OBJECTIVE: To investigate symptoms and functional impairment in women with secondary lymphoedema of the breast following surgical treatment and to assess the therapeutic benefit of treatment with low-intensity and extremely low-frequency electrostatic fields (Deep Oscillation®), supplementing manual lymphatic drainage.
METHODS: Twenty-one patients were randomized either to the treatment group (n = 11): 12 sessions of manual lymphatic drainage supplemented by Deep Oscillation®, or to the control group (n = 10): manual lymphatic drainage alone. Assessment included subjective pain and swelling evaluation, range of motion of the shoulder and the cervical spine, and analysis of breast volume using a 3D measuring system.
RESULTS: Patients had high pain and swelling scores at baseline. Shoulder mobility was impaired in all patients; restriction of cervical spine mobility was common at baseline and declined further in the control group. Deep Oscillation® resulted in significant pain reduction in the treatment group. The subjective reported reduction of swelling in both groups was confirmed objectively by 3D measurement only in the treatment group.
CONCLUSION: Additional Deep Oscillation® supplementary to manual lymphatic drainage can significantly enhance pain alleviation and swelling reduction in patients with secondary breast lymphoedema compared with manual lymphatic drainage alone.
Key words: breast cancer, deep oscillation therapy, lymphatic drainage, pain, swelling.
J Rehabil Med 2008; 40: 645–650
Correspondence address: Anett Reisshauer, Department of Physical Medicine and Rehabilitation Charité, Universitätsmedizin Berlin, Charitéplatz 1, DE-10117 Berlin, Germany. E-mail: anett.reisshauer@charite.de
Submitted July 4, 2007; accepted April 2, 2008
Introduction
Many patients with breast cancer develop secondary lymphoedema of the arm, chest and breast after cancer treatment. The reported incidence proportion of arm lymphoedema after mastectomy with subsequent radiotherapy is 0–54% (1). This wide variation can be explained by the use of different definitions, measuring techniques and study designs. From our clinical experience we presume that more women will develop secondary lymphoedema in the future as more breast-sparing operations are performed. The lack of reliable data is attributable to the facts that no generally accepted definition of secondary lymphoedema exists and that no definite diagnostic criteria are available. For instance, Meric et al. (2) reported that 28.9% of patients who underwent breast-sparing surgery developed breast fibrosis defined as thickening or fibrosis of the skin. Goffmann et al. (3) identified breast lymphoedema in 9.6% of their patients. They defined breast lymphoedema as swelling and orange-peel of the breast combined with a sensation of heaviness and pain. The largest study included 160 patients and was performed by Rönkä et al. (4). The authors reported breast swelling in 34% of the patients, orange-peel in 3.8%, and tenderness on palpation of the breast in 59% of cases. The risk of developing breast lymphoedema increases with the severity of axillary trauma (number of removed lymph nodes, radiation dose) (3–5). While lymphoedema of benign origin affecting the arms or legs causes almost no pain, patients with chronic secondary breast lymphoedema may experience severe pain and discomfort and considerable impairment of their quality of life. Velanovich & Szymanski (6) investigated quality of life in patients with secondary arm lymphoedema and concluded that better therapy of lymphoedema is needed urgently to ensure an adequate quality of life in breast cancer patients. Diagnostic evaluation usually focuses on arm lymphoedema, while breast lymphoedema does not receive the attention it deserves. The extent of arm lymphoedema can be reproducibly quantified by manual measurement of arm circumference or optoelectronic volume and circumference measurement (7). The only objective diagnostic parameter available so far for assessing the severity of breast lymphoedema is change of skinfold thickness. This parameter, however, is poorly reproducible and unreliable in monitoring treatment outcome.
Lymphoedema is treated by complex physical decongestive therapy comprising manual lymphatic drainage, compression therapy, skin care, and remedial exercises. In a systematic review of lymphoedema secondary to breast cancer Kligman et al. (8) concluded that there is some evidence suggesting that compression therapy and manual lymphatic drainage can reduce the extent of lymphoedema, but that further studies are needed to corroborate the benefits of these therapeutic approaches. A study conducted by our group demonstrated significantly increasing transcutaneous oxygen partial pressure in patients treated by manual lymphatic drainage for arm and leg lymphoedema (9). Because compression therapy is not possible in patients with breast lymphoedema, there is a need for alternative therapeutic approaches. In a study performed by Bertelli et al. (10), electrically stimulated lymphatic drainage, however, did not lead to an additional benefit when compared with compression alone. In contrast, Schönfelder & Berg (11) reported pain alleviation and improved arm function in patients with breast lymphoedema treated by Deep Oscillation®. The aim of Deep Oscillation® is to stimulate the flow of lymph and reduce swelling.
The study presented here had 2 aims: to evaluate the symptoms (swelling, pain) and functional limitations (reduction of range of movement of shoulder and cervical spine) of patients with secondary breast lymphoedema and to assess the additional therapeutic benefit of Deep Oscillation® when combined with manual lymphatic drainage.
Methods
Subjects
Patients were recruited for the study by means of bulletins, articles in the print media, and public lectures for patients and physicians. The diagnosis of breast lymphoedema was established by palpation of a pasty oedema in the affected breast in comparison with the contralateral side by the same experienced examiner throughout the study. Inclusion criteria were: age range 18–80 years, breast lymphoedema, at least 6 weeks since last irradiation, updated documentation of aftercare, patient living near study centre. Exclusion criteria were: Deep Oscillation®
treatment in the 3 months preceding the study, acute inflammation, acute thrombosis, heart disease, electronic implant, pregnancy, subjective sensitivity to electrical fields. Twenty-two subjects who fulfilled the inclusion criteria and had no exclusion criteria consented to participate in the study. Block randomization (using a list of A-B-C generated by a random-number generator with a size of 6 per block) was used to assign the patients to the treatment group (manual lymphatic drainage with additional Deep Oscillation®) or to the control group (lymphatic drainage alone). One patient discontinued the study due to recurrent breast cancer diagnosed during the study time.
The patients had a mean age of 59.2 (range 41–71) years. All women included in the study had secondary lymphoedema in the breast region; 21 after breast-sparing surgery for cancer and one after removal of a melanoma from the upper arm with axillary lymph node dissection. The left breast was operated in 9 patients and the right breast in 12 patients. The average time since surgery was 4 years and one month. All patients had adjuvant radiotherapy (4 month to 5 years ago) (Table I). Shoulder mobility was impaired in all patients (Table II). The median pain severity score of all patients in both groups was 4.9 on a visual analogue scale (VAS) from 1 to 10; the score for swelling was 5.9. There were no significant differences between the 2 groups in terms of these baseline parameters (see Tables III and IV).
| Table I. Group characteristics |
| | Treatment group | Control group |
| Age, years, mean (range) | 56.6 (41– 65) | 62.0 (42–71) |
| Months after OP, mean (range) | 47.6 (9–110) | 50.8 (9–142) |
| Affected breast | 5 right, 6 left | 4 right, 6 left |
| Years after radiotherapy, mean (range) | 3.5 (0.5–12) | 3.8 (0.5–12) |
| OP: operation. |
The study was approved by the ethics committee of the Hospital Charité – Universitätsmedizin Berlin (reference EA 1/028/05).
Study parameters
The study patients were examined before and after the 4-week course of therapy and again 8 weeks after the end of treatment. The patients subjectively assessed pain, breast swelling and the effectiveness of lymphoedema treatment on a 10-point VAS. In addition, breast lymphoedema was documented on photographs and functional tests were employed to assess the range of motion of the shoulder and cervical spine. Shoulder mobility of the affected side compared with the contralateral side was measured using the neutral-zero method for passive range of motion (12) (normal values see Table II). Active cervical spine mobility was tested using the Zebris ultrasound-based movement sensor (Zebris, Medizintechnik GmbH, Isny, Germany) (13). Limits for normal movement were: Lateral Flexion 45°, Rotation 60°, Anteflexion 40°, Retroflexion 30°. The extension capacity of the pectoral muscle was determined using Janda’s muscle function test (14).
ScanMobile (GFaI, Berlin, Germany) served as a mobile 3D measuring system for simple and rapid determination of the body surface area in the target region (15, 16). With this device, the breast surface was scanned at high resolution (about 0.3 mm) in 1.3 sec. Testing of differences through expiration, performed before the study, showed a maximum volume difference of 28 ml through the different phases of expiration. Therefore, much attention was paid to testing in resting expiratory position. The scanned dot clouds served to interpolate a closed surface (triangulation) for computation of distances and difference volumes. The data-sets acquired at different time-points were matched using the iterative closest-point technique of the Final Surface software. After matching of the data-sets, a region for computing the difference volume was selected on the surface of 1 data-set (master) and the volume between this surface and that of a second data-set was then determined for this region. Thirty measurements with the same object showed a maximal difference of 0.5 mm; mean 0.2 mm in each direction corresponding to a total of only 0.125 ml.
Treatment
The patients assigned to the treatment group underwent a 4-week course of 12 sessions of manual lymphatic drainage supplemented by Deep Oscillation®. Oscillations were performed with the Hivamat (Physiomed Elektromedizin AG, Schnaittach/Laipersdorf, Germany). After the 4-week course of combined treatment, the patients returned to manual lymphatic drainage alone for the next 8 weeks. The patients in the control group continued routine manual lymphatic drainage therapy (usual care as paid for by the national health insurance) consisting of 1 or 2 of 30–45-min sessions per week, by the therapist who treated the patient prior to the study.
Deep Oscillation® is a therapeutic approach that consists in applying an intermittent electrostatic field of low intensity (U = 100–400V; I = 150µA) and extremely low frequency (30–200Hz, rectangular, bi-phase) to the target area. The field electrostatically attracts and releases the patient’s tissue in the selected frequency, resulting in deep and lasting resonance vibration (Fig. 1). Both the patient and the therapist are connected to the Deep Oscillation® device, which serves as a source of tension with high internal resistance. A special glove serves as an insulator. The impulse of the voltage induces an electrostatic force of attraction on the tissue and leads to a higher force of friction while massaging the oedema. Each treatment session lasted for 1 hour, starting with a 15-min pre-treatment period of conventional manual lymphatic drainage, followed by Deep Oscillation®-assisted lymphatic drainage of the breast and chest wall and including the arm in patients with accompanying arm oedema. Oscillations were performed at 100 Hz
for 30 min and then at 30 Hz for 15 min.
Fig. 1. Diagram of manual lymphatic drainage assisted by Deep Oscillation®.
Statistics
Statistical analysis was performed using SPSS for Windows, version 14.0. The non-parametric Mann-Whitney U test was used for group analysis. The Friedmann-test was applied for analysing changes over time, Wilcoxon-test for changes between 2 time-points in one group. In addition, the χ2 test was used. Significance was set at 0.05.
Results
Evaluation of symptoms at baseline
Visual analogue scales. The patients in both groups taken together had a median baseline pain score of 4.9. Swelling at baseline was assigned a score of 5.9 by the patients.
Mobility of the shoulder and cervical spine. Mobility of the shoulder was impaired in all patients at baseline. The range of motion was markedly poorer on the affected side (see Table II).
| Table II. Numbers of patients with limited range of motion of the shoulder for the movements tested at baseline |
| Movement (all 21 patients) | Normal range | Ipsilateral mean | Ipsilateral, n with limited range | Contralateral mean | Contralateral, n with limited range |
| External rotation with 90° abduction | 70° | 75.9° | 7 | 83.6° | 1 |
| Internal rotation with 90° abduction | 70° | 58.9° | 15 | 65.9° | 8 |
| External rotation | 60° | 61.1° | 0 | 70.9° | 0 |
| Internal rotation | 95° | 58.2° | 20 | 62.3° | 20 |
| Forward flexion | 170° | 140.2° | 14 | 157.5° | 5 |
| Extension | 40° | 40.0° | 10 | 48.6° | 1 |
| Abduction | 180° | 132.5° | 16 | 159.3° | 6 |
| Adduction | 40° | 13.7° | 11 | 18.25° | 8 |
| Abduction without scapula | 90° | 71.7° | 20 | 80.6° | 17 |
In the initial examination at baseline, the patients in general showed good forward flexion and extension of the cervical spine. These movements were limited only in 2 (flexion) and 5 (extension) patients, respectively. Rotation and lateral flexion of the cervical spine were found to be reduced at baseline but without a significant side-to-side difference. Rotation toward the affected side was limited in 11 patients and toward the unaffected side in 12 patients. Lateral flexion to the affected side was reduced in 17 and to the unaffected side in 20 patients.
The extension capacity of the pectoral muscle at baseline was reduced on the affected side in 15 patients and on the contralateral side in one patient.
Results of intervention
Visual analogue scales. The pain scores were unaffected by treatment in the control group. In the patients treated by additional Deep Oscillation®, the pain scores decreased significantly from 4.0 before therapy to 2.1 (end of therapy) and to 2.0 (at 8-week follow-up). The pain score at the end of therapy but not at follow-up showed between both groups a significant better outcome in favour for the intervention group (Table III). Considering the differences in the pain score reached after therapy and at follow-up no significant change could be found.
| Table III. Visual analogue scale (VAS) pain scores assigned to breast lymphoedema at the 3 time-points: T1 = baseline at start of therapy, T2 = at end of therapy after 4 weeks, T3 = 8-week follow-up: median (quartile 25–75) |
| | T1 | T2 | T3 | p-value/ Friedman | Difference T2–T1 | Difference T3–T1 |
| Control group, n = 10 | 5.0 (4.0 to 6.0) | 4.8 (3.8 to 6.5) | 4.6 (1.9 to 6.5) | 1.000 | –0.5 (–1.2 to 1.3) | 0.2 (–1.5 to 0.9) |
| Treatment group, n = 11 | 4.0 (2.0 to 5.0) | 2.1 (1.0 to 4.0) | 2.0 (1.0 to 3.0) | 0.048 | –1.0 (–2.0 to 0) | –1.6 (–2.3 to 0) |
| p-value / Whitney U test | 0.284 | 0.031 | 0.104 | | 0.173 | 0.282 |
Patients’ subjective assessment in both groups showed a significant reduction in swelling (control group at follow-up, intervention group after treatment). At the end of therapy, there was only a tendency toward better therapy results in the group with additional oscillation (Table IV). Also there was a significant change between both groups for differences in VAS for swelling after therapy.
| Table IV. Visual analogue scale (VAS) swelling scores assigned to the affected breast at the 3 time-points: T1 = baseline at start of therapy, T2 = at end of therapy after 4 weeks, T3 = 8-week follow-up: median (quartile 25–75) |
| | T1 | T2 | T3 | p-value / Friedman | Difference T2–T1 | Difference T3–T1 |
| Control group (n = 10) | 5.2 (3.7–7.1) | 5.5 (3.8–8.4) | 4.5 (2.5–5.3) | 0.023 | –0.1 (–1.1–1.9) | –1.9 (–2.7– –0.2) |
| Treatment group (n = 11) | 6.0 (5.0–7.9) | 4.0 (2.7–6.0) | 3.4 (3.0–5.9) | 0.046 | –2.0 (–3.2– –1.4) | –2.0 (–4.0–0) |
| p-value / U test | 0.481 | 0.090 | 0.919 | | 0.020 | 0.512 |
The effectiveness of therapy (conventional lymphatic drainage) before inclusion in the study was not rated as “good” by the patients. The corresponding VAS score (with highest values for optimum outcome) was only 3.1. In comparison, the scores assigned to the effectiveness of combined Deep Oscillation® and lymphatic drainage were 6.0 immediately after treatment and 6.9 at 8-week follow-up. The scores in the control group were markedly lower (3.0 and 4.0, respectively). However, the differences between the groups did not reach statistical significance.
Subjective satisfaction with Deep Oscillation® as a supplement to manual lymphatic drainage was very high (score of 8.9). None of the patients reported adverse events.
Mobility of the shoulder and cervical spine. The further course at follow-up showed significant reduction in shoulder mobility in the control group for internal rotation on the affected side (from 63° to 50°, p = 0.015) and of abduction on the unaffected side (from 163° to 150°, p = 0.039). The range of mobility in the intervention group was unchanged (p > 0.30).
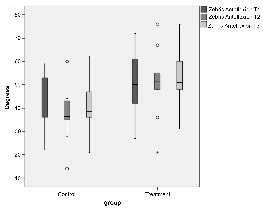
Forward flexion of the cervical spine after treatment showed a significant difference between the 2 groups: the mobility was poorer in controls and slightly improved in the oscillation treatment group (see Fig. 2). The situation was similar for rotation toward the contralateral side (intervention group: 62° at baseline to 61° at follow-up; control group: 54° to 41° respectively; p = 0.025 between groups at follow-up). Lateral flexion toward the affected side nearly reached significance level p = 0.053 at follow-up between groups (intervention group: 37° to 36°; controls: 32° to 30°).
Fig. 2. Forward flexion of the cervical spine at the 3 time-points: T1 = start of therapy, T2 = end of 4-week therapy, and T3 = 8-week follow-up (12 weeks after start of therapy); U test between groups: T1 p = 0.240; T2 p = 0.026; T3 p = 0.023, Whitney U test difference T2–T1 p = 0.240; T3–T1 p = 0.037. Outliers are defined by 1.5–3 times interquartile-distance; extreme values are defined by more than 3 times interquartile-distance.
However, besides forward flexion no significant change from baseline between the groups was found for shoulder or cervical spine mobility.
Extension capacity of the pectoral muscle. There was no significant change after treatment in both intervention and the control group, only a tendency towards less impairment on the ipsilateral side in the intervention group.
Volume measurement. Volume determination using the 3D measuring technique described above objectified a reduction of swelling after the intervention interval in the treatment group of –15.7 ml (–35.2 to –6.1 ml) in contrast to an increased volume in the control group of 13.3 ml (–3.6 to 26.3 ml) p = 0.007. At follow-up, the difference lost statistical significance: treatment group –15.4 ml (–36.4 to –1.2 ml), control group –1.1 ml (–16.9 to 7.0 ml) p = 0.191.
Discussion
Until now, little attention has been paid to breast lymphoedema as an adverse effect of breast-sparing surgery in women with breast cancer. Schönfelder & Berg (11) listed breast lymphoedema as only one of the many conditions they subsumed under skin changes while other investigators ignored it altogether (1, 6). A review article (17) depicted the remarks of woman that their treating physicians knew too little about lymphoedema.
Affected patients suffer severely from the swelling-associated pain, and therefore seek treatment urgently; a fact that is also reflected by their voluntary participation in our time-consuming study. With a value of 4.9, the initial median pain score was rather high in our study. To our knowledge no studies exist about the impact of breast oedema on pain. Velanovich & Szymansky (6) reported a significantly higher proportion of patients complaining about pain after breast cancer operations when they had lymphoedema of the arm. Schönfelder & Berg (11) reported that 37% of woman after breast-sparing operation and radiation complained about pain and hyperaesthesia in the breast but without differentiating between women with or without breast oedema. Normally only the arm volume is used as a parameter in studies dealing with lymphoedema. Therefore, little is known about the symptom pain in woman with breast oedema and its treatment.
One of the important symptoms in our study population is the pronounced restriction of cervical spine mobility, most notably rotation and lateral flexion. Shoulder mobility was impaired in all patients in our study, while the incidence reported in the literature ranges from 4.8% to 35% (2, 11, 18). The much higher incidence in our patient population might be attributable to the use of different reference values and test methods. However, it is also conceivable that the axial trauma causing breast lymphoedema was more severe in our study population. The extension capacity of the pectoral muscle was reduced in the majority of our patients. Lee et al. (19) attributed the reduced length to fibrosis of this muscle, but they did not find a correlation between the degree of reduction of shoulder mobility and the symptoms reported by their patients. Neither the stretching programme used by Lee et al. (19) nor our therapeutic approach had any appreciable beneficial effect on pectoral muscle function. However, it is possible that our measuring technique was too unspecific to register any therapeutic effect on the pectoral muscle.
Our new approach to measuring volume changes of the breast using 3D surface scanning is a promising technique for evaluating therapeutic effects on lymphoedema or other conditions accompanied by swelling. This method is fast, easy and has no side-effects. To minimize the influence of movement, we standardized the patient’s lying and breathing position. However, the results should nevertheless be treated with caution. To our knowledge, until now 3D surface scanning has been used only for measuring inanimate objects. Thus, an evaluation of its reliability and validity when applied to humans is required.
The patients included in this study were not satisfied with the results of treatment they had received before study participation. The results presented here show that the patients who underwent manual lymphatic drainage supported by Deep
Oscillation® experienced a reduction in pain and swelling and were highly satisfied with the outcome. The reduced mobility of the cervical spine and shoulder did not deteriorate further; which is in strong contrast to the situation in the control group.
To our knowledge, there are no other studies that systematically investigate breast lymphoedema therapy. In a study of the incidence of breast and arm lymphoedema, Goffman et al. (3) merely mentioned that good results were achieved with manual lymphatic drainage in 21 of 23 patients with breast lymphoedema. More data are available on physical therapy in patients with lymphoedema of the arm after breast cancer treatment (7, 8, 20). However, these results are not directly applicable to the treatment of breast lymphoedema because compression bandaging is not possible for this condition.
We could identify only 2 other in vivo studies with deep oscillation. Schönfelder & Berg (11) applied manual lymphatic drainage assisted by Deep Oscillation® in all breast cancer patients who underwent breast-sparing surgery with subsequent radiotherapy, regardless of whether they had breast lymphoedema. In an uncontrolled study performed by Gasbarro et al. (21) this combined approach was shown to significantly reduce oedema volume and the thickness of the subcutaneous layer in patients with leg lymphoedema.
The positive effect seen in the intervention group might be explained partly by the placebo effect due to the use of a new technique. Unfortunately sham oscillation therapy was not possible. Also was the number of treatment sessions per week higher in the treatment group with 2–3 sessions per week compared to control group with only 1–2 sessions per week. However, to date no attempt has been made to investigate the influence of frequency of manual lymphatic drainage on outcome.
We assume that the therapeutic benefit of manual lymphatic drainage assisted by a treatment with low-intensity and extremely low-frequency electrostatic fields (Deep Oscillation) may be attributable to a combination of different effects: stimulation of lymphatic flow, reduction in muscle tone and alleviation of pain by means of mechanical stimulation of pain receptors. However, these mechanisms have to be verified. Up to now, in vitro experiments have only been performed to demonstrate immunostimulating and antioxidative effects of Deep Oscillation® (22, 23).
The results of the pilot study presented here show that Deep Oscillation® performed in addition to manual lymphatic drainage in patients with secondary lymphoedema of the breast substantially improves outcome in terms of alleviation of pain, mobility, and lymphoedema volume reduction compared with manual lymphatic drainage alone. We therefore conclude that more attention should be paid to patients with breast lymphoedema and that treatment with low-intensity and extremely low-frequency electrostatic fields (Deep Oscillation®) could be a useful supplementary therapy in the management of patients with breast lymphoedema.
Acknowledgement
The study was supported by grants from Physiomed Elektromedizin AG, Schnaittach/Laipersdorf, Germany.
References
1. Hinrichs CS, Watroba NL, Rezaishiraz H, Giese W, Hurd T, Fassl K, et al. Lymphedma secondary to postmastectomy radiation: incidence and risk factors. Ann Surg Oncol 2004; 11: 573–580.
2. Meric, FM, Buchholz TA, Mirza NQ, Vlastos G, Ames FC, Ross MI, et al. Long-term complications associated with breast-conservation surgery and radiotherapy. Ann Surg Oncol 2002; 9: 543–549.
3. Goffman T E, Laronga C, Wilson L, Elkins D. Lymphedema of the arm and breast in irradiated breast cancer patients: risks in an ear of dramatically changing axillary surgery. Breast J 2004; 10: 405–411.
4. Rönkä RH, Pamilo MS, von Smitten KAJ, Leidenius. Breast lymphedema after breast conserving treatment. Acta Oncologica 2004; 43: 551–557.
5. Clarke D, Martinez A, Cox RS, Goffinet DR. Breast edema following staging axillary node dissection in patient with breast carcinoma treated by radical radiotherapy. Cancer 1982; 49: 2295–2299.
6. Velanovich V, Szymansky W. Quality of life of breast cancer patients with lymphedema. Am J Surg 1999; 3: 184–188.
7. Didem K, Ufuk Y, Serdar S, Zümre A. The comparison of two different physiotherapy methods in treatment of lymphedema after breast surgery. Breast Cancer Res Treat 2005; 93: 49–54.
8. Kligman L, Wong RKS, Johnston M, Laetsch NS. The treatment of lymphedema related to breast cancer; a systematic review and evidence summary. Supp Care Cancer 2004; 12: 421–431.
9. Reißhauer A, Bischoff A, Mathiske-Schmidt K. Transcutaneous oxygen partial pressure before and after manual lymphatic drainage in patients with chronic lymphedema of the limbs. Eur J Lymphol 2004; 40: 13–16.
10. Bertelli G, Venturini M, Forno G, Macchiavello F, Dini D. Conservative treatment of postmastectomy lymphedema: a controlled, randomized trial. Ann Oncol 1991; 2: 575–578.
11. Schönfelder G, Berg D. Nebenwirkungen nach brusterhaltender Therapie des Mammakarzinoms. Gynakol prax 1991; 15: 109–122.
12. Terwee CB, Winter AF, Scholten RJ, Jans MP, Deville W, van Schaardenburg D, Bouter LM. Interobserver reproducibility of the visual estimation of range of motion of the shoulder. Arch Phys Med Rehabil 2005; 86: 1356–1361.
13. Dvir Z, Prushansky T. Reproducibility and instrument validity of a new ultrasonography-based system for measuring cervical spine kinematics. Clin Biomech 2000; 15: 658–664.
14. Janda V, editor. Manuelle Muskelfunktionsdiagnostik. Munich: Urban und Fischer Verlag München, Jena; 2000.
15. Luhmann T, editor. Nahbereichsphotogrammetrie – Grundlagen, Methoden und Anwendungen. Heidelberg: Herbert Wichmann Verlag Heidelberg; 2000.
16. Sasse R, editor. Bestimmung von Entfernungsbildern durch aktive stereoskopische Verfahren; Fortschritte der Robotik, vol 23. Braunschweig/Wiesbaden: Vieweg&Sohn Verlagsgesellschaft mbH Braunschweig/Wiesbaden; 1994.
17. Johansson K, Holmstrom H, Nilsson I, Ingvar C, Albertsson M, Ekdahl C. Breast cancer patients’ experiences of lymphoedema. Scand J Caring Sci 2003; 17: 35–42.
18. Sugden EM, Rezvani M, Harrison JM, Hughes LK. Shoulder movement after the treatment of early stage breast cancer. Clin Oncol (R Coll Radiol) 1998; 10: 173–181.
19. Lee TS, Kilbreath SL, Refshauge KM, Pendlebury SC, Beith JM, Lee MJ. Pectoral stretching program for women undergoing radiotherapy for breast cancer. Breast Cancer Res Treat 2007; 102: 313–321.
20. Mirolo BR, Bunce IH, Chapman M, Olsen T, Eliadis P, Hennessy JM, et al. Psychosocial benefits of postmastectomy lymphedema therapy. Cancer Nurs 1995; 18: 197–205.
21. Gasbarro V, Bartoletti R, Tsolaki E, Sileno S, Agnati M, Conti M, et al. Role of Hivamat (Deep Oscillation®) in the treatment for the lymphedema of the limbs. Eur J Lymphol 2006; 16: 13–15.
22. Mikhalchik E, Titkova S, Anurov M, Suprun M, Ivanova A, Trakhtman I, Reinhold, J. Effects on blood parameters of Deep Oscillation®. 1st International Conference on Skin and Environment, Moscow-St. Petersburg, 2005; June 1–6: 59.
23. Titkova S, Anurov M, Suprun M, Ivanova A, Trakhtman I, Reinhold J. Wound healing effects of Deep Oscillation®. 1st International Conference on Skin and Environment, Moscow-St. Petersburg, 2005; June 1–6: 71.