OBJECTIVE: Botulinum toxin is known to relieve upper limb spasticity, which is a disabling complication of stroke. We examined its effect on quality of life and other person-centred perspectives.
DESIGN: A multi-centre, randomized, double-blind, placebo-controlled study.
Patients: Ninety-six patients were randomized (mean age 59.5 years) at least 6 months post-stroke. Mean time since stroke was 5.9 years.
METHODS: Patients received either botulinum toxin type A or placebo into the affected distal upper limb muscles on 2 occasions, 12 weeks apart. Assessment was undertaken at baseline, 8, 12, 20 and 24 weeks. The primary outcome measure was the Assessment of Quality of Life scale (AQoL). Secondary outcome assessments included Goal Attainment Scaling (GAS), pain, mood, global benefit, Modified Ashworth Scale (MAS), disability and carer burden.
RESULTS: The groups did not differ significantly with respect to quality of life, pain, mood, disability or carer burden. However, patients treated with botulinum toxin type A had significantly greater reduction in spasticity (MAS) (p < 0.001), which translated into higher GAS scores (p < 0.01) and greater global benefit (p < 0.01).
CONCLUSION: Although no change in quality of life was demonstrated using the AQoL, botulinum toxin type A was found to be safe and efficacious in reducing upper limb spasticity and improving the ability to achieve personal goals.
Key words: botulinum toxin, muscle spasticity, stroke.
J Rehabil Med 2009; 41: 536–544
Correspondence address: Melinda Munns, Ipsen Pty Ltd, PO Box 820, Glen Waverley, Victoria 3150, Australia. E-mail: melinda.munns@ipsen.com
Submitted July 3, 2008; accepted February 11, 2009
INTRODUCTION
Each year stroke affects approximately 53,000 people in Australia, 150,000 in the UK and 750,000 in the USA (1, 2). Upper limb spasticity is a common complication that can lead to abnormal limb posturing and interfere with both active and passive function, resulting in a reduction in quality of life and increased carer burden (3).
Intramuscular botulinum toxin type A (BoNT-A) offers the possibility of local treatment for focal spasticity without affecting sensation and without the systemic side-effects of oral medication. Moreover, targeting specific muscle groups without affecting others has the theoretical potential to unmask selective voluntary movement in situations where this is over-ridden by mass patterns of spasticity in antagonistic muscle groups (3).
There are now at least 12 published randomized controlled trials (with a total of over 500 patients) of BoNT-A in the treatment of upper limb post-stroke spasticity. These have consistently demonstrated that the treatment is safe and effective in reducing unwanted muscle spasticity (4–6) and that the effect is maintained over repeated treatments (7). Functional benefits are also demonstrated, in terms of reduction of disability and carer burden (8, 9), although they may lag behind the reduction in spasticity itself (10).
However, few studies have focused on the impact of BoNT-A from the patient’s personal perspective. This study addressed the effect of BoNT-A not only on disability, but also on the patient’s perceived state of well-being, using a standard Assessment of Quality of Life measure (AQoL). In addition, we examined the effect on symptoms (such as pain and mood) and on the achievement of personal goals.
METHODS
This study was a prospective, placebo-controlled, double-blind, randomized, multicentre study designed to assess the effect of the administration of BoNT-A (Dysport®, Ipsen, Slough, UK) on quality of life and other person-centred outcomes in patients treated for upper limb spasticity after stroke. All patients gave their informed consent. The study had ethics permission from independent research ethics committees at each investigational site.
Participants
Adults with hemiplegic stroke and severe or moderately severe spasticity following stroke were recruited from spasticity clinics in 6 centres in Australia (Melbourne and Sydney), through referral from hospital stroke/neurology units, rehabilitation centres, community physiotherapists and newspaper advertisements.
Patients were included if they:
• were over the age of 18 years and had had a stroke at least 6 months previously;
• had moderate to severe spasticity of the arm as defined by a minimum score of 2 on the Modified Ashworth Scale (MAS) in at least 2 out of 3 of wrist, elbow and finger flexor muscles and a minimum of 1+ for the third area;
• had sufficient cognitive and communication ability to be able to give written informed consent.
Excluded were those:
• with established severe contracture or other neurological impairments;
• receiving concurrent aminoglycoside antibiotics;
• who had received botulinum toxin treatment within the past 120 days or had been previously treated with phenol or intrathecal baclofen for arm spasticity.
Patients on oral anti-spasticity medication were included provided their dosage remained stable in the month prior to study entry.
Eligibility for treatment was further assessed at baseline. Only those with stable spasticity and disability ratings between pre-baseline assessment (week –4) and baseline (week 0) were randomized.
Intervention
Treatment comprised injections of BoNT-A or equivalent placebo (total dose range 750–1000 units) into the principal spastic muscles of the distal upper limb (restricted to muscles acting at elbow, wrist and finger joints) at week 0. The selection of muscles, use of single or multiple injection sites within a given muscle, and electromyography or nerve/muscle stimulation to assist accurate placement were all at the clinicians’ discretion.
Patients received re-treatment with the same agent as their first cycle at week 12 with a total dose range of 500–1000 units according to the response in the initial cycle. This flexible dose regimen allowed investigators to optimize the treatment on an individual basis as would occur in routine clinical practice.
Specific objectives and hypotheses
This study tested the hypothesis that reduction of spasticity with 2 cycles of BoNT-A injections would: (i) improve patient quality of life; (ii) improve symptoms such as pain and depression; and (iii) enhance the attainment of individual goals for treatment.
Outcome measurement
The outcome measures were undertaken at weeks 0 (baseline), 8, 12, 20 and 24. The principal outcome points for analysis were at the peak of each cycle (weeks 8 and 20), except for global benefit, which was evaluated at the end of each cycle (weeks 12 and 24). On the basis of past trial results, it was anticipated that the second injection would maintain response or potentially enhance the level of response, and week 20 was predetermined as the primary outcome point for the trial when the effect of the second BoNT-A injection should be maximal (7, 8).
Primary outcome measure
The primary outcome for the study was the assessment of patient well-being and quality of life at week 20, as measured by the AQoL (11–13). The AQoL is a multi-dimensional utility instrument designed to profile health status according to level of disability and handicap. The utility score is presented on a scale of 1.00 (representing full quality of life) to 0.00 (representing death-equivalent quality of life).
Secondary outcome measures
1. Person-centred secondary outcomes
• Pain associated with spasticity in the hand, wrist and elbow was rated “at rest” and “on movement” using a (100-mm) visual analogue scale.
• Depression was recorded using the Hospital Anxiety and Depression Rating Scale (HADs) (14, 16).
• The achievement of individual goals for treatment was recorded using Goal Attainment Scaling (GAS) (17, 18). The GAS was applied according to a method described previously (19). At baseline patients, with their treating team, identified up to 2 personal goals for treatment and one chosen activity from the Patient Disability Scale (PDS) (see below). Goals were weighted by importance and difficulty. Goal attainment was rated at weeks 8 and 20 on a 5-point scale, where “0” denotes the expected level of achievement; “+1” and “+2” are respectively “a little” and “a lot” better than expected, whilst “–1” and “–2” are correspondingly a little and a lot less than the expected level. These attainment levels were combined in a single score by applying the formula recommended by Kiresuk & Sherman (17). This formula is designed to transform the sum of the attainment levels for each goal (× their relative weights) into a standardized measure or “T score” normally distributed around a mean of 50 and standard deviation of 10, if the results exceed and fall short of expectation in roughly equal proportion.
2. Other measures
• Spasticity. In order to confirm an effect on spasticity, muscle tone was assessed during passive range of movement at the elbow, wrist and finger joints using the MAS (20). The MAS was assessed 4 weeks post-injection in addition to the standard outcome measure time-points of 8, 12, 20 and 24 weeks. The 3 scores were summed to give a composite value across all joints, as described by Bakheit et al. (4, 5, 7) and Francis et al. (10), reflecting the overall reduction in muscle tone in the limb. A score of 1+ was assigned the value of 1.5.
• Ability to perform hand movements and upper arm function were captured using a Modified Motor Assessment Scale (MMAS) (21) consisting of the 3 upper limb domains (18 items in total).
• Patient disability and carer burden were measured using the PDS and Carer Burden scale (CBS) (22). The PDS (8 items) identifies the patients’ ability to care for their affected limb (dressing, maintaining hygiene, etc.) and to use it actively, for example, for standing/walking balance. The CBS (4 items) identifies care tasks, such as dressing and maintaining hygiene, where these are performed by a carer. Each item is scored on a 5-point Likert scale. Items scores were summed and divided by the number of items answered to give a summary score from 0 = none to 4 = maximum disability or carer burden.
• At baseline, patients chose one activity in the PDS as their preferred activity to improve following treatment. This was designated the Patient Functional Outcome Measure (PFOM).
• Global Assessment of Benefit. A global assessment of benefits was made at the last visit of each cycle (week 12 and week 24), by both the investigator and the patient (or guardian), taking into account the peak efficacy and duration of efficacy since last treatment. The patient was asked “How would you rate the overall benefit you have received to your arm in the time since your last injection?”, and the response was categorized on a 5-point Likert scale.
Also recorded were:
• all adverse events, regardless of causality and intensity;
• any changes in medication;
• the number of physiotherapy and/or occupational therapy sessions that were undertaken in the preceding 4 weeks.
Sample size and power calculations
We calculated a priori that a sample size of 21 patients would be required for each study group, to have an 80% chance of detecting as significant (at the 2-sided 5% level) a mean difference of > 0.15 (SD 0.23) in AQoL scores between the 2 groups at week 20. Similarly, 51 patients per group were needed to detect with similar power a significant group interaction in change of AQoL score from baseline to week 20, based on a mean difference in change score of 0.13 (SD of 0.23).
Randomization and allocation concealment
At baseline, patients were randomized to receive treatment with either BoNT-A or matching placebo. A computer-generated master list of randomized treatment allocation codes was prepared centrally by an independent organization (Penn Pharmaceutical Services Ltd, Tredegar, UK) with a 1:1 proportion of patients assigned to each group. Group assignment was determined independently of the treating team by sequential allocation of treatment packs from the hospital pharmacy with pre-assigned randomization numbers.
Blinding
The patients, investigators and treating teams were all blinded to group assignment. Vials of investigational drug (BoNT-A or placebo) contained a white freeze-dried pellet that was reconstituted by the investigator in sodium chloride solution (0.9%) at the time of injection. The blinding was not broken at any stage during this trial. The master randomization list was supplied only after the database had been locked and the statistical plan approved.
Statistical analysis
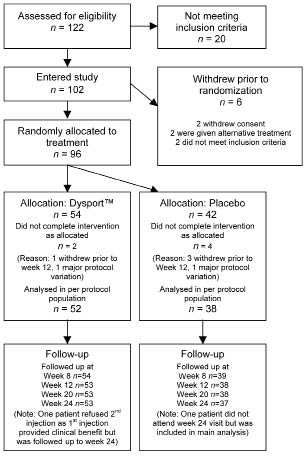
Efficacy data for primary and secondary end-points were analysed using an intention-to-treat population, defined as all patients who were randomly assigned and who received at least one dose of study medication (54 BoNT-A, 42 placebo). Six patients did not complete as allocated leaving 90 for analysis per protocol (52 BoNT-A, 38 placebo). Missing data were imputed using a Last Observation Carried Forward (LOCF) method.
The between-treatment comparison was carried out by fitting an analysis of covariance model (ANCOVA) to the data with baseline AQoL included as a covariate. Treatment effect estimates, 95% confidence intervals (95% CI), and p-values were calculated. Standard diagnostic tests showed no evidence of non-linearity of the regression relationship, non-normality of the residual distribution, or non-constancy of variance. Therefore no transformations of the data were undertaken, and parametric tests were applied throughout for consistency.1
1An alternative analysis using non-parametric statistics gave similar results and is available on request from the corresponding author.
Mean change in AQoL scores was also assessed separately for BoNT-A and placebo within each treatment arm. Mean change, and 95% CI were calculated for the difference in final AQoL vs baseline.
Descriptive summary statistics for differences between the mean scores and mean change from baseline (SD) for all other secondary measures are presented. Within-group and between-group comparisons were carried out using, respectively, paired and independent t-tests. Dichotomous variables were analysed using χ2 tests.
RESULTS
Between November 2004 and January 2006 a total of 122 subjects were screened, of which 102 were recruited into the study and 96 were randomized to treatment (54 BoNT-A, 42 placebo) (see Fig. 1 and discussion). Table I summarizes the demographic data in the 2 groups. This was a relatively young group (mean age 59 years) with long-term spasticity, on average nearly 6 years after stroke onset. In the 102 patients recruited, left-sided hemiparesis was more common than right-sided hemiparesis (67.7% vs 32.3%, respectively). A similar proportion of patients had left-sided hemiparesis in the placebo group (69%) and the BoNT-A-treated group (67%). Table II summarizes the different muscles chosen by the physicians in this study to treat upper limb spasticity and the corresponding BoNT-A and equivalent saline doses administered.
Fig. 1. CONSORT diagram showing participant flow and retention.
| Table I. Subject demographic data |
| Demographic parameter | Placebo n = 42 | BoNT-A (Dysport®) n = 54 |
| Age, years Mean (SD) Range | 58.4 (14.6) 21–83 | 59.7 (12.2) 32–82 |
| Body mass index Mean (SD) Range | 26.7 (4.9) 18.6–38.4 | 27.9 (5.8) 18.2–50.2 |
| Gender, male/female. | 26/16 | 32/22 |
| Race, n (%) Caucasian Asian | 40 (95.2) 2 (4.8) | 49 (90.7) 5 (9.3) |
| Time since onset of stroke, years Mean (SD) Range | 6.6 (12.6) 0.6–61.1 | 5.3 (8.7) 0.5–60.8 |
| BoNT-A: botulinum toxin type A; SD: standard deviation. |
| Table II. Dose of botulinum toxin type A (BoNT-A) and saline equivalent injected in upper limb muscles at the first and second cycle |
| Muscle | First cycle | Second cycle |
| BoNT-A units, median (min, max) | Patients injected, n (%) | Placebo (saline: equivalent to dose in U), median (min, max) | Patients injected, n (%) | BoNT-A units, median (min, max) | Patients injected, n (%) | Placebo (saline: equivalent to dose in U), median (min, max) | Patients injected, n (%) |
| Biceps brachii | 300 (0, 400) | 45 (83) | 300 (0, 400) | 34 (81) | 300 (0, 400) | 41 (77) | 300 (0, 400) | 25 (66) |
| Brachialis | 100 (0, 200) | 9 (16) | 100 (0, 300) | 5 (12) | 100 (0, 250) | 8 (15) | 100 (0, 250) | 4 (11) |
| Brachioradialis | 100 (0, 250) | 15 (28) | 250 (0, 250) | 8 (19) | 100 (0, 300) | 9 (17) | 200 (0, 400) | 10 (26) |
| Triceps | 275 (0, 300) | 2 (4) | 150 (0, 200) | 2 (5) | 250 (0, 500) | 5 (9) | 175 (0, 250) | 2 (5) |
| Flexor digitorum profundus | 150 (0, 300) | 46 (85) | 200 (0, 400) | 38 (90) | 150 (0, 400) | 41 (77) | 150 (0, 400) | 34 (89) |
| Flexor digitorum superficialis | 200 (0, 400) | 53 (98) | 225 (0, 400) | 40 (95) | 200 (0, 400) | 50 (94) | 200 (0.400) | 35 (92) |
| Flexor carpi ulnaris | 150 (0, 500) | 40 (74) | 150 (0, 375) | 31 (74) | 150 (0, 400) | 34 (64) | 150 (0, 300) | 30 (79) |
| Flexor carpi radialis | 150 (0, 200) | 31 (57) | 150 (0, 200) | 26 (62) | 150 (0, 250) | 27 (51) | 150 (0, 250) | 24 (62) |
| Finger/thumb flexors | 100 (0, 300) | 21 (39) | 100 (0, 250) | 19 (45) | 200 (0, 375) | 25 (47) | 150 (0, 250) | 15 (39) |
Primary outcome
MAS measurements confirmed a significant reduction in muscle spasticity for BoNT-A-treated patients compared with those on placebo, at all time-points (Table III). Despite this effect on muscle tone, no significant difference between the groups was observed with respect to change in quality of life from week 0 to week 20 as measured by the AQoL. Indeed, between-group comparison in the intention-to-treat population (n = 96) yielded a treatment effect estimate (ANCOVA) of –0.031 (standard error 0.03, p = 0.27). This indicates that, if anything, the trend was towards greater improvement in the placebo group, although this did not reach significance. These results did not alter when the analysis was adjusted for age, gender, cluster (hospital grouped by Australian state) or centre. Sub-group domain analyses also showed no evidence of benefit in favour of active treatment.
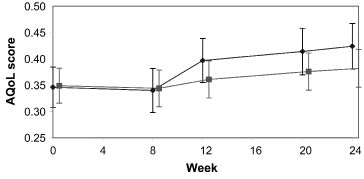
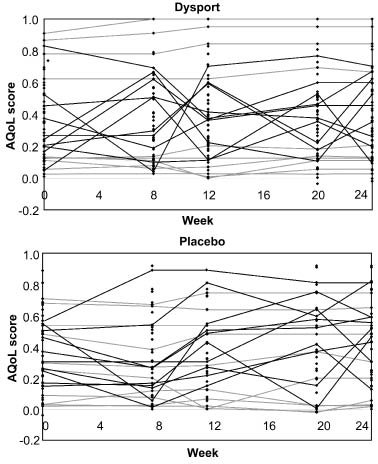
However, throughout the study period, the serially-recorded AQoL scores demonstrated a very high degree of individual variation over time. This is illustrated in Figs. 2 and 3, which show the mean AQoL scores at each time-point together with a trajectory of single patient AQoL scores from the 10 most variable and 10 least variable patients in each group. Some patients showed changes in the region of ± 0.4–0.6 over a single 4-week period, and this variation was evident in both treatment groups.
| Table III. End-points for primary and secondary outcomes at weeks 0, 8 and 20 and change from baseline |
| | End-points | Change from baseline (week 0) |
| Placebo (n = 42) Mean (SD) | BoNT-A (n = 54) Mean (SD) | Between-group difference in end-point | Placebo (n = 42) Mean (SD) | Within-group 95% CI | BoNT-A (n = 54) Mean (SD) | Within-group 95% CI | Between-group difference in change from baseline |
| Difference 95% CI | p-value | Difference (95% CI) | p-value |
| Primary outcome AQoL |
| Week 0 Week 20 | 0.35 (0.25) 0.41 (0.27) | 0.35 (0.24) 0.38 (0.25) | 0.00 (–0.10, 0.10) 0.03 (–0.76, 1.36) | 1.00 0.57 | 0.06 (0.13) | 0.02, 0.10 | 0.03 (0.15) | –0.01, 0.07 | –0.03 (–0.09, 0.02) | 0.27 |
| Secondary outcomes |
| Pain | | | | p-value | | p-value | | |
| Week 0 Week 8 Week 20 | 36.0 (35.8) 30.9 (37.9) 33.1 (40.3) | 44.3 (46.6) 34.4 (43.2) 34.3 (47.6) | –8.30 (–25.56, 8.96) –3.40 (–20.53, 13.73) –1.25 (–20.14, 17.64) | 0.34 0.69 0.89 | NA –4.9 (36.4) –0.7 (39.1) | 0.41 0.92 | NA –9.9 (39.5) –10.8 (42.0) | 0.07 0.07 | 5.07 (–10.9, 21.0) 10.14 (–8.1, 27.4) | 0.53 0.25 |
| HADS |
| Week 0 Week 8 Week 20 | 5.2 (3.5) 4.8 (3.2) 5.2 (4.1) | 5.7 (3.2) 5.7 (3.2) 5.6 (3.4) | –0.50 (–1.86, 0.86) –0.84 (–2.19, 0.51) –0.41 (–1.99, 1.67) | 0.48 0.22 0.61 | NA –0.3 (2.2) 0.1 (3.2) | 0.44 0.80 | NA 0.0 (2.4) –0.17 (2.4) | 1.00 0.61 | –0.3 (–1.24, 0.68) –0.07 (–0.87, 1.47) | 0.56 0.61 |
| GAS |
| Week 0 Week 8 Week 20 | 29.5 (5.6) 29.8 (5.7) 31.1 (7.6) | 28.7 (5.5) 32.2 (10.3) 35.7 (10.1) | 0.80 (–1.54, 3.14) –2.08 (–6.07, 1.27) –4.32 (–8.50, –0.70) | 0.49 0.20 0.02 | NA 0.2 (7.0) 1.8 (8.5) | 0.83 0.20 | NA 3.5 (9.4) 7.0 (8.9) | 0.01 < 0.001 | –3.30 (–6.80, 0.36) –5.20 (–9.08, 1.28) | 0.07 0.01 |
| MAS across all joints1 |
| Week 0 Week 8 Week 20 | 6.9 (1.1) 6.5 (1.3) 6.6 (1.3) | 7.1 (1.2) 5.3 (1.7) 5.3 (1.7) | –0.22 (–0.69, 0.25) 1.20 (0.57, 1.83) 1.34 (0.68, 2.00) | 0.35 < 0.001 < 0.001 | NA –0.3 (1.2) –0.2 (1.2) | 0.08 0.28 | NA –1.8 (1.7) –1.8 (1.6) | < 0.001 < 0.001 | 1.47 (0.84, 2.10) 1.59 (0.98, 2.00) | < 0.001 < 0.001 |
| MMAS |
| Week 0 Week 8 Week 20 | 1.5 (1.7) 1.5 (1.9) 1.4 (2.0) | 1.1 (1.8) 1.4 (1.8) 1.3 (2.2) | 0.39 (–0.32, 1.10) 0.06 (–0.75, 0.87) 0.10 (–0.80, 1.00) | 0.28 0.88 0.83 | NA 0 –0.03 (1.4) | 1.0 0.91 | NA 0.3 (1.2) 0.2 (1.2) | 0.07 0.24 | –0.32 (–0.79, 0.15) –0.22 (–0.75, 0.31) | 0.18 0.41 |
| PDS |
| Week 0 Week 8 Week 20 | 1.8 (0.7) 1.5 (0.7) 1.5 (0.7) | 1.8 (0.8) 1.6 (0.8) 1.6 (0.7) | –0.04 (–0.37, 0.29) –0.12 (–0.45, 0.20) –0.07 (–0.38, 0.24) | 0.81 0.46 0.66 | NA –0.3 (0.5) –0.3 (0.7) | 0.003 0.02 | NA –0.2 (0.6) –0.3 (0.5) | 0.01 < 0.001 | –0.05 (–0.28, 0.18) –0.01 (–0.27, 0.25) | 0.66 0.94 |
| CBS |
| Week 0 Week 8 Week 20 | 1.9 (1.2) 1.7 (1.2) 1.5 (1.2) | 1.5 (1.1) 1.2 (0.9) 1.0 (0.8) | 0.39 (–0.20, 0.97) 0.45 (–0.15, 1.05) 0.42 (–0.17, 1.01) | 0.19 0.14 0.16 | NA –0.1 (0.6) –0.4 (1.0) | 0.41 0.08 | NA –0.2 (1.1) –0.4 (1.1) | 0.35 0.04 | 0.06 (–0.48, 0.60) –0.02 (–0.65, 0.61) | 0.83 0.95 |
| 1Combined MAS score and change in MAS were also significantly improved in botulinum toxin type A (BoNT-A; Dysport®)-treated patients compared with placebo-treated patients at week 4 and week 16 (independent t-test, p < 0.001). Between-group difference in end-point is for primary outcome AQoL made by analysis of covariance model and for secondary outcomes is it independent t-test. Significant values are shown in bold. CI: confidence interval; SD: standard deviation: AQoL: Assessment of Quality of Life; NA: not applicable; HADS: Hospital Anxiety and Depression rating scale; GAS: Goal Attainment Scaling; MAS: Modified Ashworth Scale: MMAS: Modified Motor Assessment Scale; PDS: Patient Disability Scale; CBS: Carer Burden Scale. |
Fig. 2. Mean (standard error of mean) Assessment of Quality of Life (AQoL) score in patients treated with botulinum toxin type A (Dysport® ()) or placebo ().
Fig. 3. Trajectory plot of Assessment of Quality of Life (AQoL) scores at each time-point for the most and least variable patients in each treatment group. The intra-patient variability of AQoL is demonstrated in these trajectory plots of AQoL scores at weeks 0, 8, 12, 20, and 24. The 10 least variable (grey lines) and the 10 most variable patients (black lines) are presented in each treatment group. Individual dots represent all patient observations.
Person-centred secondary outcomes (Table III)
Less than half of patients reported significant pain. Table III provides data for all patients combining the scores of pain at rest and pain on movement. BoNT-A treatment failed to show significant improvement in pain compared with placebo-treated patient at rest and movement at any time-point. A sub-analysis of change in pain score on movement was conducted only for those with a pain rating ≥ 25 on movement at baseline (n = 40). Within this subset, both groups showed significant reduction in pain levels at both weeks 8 and 20 (paired t-test, p < 0.05 for placebo group, p < 0.01 for BoNT-A group, not shown in Table III). Those treated with BoNT-A had significantly higher levels of pain on movement at baseline (65.05 vs 47.37 for placebo, p = 0.01), and this may account for the relatively greater pain reduction in this group, which reached significance at week 12 (paired t-test, p < 0.05, not shown in Table III). However, no significant difference between the 2 groups was observed at either of the main time-points for this study (weeks 8 and 20).
Similarly, there was no significant change in mood, as measured using the HADS at any time-point in this study. However, less than a quarter (20/96 (21%)) screened positive for depression (HADS ≥ 8) at baseline, of which only 8 had scores in the abnormal range (HADS ≥ 11). These numbers were too small for statistical analysis.
The goal attainment scores, however, did reflect a functional benefit of BoNT-A-treatment at week 20. Goal attainment scores were 35.7 at week 20 for BoNT-A compared with 31.1 in placebo-treated patients (independent samples t-test, p < 0.01) (Table III and Fig. 4).
Fig. 4. Mean goal attainment scaling (GAS) scores for placebo and botulinum toxin type A (BoNT-A; Dysport®)-treated patients at week 0, 8 and 20 (∗∗p < 0.01).
Other secondary outcomes (Table III)
As might be expected in this population with a mean duration of spasticity of nearly 6 years, the MMAS did not show any motor gains in either group.
Both groups showed a modest reduction in patient disability (PDS) and carer burden (CBS) over the course of this study (possibly as a result of the concomitant therapy that was provided for the majority of patients), but there was no statistical difference between treatment groups. However, it should be noted that the baseline PDS scores were low in this series (mean 1.8). Moreover, 37/96 had no carer, confirming that this was a relatively independent study population.
On the other hand, at week 8 the chosen item from the patient disability rating scale (PFOM) was improved from baseline in 20/54 (37%) of the BoNT-A group compared with only 6/38 (15%) of the placebo group (χ2, p = 0.02). At week 20 the percentages remained steady at 34% for the BoNT-A group and 16% for the placebo group (χ2, p < 0.05).
Overall, a significantly higher proportion of both patients and investigators reported a benefit from treatment in the BoNT-A group compared with the placebo group at the end of the first treatment (week 12) and also the second treatment (week 24) (p = 0.01) (Table IV).
| Table IV. Global assessment of benefit (proportion reporting benefit) |
| | Placebo | BoNT-A | χ2* | p-value |
| Investigator Week 12 Week 24 | 12/38 (32%) 10/37 (27%) | 31/53 (58%) 31/53 (58%) | 6.54 8.94 | 0.01 0.003 |
| Patient Week 12 Week 24 | 13/38 (34%) 9/37 (24%) | 32/53 (60%) 35/53 (66%) | 6.14 15.74 | 0.01 < 0.001 |
| *Likelihood ratio χ2 values. BoNT-A: botulinum toxin type A. |
Concomitant treatment
Concomitant medications were started during the study in 52.1% of patients (57.4% BoNT-A, 45.2% placebo). There were no obvious patterns in concomitant medications ongoing, stopped or started during the study.
Whilst it is generally recommended that BoNT-A should be followed up by appropriate physical therapy to maximize benefit, such treatment is not currently universally available (23). In order to reflect current practice, the protocol stipulated that any concomitant therapies (physiotherapy and occupational therapy) would be given according to routine practice for the centre (23). In the event, therapies were continued in 9.4% of patients, and started in 57.3% (55.6% BoNT-A, 59.5% placebo) of patients during the study. The number of treatment sessions ranged from 0 to 91, with a mean 10.8 (SD 14.5), but there was no difference between the 2 groups (t –0.159, df 88, p = 0.87). However, 33% of patients did not receive concomitant therapy.
Adverse events
A list of adverse events is given in Table V. In this study, 67% of the BoNT-A and 62% of the placebo group experienced at least one adverse event, which were generally mild. These were considered treatment-related in 5.5% of BoNT-A-treated patients and 9.5% placebo-treated patients. No deaths occurred and no hospitalizations for treatment-related events. In particular, there were no instances of respiratory failure, swallowing problems or other evidence of remote spread of toxin.
| Table V. Summary of adverse events. Treatment-emergent adverse events experienced in > 3% of patients and treatment-related adverse events |
| | Placebo (n = 42) | BoNT-A (n = 54) | Combined (n = 96) |
| Treatment-emergent events (n (%)) |
| Fall Arthralgia Convulsion Contusion Shoulder pain UTI Back pain Chest pain Fatigue | 8 (19) 3 (7.1) 2 (4.8) 2 (4.8) 2 (4.8) 3 (7.1) 2 (4.8) 0 (0) 0 (0) | 6 (11.1) 3 (5.6) 4 (7.4) 3 (5.6) 3 (5.6) 2 (3.7) 2 (3.7) 3 (5.6) 3 (5.6) | 14 (14.6) 6 (6.3) 6 (6.3) 5 (5.2) 5 (5.2) 5 (5.2) 4 (4.2) 3 (3.1) 3 (3.1) |
| Treatment-related events* (n (%)) |
| Atopic reaction1 Arm numbness Burning sensation Elbow twitch Increased pain2 Myoclonic jerk3 Nausea Thumb tremble Vomiting | 0 (0) 0 (0) 1 (2.4) 0 (0) 1 (2.4) 1 (2.4) 1 (2.4) 0 (0) 1 (2.4) | 1 (1.9) 1 (1.9) 0 (0) 1 (1.9) 0 (0) 0 (0) 0 (0) 1 (1.9) 0 (0) | 1 (1.0) 1 (1.0) 1 (1.0) 1 (1.0) 1 (1.0) 1 (1.0) 1 (1.0) 1 (1.0) 1 (1.0) |
| *All treatment-related adverse events were classified mild, except for atopic reaction, which was classified moderate in intensity. At the completion of the study, the investigator concluded that the reaction was due to the alcohol swabs rather than to the study drug. 1Atopic reaction occurred at the injection site. 2Increased pain occurred back of hand on limb that was injected. 3Myoclonic jerk occurred in biceps and elbow of the arm injected. BoNT-A: botulinum toxin type A; UTI: urinary tract infection. |
DISCUSSION
In this study, intramuscular treatment with BoNT-A for chronic upper-limb spasticity in stroke patients was found to be well-tolerated and efficacious in terms of reducing muscle spasticity and improving the ability to achieve personal functional goals. However, no significant benefit in improving quality of life was demonstrated using the AQoL. Neither was there any consistent difference in reduction of pain or depression. However, these symptoms were not prominent among the study population and the groups were not well matched with respect to pain at baseline. Further study in symptomatic individuals will be required to evaluate the impact of BoNT-A on these parameters.
At a mean age of 59 years, the patients in this study were somewhat younger than the general stroke population, but of similar age to those in other studies of BoNT-A in post-stroke spasticity, which range around a mean age of approximately 60 years (4–8, 25). The mean time since stroke (5.9 years), however, was rather longer than in other trials where this more typically ranges between 2 (25) and 4 (8) years. The observed reduction in muscle spasticity with BoNT-A is well documented in previous studies (5, 6, 7, 24). The lack of improvement in active muscle function (MMAS) was not unexpected given the chronicity of hemiplegia in this study population. Previous studies (5) have similarly shown no concomitant change in motor function.
This study did not demonstrate significant benefits in terms of patient disability or carer burden, although both groups improved, and previous studies have also varied in this respect. Bakheit et al. (5) and Childers et al. (25) also failed to show any functional change using global disability measures, such as the Barthel Index or the Functional Independence Measure. In contrast, Bhakta et al. (22) demonstrated change in disability specifically with respect to the upper limb using the same Patient Disability and Carer Burden scales that were used in this study. A possible reason for the apparent discrepancy is that our study population was relatively independent. Bhakta’s patients had a substantially higher level of disability score at baseline (2.5) compared with our group (1.8) and also a much higher proportion of patients with carers (90% compared with 62%) (22).
However, the primary purpose of this study was to determine whether these effects on spasticity were reflected in a change from the patient’s personal perspective – in particular on quality of life. The AQoL was chosen as the primary end-point in this study as it had been previously validated on a community stroke population in the North East Melbourne Stroke Incidence Study (NEMESIS) study (26–28). The mean baseline AQoL score in this population (mean = 0.35) was similar to that observed in upper-limb impaired stroke patients in the NEMESIS study (mean = 0.38). Also in common with the NEMESIS study, a high proportion of patients in this population (17%) had a baseline AQoL score of less than 0.1, although there was considerable variability. However, no significant improvement in quality of life was demonstrated using this instrument, even with subgroup domain scores and after potential confounders such as baseline AQoL scores and age were taken into consideration.
There are a number of possible reasons for this negative result. Clearly the BoNT-A achieved an effect on spasticity, but either this was insufficient to impact at the level of the person’s well-being, or the tool was too insensitive to detect the impact. Alternatively, other external influences may simultaneously have affected their perception of well-being, so that the effects of relieving spasticity were obscured.
The AQoL is a multifaceted questionnaire addressing a range of issues, including eyesight, hearing, sleep, social relationships and independence. With the benefit of hindsight it was possibly over-ambitious to have expected changes resulting from focal relief of upper limb spasticity to have impacted significantly on these issues, especially in the context of longstanding upper limb dysfunction. Whilst many trial designs incorporate the primary outcome as part of the criteria for recruiting patients, poor quality of life at baseline was not a selection criterion for this study. The mean baseline AQoL of patients in this study was comparable to that measured in a large Australian epidemiological study of post-stroke patients (NEMESIS study) (26–28), and it was a change from this score that was used to calculate the power of the study. However, we failed to predict a very high degree of intra-patient variation in AQoL scores that was observed across the 24-week study period, as illustrated in Figs 2 and 3. In the presence of such variability, it is not surprising that no overall trend was observed. This also suggests that other external factors have much more important influence on day-to-day well-being, as measured by the AQoL. Whilst in the NEMESIS study, the AQoL provided a valid indication of well-being in a community stroke population in comparison with a normal population, its usefulness as a serial measure to compare change related to focal intervention must remain in question. Only one other study has attempted to evaluate the effects of BoNT-A using a global quality of life measure, that of Childers et al. (25), who used the SF-36 and similarly showed no benefit.
A common limitation of all these standardized rating scales is that they assess outcomes that are assumed to be of importance, rather than allowing patients to determine the outcomes that matter to them. This was a relatively able group in which even symptoms such as pain and depression affected only a minority. GAS, on the other hand, provides a measure of achievement of personal goals for treatment. First described by Kiresuk and colleagues (17, 18), it has been used as a method for evaluation of complex interventions in a number of health settings. It has also been used successfully to demonstrate person-centred functional gains in the context of BoNT-A treatment for spasticity (19). In this study, the GAS T scores averaged well below 50, indicating that goals were ambitiously set. Nevertheless, patients treated with BoNT-A showed significantly higher levels of personal goal attainment at week 20 than those treated with placebo. Although their detailed description is beyond the scope of this paper, it was interesting to note that many of the personal goals set would not have been reflected in the standardized measures used in this study (for example the reduction of associated reactions, or the use of the limb for specific bimanual tasks related to hobbies or domestic activities). A secondary analysis of individual goal attainment will be presented separately for publication.
Finally, although in some senses subjective, the perception of overall benefit reported by investigator and patient should not be underestimated as an outcome since it brings together the various factors that treatment was aimed to address in a single global measurement. In this study, both investigators and patients independently rated the overall benefit significantly higher in patients treated with BoNT-A compared with placebo, which underlines the importance of reducing spasticity and improving function at a personal level regardless of performance on standardized scales.
There are a number of recognized limitations to this study:
• Although the randomization process was 1:1 and designed to provide equal numbers in the 2 groups, there were in fact 12 more patients on active treatment than placebo. A confirmation of the validity of the randomization code was provided by Penn Pharmaceutical Services Ltd, so we are unable to explain this apparent failure of randomization. Despite the discrepancy in numbers, the groups were relatively well matched for baseline variables, except with respect to pain levels, which may have accounted for the apparent benefits in pain reduction at week 12.
• The study set out to examine the effects of BoNT-A on quality of life. Although the AQoL was chosen in good faith as a valid tool for the evaluation of personal well-being following stroke, for the reasons described above it was probably not the best choice, being both highly variable within individuals over time (presumably in response to unidentified external factors) and relatively insensitive to the degree of change anticipated from intervention for focal spasticity. We have therefore neither proven nor disproven impact at the level of quality of life.
• For pragmatic reasons and to reflect current practice, follow-up therapy was only provided as part of routine practice. In fact one-third of both groups did not receive such treatment. Failure to capitalize on the effects of muscle relaxation with sustained muscle stretching could have led to a poorer outcome for the BoNT-A group than may have been expected had the recommendations for best practice been fully followed.
In conclusion, intramuscular treatment of upper-limb spasticity with BoNT-A in post-stroke patients, was found to be well-tolerated and efficacious in terms of reducing muscle spasticity and improving the ability to achieve personal functional goals. These benefits were not reflected in a change in quality of life as measured using the AQoL. However, this may relate more to the metrics of the instrument than to a true reflection of person-centred outcome, and further exploration of other tools (such as the GAS) is recommended in this patient population.
Conflicts of interest disclosure
All authors of this publication have made a full and complete disclosure of interests, memberships, relationships, arrangements, investments and holdings that potentially could result in a conflict between the author’s personal, professional or business interests and the integrity of the author and journal (signed copies available on request).
Apart from modest research grant payments provided to the investigators for conducting this study, all other potential conflicts of interest are listed below:
Mc Crory: None; Turner-Stokes: Research Grant from Ipsen – Significant, Advisory Board for Ipsen – Significant, Honoraria – Modest; Baguley: Honoraria – Significant (Ipsen) and Modest (Allergan); De Graaff: Consultant – Modest; Katrak: Honoraria – Modest; Sandanam: None; Davies: None; Munns: Employment – Significant; Hughes: Advisory Board for Ipsen – Modest.
ACKNOWLEDGEMENTS
We would like to thank all the clinical staff and patients who took part in this study.
The research discussed in this study was funded fully by Ipsen Pty Ltd, Australia, who had no influence on the interpretation of data and the final conclusions drawn.
Statistical support was provided by Peter Mullins of Sage Consulting, New Zealand.
REFERENCES