. Furthermore, Demirel et al. (5) used factors identified in the guidelines of the National Cholesterol Education Project Adult Treatment Panel II (NCEP-ATP) to assess risk in a Turkish population of 45 people with paraplegia and 24 with tetraplegia. Compared with controls, DM, elevated TC and low-density lipoprotein (LDL-C), and low HDL-C were significantly more prevalent in persons with SCI, although lipid profiles were unrelated to neurological level and completeness of lesions. Using the updated NCEP Guidelines (ATP III), Nash & Mendez (8) analysed a young, healthy, and predominantly non-smoking cohort of persons with paraplegia and found that 63.4% were in need of intervention.
It has been reported that, when using SCORE on a Swedish population, it overestimates the number of CVD-related deaths (27). This may be a consequence of the fact that CVD risk models overestimate risk in populations with a decreasing CVD mortality and the opposite if CVD mortality is increasing (17). In one of our previous articles, we reported that our population with wheelchair-dependent paraplegia had an increased risk of myocardial infarction compared with the general population (22). That may indicate that, even if SCORE overestimates CVD in the general population, it may be correct or even underestimate the risk in the SCI subpopulation. In order to develop SCI-specific CVD risk equations, large epidemiological mortality studies are needed, which will require long-term multi-centre studies.
To gain a better understanding of the clinical need for intervention on CVD, overweight/obesity was also added to the SCORE and the FRE analyses. Participants with low CVD risk according to SCORE and FRE were stratified by the following BMI categories, i.e. < 22 and ≥ 22; < 23 and ≥ 23; and < 25 and ≥ 25. Inclusion of additional risk factors is a common practice, when their contribution appears obvious and they improve prognostic strength. However, selection of a criterion for overweight/obesity in those with SCI poses a challenge and may be considered a study limitation. The traditional cut-off for the general population at BMI > 25 fails to identify most obese persons with SCI (28). In fact, at a matched BMI, persons with SCI have significantly more body fat than persons without SCI. Due to lack of consensus in defining “normal” BMI values for persons with SCI, we used 3 different BMI levels. It has been suggested that “healthy” BMI for persons with SCI is below 23 (24) or 22 kg/m2 (25). Even when using a more conservative BMI criterion of 23 kg/m2 as overweight, nearly 75% of persons studied were classified at a risk stratum that qualified for intervention. These findings are consistent with previous studies reporting a high prevalence of overweight/obesity in the SCI population (29).
This paper adds to earlier studies suggesting a heightened risk for all-cause CVD in the population of persons with SCI. This finding suggests a need for diligent surveillance of the population, although methods vary as to how this might be achieved. To address this issue, our early assessment has included a wide range of outcomes for determination of all-cause risk. While the most often-cited lipid risk for persons with SCI involves low levels of the cardioprotective HDL-C, the need for intervention in guideline-driven therapeutic algorithms is ultimately based on LDL-C, not HDL-C, satisfying its target. This explains our investigation of LDL-C as a health risk. As the interplay of lipids and lipoproteins is often instructive of more global risks than isolated lipoprotein cholesterols we have included in the analysis the TC, HDL ratio, which has power for future heart disease that approaches the predictive power of the Framingham model. “Prevalence” doesn’t determine need for intervention, but the variable of LDL-C satisfying a target level for each individual does.
Some limitations of this study should be mentioned. First, it has been suggested that the FRE model overestimates hazards sustained by European populations. As CVD incidence and mortality can diverge between populations, this would appear to disqualify FRE as the comparison method in our study. Conversely, an editorial exploring this limitation concluded, “with some caution, relative hazard estimates appear portable between populations” (30). Secondly, the all-cause risks included in hazard estimation models have not been derived from study of persons with SCI and may differ from those of the non-disabled population or contribute with different strength to disease outcomes. However, Wilt et al. (2) stated in a meta-analysis that “patients with SCI should be assessed and treated according to existing guidelines for able-bodied individuals”. Thirdly, the operationalized definition of DM was based both on self-report (8 subjects) and on actual blood testing (5 subjects). True, DM may thus have been slightly overestimated, incorrectly allocating persons to the high-risk groups by a maximum of 3%. Fourthly, the total need for CVD intervention might also have included level of physical activity and fitness, something which was not the case in this study. Fifthly, BMI ≥ 25 as an indicator for overweight and obesity in the SCI population has been questioned because of the consequences with muscular atrophy after SCI. New cut-off points have been suggested, and these have also been used in this article (24, 25). However, little is known about this topic, and the true value of BMI related to level of injury that predicts CVD in persons with SCI has not yet been determined. Waist circumference is another indicator for overweight/obesity that might be more valid in this group (31). One of the problems with using waist circumference in persons with SCI is that paralysed abdominal muscles might affect the results. Additional studies are needed to elucidate which indicator of overweight/obesity is more valid in this population.
In conclusion, this study showed that one-third of persons with wheelchair-dependent paraplegia due to traumatic SCI were eligible for CVD risk intervention. When overweight/obesity was included in the risk assessment, over 80% of participants were eligible for intervention. Unacceptably high levels of risk observed in this study indicate a need for routine clinical assessment of CVD hazards in persons with SCI. This will require population-sensitive screening tools. As the study data also indicate a pattern of multiple risks, evidence-based multi-factorial health promotion programmes that address these risks must be developed and implemented.
AcknowledgEments
This study was supported by the Norrbacka-Eugenia Foundation, Cancer and Traffic Injury Fund, Stockholm County Council, The Spinalis Foundation, The Swedish Association for Persons with Neurological Disabilities, and Praktikertjänst (Research and Development Board).
No commercial party having a direct financial interest in the results of the research supporting this article has or will confer a benefit upon the author(s) or upon any organization with which the author(s) is/are associated.
References