OBJECTIVE: The aims of this study were to examine: (i) baseline pressure pain thresholds in patients with chronic fatigue syndrome and those with chronic low back pain compared with healthy subjects; (ii) the change in mean pain threshold in response to exercise; and (iii) associations with exercise-induced increase in nitric oxide.
PARTICIPANTS: Twenty-six patients with chronic fatigue syndrome suffering of chronic pain, 21 patients with chronic low back pain and 31 healthy subjects.
METHODS: Participants underwent a submaximal aerobic exercise protocol on a bicycle ergometer, preceded and followed by venous blood sampling (nitric oxide) and algometry (hand, arm, calf, low back).
RESULTS: Patients with chronic fatigue syndrome presented overall lower pain thresholds compared with healthy subjects and patients with chronic low back pain (p < 0.05). No significant differences were found between healthy subjects and patients with chronic low back pain. After submaximal aerobic exercise, mean pain thresholds decreased in patients with chronic fatigue syndrome, and increased in the others (p < 0.01). At baseline, nitric oxide levels were significantly higher in the chronic low back pain group. After controlling for body mass index, no significant differences were seen between the groups at baseline or in response to exercise. Nitric oxide was not related to pain thresholds in either group.
CONCLUSION: The results suggest hyperalgesia and abnormal central pain processing during submaximal aerobic exercise in chronic fatigue syndrome, but not in chronic low back pain. Nitric oxide appeared to be unrelated to pain processing.
Key words: exercise; pain threshold; nitric oxide; chronic pain; central sensitization; pain inhibition.
J Rehabil Med 2010; 42: 884–890
Correspondence address: Jo Nijs, Artesis Hogeschool Antwerpen (AHA), Department of Health Sciences, Division of Musculoskeletal Physiotherapy, Van Aertselaerstraat 31, BE-2170 Merksem, Belgium. E-mail: jo.nijs@vub.ac.be
Submitted June 1, 2009; accepted June 3, 2010
INTRODUCTION
While there is a body of literature on chronic pain, few studies have made direct comparisons between different chronic pain conditions. Comparing pain disorders is crucial for unravelling the differences and similarities in the nature of chronic pain. Chronic low back pain (CLBP) is a well-known example of chronic pain. We are less familiar with chronic pain experienced by patients with chronic fatigue syndrome (CFS). Although the majority of patients with CFS experience chronic widespread musculoskeletal pain, evidence on the nature of this pain is lacking (1).
Central sensitization has been documented as an important mechanism in several chronic pain populations, including fibromyalgia, whiplash-associated disorder, and osteoarthritis (2, 3). In patients with CLBP, augmented pain sensitivity and cortical reorganization have been described, suggesting abnormal central pain processing (4, 5). Generalized hyperalgesia at locations in the lower back (6, 7) and at sites unrelated to the lower back (7) have been reported. Central pain mechanisms may contribute to recurrent pain in patients with CLBP.
In CFS the central sensitization hypothesis has been suggested (8). Central mechanisms may be responsible for the deregulated anti-nociception. Whitheside et al. (9) reported a decrease in pressure pain threshold (PPT) in response to exercise in 5 patients with CFS. In healthy individuals PPTs increase following different types of exercise. Endogenous opioid release, however, with mixed support (10), or other spina/supraspinal nociceptive inhibitory mechanisms (11) have been reported to be involved in the increased pain thresholds following exercise. Increased nociceptive perception after exercise may be suggestive of a deregulated anti-nociceptive mechanism.
The latter may partly explain the typical exacerbation of symptoms (including pain) following vigorous physical activity in patients with CFS (12), a primary characteristic that is evident in up to 95% of patients with CFS (13). This is not present in other disorders where fatigue is a predominant symptom, such as depression, rheumatoid arthritis, or multiple sclerosis (14). Post-exertional malaise is one of the best predictors of the differential diagnosis of CFS and major depressive disorder (15).
Nitric oxide (NO) plays a complex role in nociceptive processing (11). Although evidence exists regarding the beneficial effects of the release of small amounts of NO during inhibition of nociceptive pathways (16), excessive amounts of NO, could contribute to central sensitization in CFS (8). Excessive amounts of NO have been documented in patients with CFS (17). NO is able to reduce the nociceptive inhibitory activity of the central nervous system, leading to central sensitization of dorsal horn neurones (18), which could explain chronic widespread pain in patients with CFS (8).
A single bout of physical activity triggers release of NO (19) and could consequently further raise elevated NO levels in patients with CFS. The above-mentioned hypothesis on NO could account for the increase in pain following vigorous exercise in patients with CFS. Besides the evidence in support of abnormal central pain processing in CLBP, nociceptive physiology in relation to exercise/physical activity has not been studied in CLBP. Furthermore, no data addressing levels of NO and exercise are available for patients with CLBP.
The goal of the present study was to examine the change in PPTs and serum NO levels in response to a submaximal aerobic bicycle exercise in patients with CFS with chronic widespread pain, pain-free healthy subjects, and patients with CLBP. Furthermore, the association between NO and PPT changes were studied. It was hypothesized that: (i) baseline PPTs are lower in the patient groups compared with healthy subjects, (ii) PPTs decrease following submaximal aerobic exercise in patients with CFS, and (iii) impaired pain response to exercise is associated with an exercise-induced increase in NO levels.
MATERIALS AND METHODS
Research design
We compared the response to a submaximal exercise test on a bicycle ergometer between patients with CFS with chronic widespread pain, patients with CLBP, and healthy sedentary subjects. First the participants received a leaflet explaining the purpose of the research. Participants were asked to read the information leaflet carefully and to sign the informed consent form to indicate agreement to participate in the study. The hypothesis of the study was not discussed with the patients prior to the completion of the tests. The protocol and the information leaflet were approved by the local ethics committee (University Hospital Vrije Universiteit Brussel; O.G. 016). Demographic data were recorded, including age, height and weight.
All participants underwent algometry and venous blood sampling for NO quantification immediately before and after a submaximal aerobic exercise bicycle test. Changes in NO and mean PPT following the exercise protocol were compared between the 3 groups.
Participants
All participants were contacted by telephone to verify a number of study requirements and to ask whether they were interested in participating in the trial. To fulfil the general inclusion criteria, participants had to be aged between 18 and 65, and had to be able to perform an ergometer bicycle test. Participants were excluded in case of pregnancy or up until one year postnatally, or if they had severe neurological or cardiovascular problems. In order to minimize confounding of the PPTs and NO analyses, all participants were asked to cease the use of analgesics and anti-depressants 24 h prior to study participation, and to avoid any intake of alcohol, caffeine, and nicotine on the day of study participation.
Chronic fatigue syndrome patients. Twenty-six patients with CFS were randomly selected (randomly defined file numbers) from the medical files available at our university-based chronic fatigue clinic. Patients had to be diagnosed with CFS following the Centre for Disease Control and Prevention (CDCP) diagnostic criteria (12) and report Chronic Widespread Pain (CWP) following the criteria of the American College of Rheumatology (20). All patients underwent an extensive medical evaluation by the same physician (specialized in internal medicine) prior to study participation. This evaluation consisted of a standard physical examination, medical history, exercise capacity test and routine laboratory tests. Any active medical condition that may explain the presence of chronic fatigue, prohibits the diagnosis of CFS. The laboratory tests included a complete blood cell count, determination of the erythrocyte sedimentation rate, serum electrolyte panel, measures of renal, hepatic and thyroid function, as well as rheumatic and viral screens. If the patient’s medical history did not exclude a psychiatric problem at the time of onset, then a structured psychiatric interview was performed. In a number of cases further neurological, gynaecological, endocrine, cardiac and/or gastrointestinal evaluations were performed. The medical records were also reviewed to determine whether patients had any organic or psychiatric disorder that could explain their symptoms. If any of the additional analyses revealed any active medical condition that could explain the presence of a patient’s symptoms, this subject was excluded from the sample.
Chronic low back pain-patients. In order to compare patients with CFS with another chronic pain population, 21 patients with CLBP were recruited via pamphlets in the University Hospital Brussels and via physical therapists. We used the criteria for non-specific LBP consistent with those used in Flynn et al.’s study (21). Patients with CLBP had to experience at least 3 months of non-specific and chronic LBP, had to be sedentary (defined as having a sedentary job and performing < 3 h moderate physical activity/week; moderate activity defined as activity demanding at least 3 times the level of energy spent passively (22)), and had to be aged between 18 and 65 years. Patients with specific underlying pathology as the cause of CLBP (e.g. tumour, known disc derangement, trauma, infection, diagnosed inflammatory joint disease, spinal stenosis, spondylolysis or spondylolisthesis) were excluded. In addition, patients with a history of spinal fracture, severe degenerative change, severe scoliosis, osteoporosis, obesity, radicular signs, malignancies, and metabolic or rheumatological diseases, were excluded. Patients with a history of spinal surgery were not included in the study.
Healthy control subjects. A healthy sedentary (see above) control group of 31 subjects was recruited among researcher’s acquaintances and by distributing pamphlets in the University Hospital Brussels, the Vrije Universiteit Brussel and the Artesis University College of Antwerp. Besides the above common criteria they could not suffer any pain complaints or severe systemic or psychiatric problems. This corresponds to activities with Metabolic Equivalents (METs) scores of 3 or more in the compendium of physical activities (23). These criteria were assessed using the demographic questionnaire.
Outcome measures
Nitric oxide assay. Venous blood was collected in heparinized vacuum tubes. NO concentrations in the serum were analysed with a NO Quantitation Kit (Active Motif Inc., Carlsbad, CA, USA). NO measurements were based on total serum nitrite and nitrate levels. Preparation and analysis of the samples was performed following the manufacturer’s protocol. Blood samples were coded and transferred to RED Laboratories (Zellik, Belgium), where the samples were analysed blindly.
Algometry. PPTs were measured with an analogue Fisher algometer (Force Dial model FDK 40 Push Pull Force Gage, Wagner Instruments, Greenwich CT, USA) immediately before and after the exercise bout in the skin web between thumb and index finger (9) (referred to as hand PPT), 5 cm lateral to the spinous process of L3 (6) (referred to as back PPT), at the insertion of the deltoid muscle (referred to as arm PPT), and at the proximal third of the calf (referred to as calf PPT). These sites were chosen in order to test PPTs on non-specific locations both on the extremities and the trunk. The order of PPT testing was randomized by lottery and always performed bilaterally. The force was gradually increased at a rate of 1 kg/s (24), by silently counting seconds while increasing pressure. PPT was defined as the point at which the pressure sensation changed to pain (2). The threshold was determined as the mean of the 2 last values out of 3 consecutive measurements, with a pause of 10 s between the measurements, a procedure found to be reliable in healthy subjects (24, 25). Pressure algometry has been found to be efficient and reliable in the exploration of physiopathological mechanisms involved in pain (26) and is useful for the evaluation of treatment outcome, as reviewed by Fischer (27). Whiteside et al. (9) previously used algometry in the skin between thumb and index to evaluate the difference in PPT before and after an exercise test in patients with CFS. Algometry was always conducted by the same researcher (MM).
Self-reported measurements. Self-report measures included a visual analogue scale (VAS) (28), ranging from 0 (no pain) to 100 (worst imaginable pain) mm to assess current pain intensity, and the Oswestry Disability Questionnaire (ODQ) (29) to evaluate disability in patients with CLBP. High levels of reliability and validity have been described for these questionnaires (28–30). The Short-Form Health Status Survey 36 (SF-36) assesses functional status and well-being and quality of life (31). The SF-36 has been documented to have reliability and validity in a wide variety of patient populations (31) and it appears to be the most frequently used measure in CFS research (32).
Exercise protocol
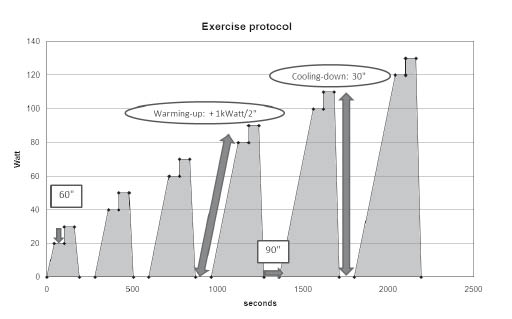
The complete exercise protocol (Fig. 1) consisted of a maximal of 6 bouts of exercise on a bicycle ergometer, each bout followed by 90 s of rest. The exercise programme was incremental, starting at 20 Watt and augmenting in steps of 10 Watt/minute. Each exercise bout was introduced by a short warming-up period to overcome the inertia, starting from zero and gradually increasing by 1 Watt every 2 s. Thereafter, the actual exercise period consisted of 2 incremental 1-min steps. Finally, each bout finished off with a short cooling-down of 30 s to prevent venous pooling. The participants were instructed to stop the test when they were getting tired and they felt that they could no longer reach a pedalling frequency of at least 70 revolutions per min. After the sixth exercise bout (ending at 130 Watt), the programme was completed. Depending on the individual capacity the complete test could be performed or prematurely discontinued. The rationale for this protocol was that everyone would be able to perform a submaximal exercise test by completing at least one or more exercise bouts. The rest periods between bouts were planned in order to permit relative recuperation, so that it would not be peripheral muscle pain or fatigue that caused premature ending of any bout. Intermittent exercise protocols are in line with exercise pathophysiology seen in patients with CFS. It has been demonstrated that a discontinuous graded exercise test did not greatly exacerbate the underlying disorder of patients with CFS (33). By interrupting the exercise, we aimed to postpone early acidosis, peripheral pain and fatigue and reduce the typical exacerbation of symptoms. The purpose of providing 6 bouts was that even the healthy sedentary subjects would experience sufficient submaximal exercise stimulus.
Fig. 1. Complete exercise protocol showing the 6 exercise bouts (shaded areas) and rest periods between bouts.
Warming up: Each exercise bout started with a warming-up period in which the workload was gradually increased by 1 kWatt/2 s until the intended workload was reached.
60”: Each plateau phase at a certain workload lasted for 60 s. Each exercise bout consisted of 2 plateau phases at incremental workloads.
Cooling down: Each exercise bout finished with a cooling-down period of 30 s.
90”: After each exercise bout, a rest period of 90 s was provided.
Statistical analysis
All data were analysed using SPSS 12.0© for Windows (SPSS Inc. Headquarters, Wacker Drive, Chicago, IL, USA). Given the normality of the variables (one-sample Kolmogorov-Smirnov goodness-of-fit test), parametric statistics were used.
Group characteristics. Mean and standard deviation (SD) for age, serum NO levels and the 8 bilateral PPT sites before and after exercise were calculated. Comparability of the 3 groups for age, length, height, PPTs and NO was assessed with a one-way analysis of variance (ANOVA) and for gender with the χ2 test. Since obesity is related to endothelial dysfunction (34), results for NO were corrected for body mass index (BMI).
Changes in nitric oxide and mean pressure pain thresholds. The change in NO and PPT measurements were compared between the 3 groups prior to and following exercise with a 2-factor repeated measures ANOVA with group (CLBP, CFS and controls) and time (pre- and post-exercise) as factors. Age, gender, and exercise duration were entered as covariates. Since 26 participants were unable to complete all 6 bouts in the exercise protocol, exercise duration was entered as covariate in the repeated measures ANOVA. Bonferroni post hoc tests were used. The ANOVA results for NO were corrected for BMI as well as for age, gender, and exercise duration. Mean PPT was the average of the 8 PPTs measured on the right-hand and left-hand sides prior to and following exercise.
Correlations between nitric oxide and mean pressure pain thresholds. Pearson correlation coefficients were calculated to reveal possible correlations between NO and mean PPTs. A power analysis determined that at least 21 participants per group were necessary to establish statistical significance at a power of 0.80. The significance level was set at 0.05.
RESULTS
Completion exercise protocol
Two healthy subjects, 20 patients with CFS and 4 patients with CLBP were unable to complete the exercise test. The total exercise duration (rest periods not included) is shown in Table I.
| Table I. Number and percentage of patients completing the exercise protocol for each of the 6 respective bouts |
| | 1 bout (190’’) n (%) | 2 bouts (415’’) n (%) | 3 bouts (690’’) n (%) | 4 bouts (1000’’) n (%) | 5 bouts (1348’’) n (%) | 6 bouts (1741’’) n (%) |
| Healthy (n = 31) | | | | | 2 (7) | 29 (93) |
| CFS (n = 26) | 1 (4) | 2 (8) | | 10 (38) | 7 (27) | 6 (23) |
| CLBP (n = 21) | | | | 1 (5) | 3 (14) | 17 (81) |
| (190’’) etc.: time (s) spent on the bicycle ergometer for each respective bout. CFS: chronic fatigue syndrome; CLBP: chronic low back pain. |
Group characteristics
Mean and SD for age, pain intensity, quality of life, NO serum concentrations, and PPTs prior to and following the ergometer bicycle test are shown in Table II. The 3 groups were comparable regarding age (p = 0.843), body length (p = 0.130), weight (p = 0.152), and gender distribution (p = 0.116). Patients with CFS showed lower PPTs, both prior to and following exercise, compared with healthy controls and patients with CLBP (p varying between < 0.001 and 0.023). No significant differences were found between the control subjects and patients with CLBP for any of the PPTs. Following exercise, NO concentrations were higher in patients with CLBP compared with patients with CFS (p = 0.044), but when accounting for BMI the difference was, however, no longer statistically significant.
| Table II. Demographic data, outcome measures, nitric oxide (NO) and pressure pain thresholds for 8 bilateral sites (PPTs; absolute and mean, right and left sides) before and after exercise. |
| Demographic data | CFS (n = 26) 21♀ 5♂ Mean (SD) | CLBP (n = 21) 11♀ 10♂ Mean (SD) | Healthy (n = 31) 21♀ 10♂ Mean (SD) |
| Age | 41.52 (11.38) | 41.55 (12.40) | 39.88 (12.63) |
| Length | 168.54 (8.11) | 173.29 (9.62) | 170.06 (6.61) |
| Weight | 66.69 (13.80) | 73.24 (11.13) | 67.65 (11.33) |
| Outcome measures |
| Pain VAS | 5.11 (2.31) | 3.63 (2.50) | 0.20 (0.44) |
| SF-36 | 300.81 (122.50) | 540.74 (129.60) | 473.72 (337.49) |
| ODQ | – | 18.57 (15.09) | – |
| BDI | 16.46 (8.77) | 5.59 (4.84) | 1.68 (4.339) |
| Measurements before exercise (NO and PPTs) |
| NO | 8.3 (6.45) | 13.36 (11.62) | 9.38 (5.14) |
| Arm L | 3.21 (2.44) | 7.08 (4.38) | 5.70 (3.34) |
| Arm R | 2.95 (2.06) | 6.72 (4.48) | 5.64 (3.66) |
| Hand L | 4.61 (3.18) | 9.77 (3.90) | 8.23 (3.06) |
| Hand R | 4.78 (3.46) | 9.35 (3.78) | 8.02 (2.65) |
| Back L | 5.12 (3.81) | 8.91 (3.05) | 7.77 (3.04) |
| Back R | 4.44 (2.66) | 8.43 (2.85) | 7.68 (2.92) |
| Calf L | 3.93 (2.67) | 7.20 (3.16) | 6.96 (3.15) |
| Calf R | 3.75 (2.53) | 7.37 (3.12) | 6.88 (3.12) |
| Mean | 4.10 (2.59) | 8.10 (3.02) | 7.11 (2.74) |
| Measurements after exercise (NO and PPTs) |
| NO | 8.38 (6.92) | 14.25 (11.94) | 8.88 (5.04) |
| Arm L | 2.51 (2.27) | 7.46 (4.38) | 6.10 (3.68) |
| Arm R | 2.38 (2.23) | 7.16 (4.48) | 5.35 (2.93) |
| Hand L | 3.77 (3.63) | 8.47 (4.17) | 7.67 (3.69) |
| Hand R | 3.77 (3.09) | 8.35 (4.35) | 7.25 (3.62) |
| Back L | 4.24 (2.46) | 9.98 (3.85) | 9.23 (4.28) |
| Back R | 4.14 (3.27) | 8.88 (3.45) | 8.62 (3.70) |
| Calf L | 3.59 (2.35) | 8.08 (3.52) | 7.84 (3.18) |
| Calf R | 3.55 (2.40) | 7.89 (3.80) | 8.10 (3.26) |
| Mean | 3.51 (2.48) | 8.28 (3.49) | 7.56 (3.17) |
| Different parts of the body represent the locations where pressure pain thresholds (PPT) were assessed, the values are expressed in kg/cm3. CFS: patients with chronic fatigue syndrome; CLBP: patients with chronic low back pain; SF-36: Short-Form Health Survey 36; VAS: visual analogue scale (cm); ODQ: Oswestry Disability Index; NO: nitric oxide (µM/l); L: left; R: right; –: questionnaire not completed; BDI: body density index. |
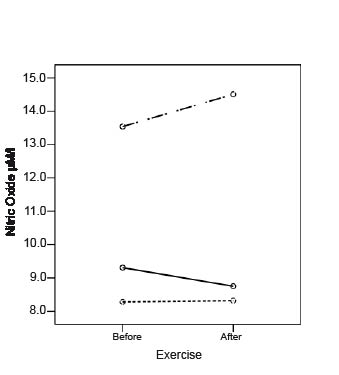
Changes in nitric oxide. As shown in Fig. 2, NO concentrations were considerably higher at baseline and increased in response to exercise in the CLBP-group, while they were initially lower and only marginal changes were seen in the CFS and healthy groups after exercise. However, the differences were not statistically significant (p = 0.207; and after accounting for BMI p = 0.267).
Fig. 2. Changes in nitric oxide (μM/l) following exercise.
Control subjects.
Patients with chronic low back pain.
Patients with chronic fatigue syndrome.
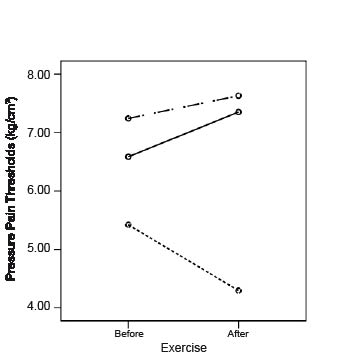
Changes in mean pressure pain thresholds. The mean PPT increased following exercise in healthy subjects and in patients with CLBP, and decreased in patients with CFS (Fig. 3). These changes were statistically significant (p = 0.001) for all 3 groups. When comparing the patients with CFS with the 2 other groups, they differed significantly from patients with CLBP (p = 0.002) and from healthy controls (p = 0.009).
Fig. 3. Changes in pressure pain thresholds (PPT; kg/cm3) following exercise.
Control subjects.
Patients with chronic low back pain.
Patients with chronic fatigue syndrome.
Correlations between nitric oxide and mean pressure pain thresholds. No significant correlations between NO and PPTs were found (Table III).
| Table III. Correlations between serum nitric oxide (NO) and mean pressure pain thresholds (PPTs) before and after exercise |
| | Mean PPT before exercise | Mean PPT after exercise | Difference PPT |
| CFS (n = 26) | CLBP (n = 21) | CON (n = 31) | CFS (n = 26) | CLBP (n = 21) | CON (n = 31) | CFS (n = 26) | CLBP (n = 21) | CON (n = 31) |
| NO before | R p-value | 0.101 0.624 | –0.278 0.223 | –0.068 0.716 | –0.035 0.865 | –0.388 0.082 | –0.133 0.476 | 0.244 0.230 | 0.363 0.106 | 0.174 0.349 |
| NO after | R p-value | 0.101 0.624 | –0.380 0.089 | 0.015 0.936 | –0.065 0.751 | –0.432 0.050 | –0.044 0.813 | 0.297 0.141 | 0.253 0.269 | 0.134 0.471 |
| Difference NO | R p-value | –0.028 0.894 | 0.285 0.210 | –0.009 0.961 | 0.125 0.541 | 0.141 0.542 | –0.122 0.512 | –0.269 0.185 | 0.260 0.256 | 0.269 0.144 |
| R: Pearson’s correlation coefficient; p-value: significance level; CFS: chronic fatigue syndrome; CLBP: chronic low back pain; CON: control. |
DISCUSSION
While many patients with low back pain recover after a few weeks, some patients develop CLBP with recurrent episodes of pain (35). We have found previously that patients with CFS often experience chronic widespread pain, with typical exacerbations of pain following exercise (36). Central mechanisms may be responsible for the deregulated anti-nociception. Addressing patients with CFS, the results of the present study are consistent with our initial hypothesis. The decreases in PPTs that we found following exercise suggest abnormal central pain processing in CFS, but not in CLBP. Serum NO appears to be unrelated to PPTs.
Group characteristics at baseline
The overall reduced PPTs in patients with CFS, compared with healthy subjects and patients with CLBP suggest generalized hyperalgesia and indicate abnormal pain processing. It is somewhat surprising that no hyperalgesia was found in patients with CLBP, while several other studies have found lower PPTs in patients with CLBP compared with healthy subjects, at locations related to the lower back (6, 7), and also for sites unrelated to the back (7). Our patients with CLBP were only mildly to moderately disabled, as suggested by the lower scores on the Oswestry Disability Questionnaire and the high scores on the SF-36. This may suggest that hyperalgesia is present only in the severely disabled patients with CLBP rather than in the entire CLBP population.
Compared with CFS and healthy subjects, serum NO levels were significantly higher in the CLBP-group. After accounting for BMI, the difference was, however, no longer present. The high NO levels in patients with CLBP may indicate an inflammatory or degenerative process, and are in line with earlier reports on elevated NO production in low back pain (37). The observations of similar NO levels in patients with CFS and healthy controls is surprising, since previous studies have reported elevated NO levels in patients with CFS (17). However, Kurup & Kurup (17), analysed plasma rather than serum NO levels.
Changes in nitric oxide after exercise
The NO-response to submaximal exercise did not differ between groups, even though NO levels increased in patients with CLBP and remained more or less stable in patients with CFS and healthy subjects. It is known that exercise triggers NO release (19), but no changes were observed in the healthy controls and patients with CFS. NO is a very volatile molecule compliant with many external factors. Furthermore, the procedure of NO analysis is based on the analysis of the breakdown products nitrite and nitrate. The procedure may be insufficient to reveal acute changes in NO concentrations.
Changes in mean pressure pain thresholds after exercise
The decrease in mean PPT in response to submaximal aerobic exercise in patients with CFS is an important finding. Our results extend the preliminary findings, provided by Whiteside et al., that exercise lowers PPTs in CFS (9). Our data also indicate that patients with CLBP and healthy subjects reacted “normally” to exercise: the PPTs increased, while they decreased in patients with CFS. This finding suggests a pathophysiological reaction to exercise. In normal circumstances, pain thresholds increase during and following exercise. This is due to the release of endogen opioids and growth factors (10) and other strong pain inhibitory mechanisms (“descending inhibition”) orchestrated by the central nervous system (11). Our data suggest a lack of descending inhibition during submaximal aerobic exercise in patients with CFS with chronic widespread pain.
Exercise is frequently encountered as a central component of the treatment of chronic musculoskeletal pain. Because patients’ tolerance to exercise is a determining factor in treatment compliance, it is important to know how chronic pain patients react to physical efforts. Patients with CFS are known to tolerate physical efforts badly, with a worsening of their symptoms up to 48 h after an acute bout of exercise and to have a very slow recovery (12, 38). Pain complaints worsen following exercise in patients with CFS, possibly as the result of decreased pain thresholds in response to exercise. Also the findings of Jammes et al. (39) may explain the muscle pain following exercise reported by patients with CFS. It has been reported that patients with CFS respond to incremental exercise with lengthened and accentuated oxidative stress, together with marked alterations of the muscle membrane excitability (38). Furthermore, evidence for immune deregulation in patients with CFS following (sub)maximal exercise (14), causing post-exertional malaise, which can include pain, has been published. Together, these findings may explain the symptom exacerbation and decreased PPTs following exercise.
In our healthy subjects and in the patients with CLBP, PPTs increased in response to submaximal aerobic exercise. These results are consistent with the pilot study by Hoffman et al. (40), which to our knowledge is the only study analysing pain response in relation to exercise in patients with CLBP. Thus, pain physiology during exercise seems to be normal in mildly disabled patients with CLBP.
Correlation between nitric oxide and mean pressure pain thresholds
NO is able to reduce central pain inhibition (18), and may induce peripheral and central sensitization by reducing receptor thresholds (37). However, we found no correlations between NO and PPTs. Thus, our study supports the hypothesis that central nociceptive mechanisms are deregulated in CFS, but NO does not seem to play a role and NO also appeared to be unrelated to pain in the patients with CLBP.
Study limitations. Our results should be interpreted in light of the study limitations. It was not easy to standardize the exercise protocol for the 3 groups, given the great inter-individual differences in exercise capacity. It is probable that not all participants ended the exercise tests at a similar point of exhaustion. Therefore, the exercise time was entered as covariate in the analysis. Further research should use more standardized submaximal exercise protocols and also address a dose-response relationship to determine which exercise stimuli cause which response in order to steer rehabilitation programmes. Also, the pathophysiology behind the impaired pain inhibition deserves further attention. Although NO quantification is often sensitive to bias, the present results do not point to a specific role for NO in chronic pain experienced by patients with CFS or CLBP.
Furthermore, the issue of selection bias should be addressed. Patients with CFS were asked whether they were able to cycle prior to inclusion into the study. This may lead to recruitment of less severely disabled patients with CFS. The recruitment of healthy controls and patients with CLBP via pamphlets and physical therapists may also imply selection bias. Pamphlets were distributed in the university, university hospital and in physical therapy practices, and consequently reached a select population. The same applies to relatives and acquaintances of the researchers. Finally, participants reported having followed instructions and ceased medication use 24 h prior to study participation, but we cannot be sure that this was in fact the case.
In conclusion, patients with CFS manifest generalized hyperalgesia. The results of the present investigation might suggest the presence of abnormal central pain processing during submaximal aerobic exercise. Mean PPTs in patients with CFS decreased following sub-maximal bicycle ergometer exercise, while increased PPTs were seen in the CLBP and the healthy groups. Because of the great inter-individual differences in exercise capacity, more research may be warranted to address a dose-response relation. NO concentrations were higher among patients with CLBP, and increased following submaximal aerobic exercise, while they were considerably lower and changed only marginally in patients with CFS and healthy controls. NO appears to be unrelated to widespread pain and exacerbation of pain following exercise in the CFS and CLBP groups.
Acknowledgements
Mira Meeus and Nathalie Roussel are financially supported by a PhD grant supplied by the Department of Health Sciences, Artesis University College Antwerp, Antwerp, Belgium (G 807 and G 806) and co-financed by the Faculty of Physical Education and Physiotherapy, Vrije Universiteit Brussel (VUB), Brussels, Belgium (OZR project OZ.R. 1234/MFYS Wer2). Mira Meeus is now a postdoctoral fellow of the Fund for Scientific Research Flanders (FWO).
The first 2 authors had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. No financial or other relationships could lead to any conflict of interest.
The authors are grateful to Kenny De Meirleir for diagnosing the study participants, Lieve De Hauwere for taking the blood samples and assisting in the exercise tests, to Annemie Wielant and Marc Frémont (RED Laboratories, Zellik, Belgium) for analysing the blood samples, and to Andre Farasyn for kindly providing his expertise on PPT assessment.
REFERENCES