OBJECTIVE: To identify independent predictors of depressive symptoms in a cohort of patients with coronary artery disease entering cardiac rehabilitation.
DESIGN: Cross-sectional cohort study.
Patients and methods: Consecutive patients entering a cardiac rehabilitation and secondary prevention programme underwent screening for depressive symptoms using the Center for Epidemiological Studies Depression (CES-D) scale and cardiopulmonary fitness testing to quantify peak oxygen consumption.
RESULTS: Of the 366 patients with coronary artery disease, 22.3% reported at least mild (CES-D ≥ 16) and 10.4% reported significant (CES-D ≥ 23) depressive symptoms. Antidepressant medications were being used by 6.3% of patients. Sociodemographic, cardiopulmonary and cardiac characteristics, and medical co-morbidities previously associated with depression accounted for 14.7% of the variance in a multiple linear regression model (F = 8.713, p < 0.001) predicting CES-D scores. Significant independent predictors of CES-D scores were lower peak oxygen consumption, younger age, female sex, lower maximum diastolic blood pressure, angina pectoris and antidepressant use.
CONCLUSION: Reduced physical fitness, younger age, female sex and ischaemic symptoms of coronary artery disease predict higher depressive symptoms in patients entering cardiac rehabilitation.
Key words: depression, coronary disease, exercise test, rehabilitation, physical fitness.
J Rehabil Med 2008; 40: 213–218
Correspondence address: Krista Lanctôt, Sunnybrook Health Sciences Centre, 2075 Bayview Avenue, Suite FG05, Toronto, Ontario, Canada. E-mail: Krista.Lanctot@sunnybrook.ca
Submitted April 27, 2007; accepted October 16, 2007
INTRODUCTION
Coronary artery disease (CAD) is a leading cause of disability, mortality and healthcare utilization in developed countries. In Canada, one in 4 men and one in 5 women suffer from some form of heart disease. Depressive symptoms are common in persons with CAD, and they increase the risk of mortality independently of medical risk factors (1) and interfere with cardiac rehabilitation (2).
Despite demonstrated correlations between depressive symptoms and low activity levels (3), relatively few investigations have focused on the association between depressive symptoms and cardiopulmonary fitness in patients with CAD (4). Since a primary focus of rehabilitation is to improve cardiopulmonary fitness, the interaction between fitness and depressive symptoms may be of substantial clinical importance. In addition, CAD is associated with multiple co-morbid medical conditions and risk factors. Hypertension, diabetes, chronic obstructive pulmonary disease (COPD), and symptoms of CAD such as angina pectoris have been associated with an increased prevalence of depression (5–8), and medications used in the management of these conditions, such as β-blockers (9) and statins (10), have been suggested to interact with depressive symptoms.
The goal of this study was to measure the prevalence of depressive symptoms and identify independent predictors of depressive symptoms in patients with stable CAD entering cardiac rehabilitation. Knowing predictors may be useful to clinicians and they can be used to inform future studies designed to understand the aetiology of mood complications in this population. The use of psychotropic medications was also characterized in this population. These findings may help to establish strategies to better manage depressive symptoms associated with CAD within the setting of cardiac rehabilitation and to aid in the optimization of rehabilitation for patients suffering depressive symptoms.
METHODS
Setting
This was a cohort study of 366 patients selected from the Toronto Rehabilitation Institute Cardiac Rehabilitation and Secondary Prevention Program (TRI-Cardiac). The TRI-Cardiac is a university-affiliated rehabilitation facility serving an urban population with a catchment area of approximately 2.5 million. Patients recovering from various manifestations of CAD and those with significant cardiac risk factors, various forms of congenital heart defects or diabetes are accepted at the TRI-Cardiac. Enrolment is based on physician referral only. All participants provide written informed consent for their clinical data to be used in research investigations in accordance with the Research Ethics Board.
Subjects
Patient demographics, cardiac and other medical morbidities, medical history, cardiopulmonary fitness testing data and concurrent medications were obtained by electronic chart review. A clinical cohort of patients with CAD entering rehabilitation was defined based on a history of myocardial infarction (MI), coronary angiographic evidence of ≥ 50% blockage in at least one major coronary artery, or prior revascularization, such as percutaneous coronary intervention (PCI) procedures or coronary artery bypass graft (CABG) surgery at the time of enrolment at the TRI-Cardiac (Fig. 1). All subjects entered rehabilitation a minimum of 6–8 weeks post-CABG (6 weeks for uncomplicated CABG), a minimum of 6 weeks post-MI, or a minimum of 3 weeks post-PCI. Patients were excluded if they did not perform cardiopulmonary fitness testing, if they were unable to complete the Center for Epidemiological Studies Depression (CES-D) scale, or if their records were otherwise incomplete.
Assessments
Depressive symptoms. All patients were screened for depressive symptoms using the CES-D scale. The CES-D scale has been used extensively in CAD populations (9). The concurrent validity, construct validity and reliability of the CES-D have also been established in community samples (11). In addition, a cut-off score of 16 on the CES-D has demonstrated high sensitivity (100%) and specificity (88%) for detecting Diagnostic and Statistical Manual of Mental Disorders, Third Edition, Revised (DSM-IIIR) diagnosed depression in older subjects (12) and an adequate sensitivity (60% major depression and 71% minor depression) and specificity (83% and 83%, respectively) in community samples (13). Importantly, increasing rates of medical illnesses are not related to the number of false positives using the CES-D (12).
Cardiopulmonary fitness. As part of standard clinical protocol, a cardiopulmonary fitness assessment was carried out. Clinical assessment was performed under medical supervision by a multidisciplinary team including trained exercise physiologists. The exercise stress test, as previously described (14), was used to measure cardiopulmonary responses to increasing intensities of exercise. Measures included the anaerobic threshold, peak heart rate, maximum blood pressure and peak oxygen consumption (VO2 peak). The VO2 peak is the most reliable and reproducible indicator of cardiopulmonary fitness (15). Resting physiological measurements, including heart rate and blood pressure, and anthropometrics including height, mass, girth, and percentage body fat, were recorded. Body mass index (BMI) and waist-to-height ratio (WHtR) were calculated as per standard definitions. Since body mass is a composite of fat and muscle masses, and reduced ratios of muscle to fat have been previously documented in the depressed elderly (16), BMI may be insufficient as an index of body composition. WHtR is an index of relative central adiposity, which does not rely on the measurement of body mass and may therefore demonstrate superiority as a risk factor.
Statistical analysis. Descriptive statistics were calculated for all variables of interest. Continuous measures, such as age, were summarized using means and standard deviations (SD), whereas categorical measures were summarized using counts and percentages.
To determine which clinical variables were the most significant independent predictors of depressive symptoms, a backward elimination linear regression model was performed. Variables associated with depression or depressive symptoms in previous studies of patients with CAD were entered into the model including the sociodemographic characteristics age, sex, smoking status and marital status, the anthropometric characteristics BMI and WHtR, resting physiology including heart rate, systolic and diastolic blood pressure, and cardiopulmonary fitness parameters, including VO2 peak, maximum heart rate, and maximum systolic and diastolic blood pressures (4–8, 17–20). Cardiac factors, including history of MI, PCI, CABG, congestive heart failure and cardiac arrest, and the presence of hypertension and angina pectoris, and the medical co-morbidities diabetes, COPD, cancer, renal disease and stroke that have been previously associated with depression or depressive symptoms were also entered (5, 21). Concurrent use of statins (10), β-blockers (9), antidepressant or anxiolytic medications that may affect depressive symptoms was also entered into the model. Parameters below 5% significance were omitted until parameters could no longer be rejected (exit criterion p > 0.05). All analyses were two-tailed with results considered significant at p < 0.05. All analyses were performed using SPSS statistical software (version 13.0, SPSS Inc., Chicago, IL, USA).
RESULTS
Patient characteristics and incidence of depressive symptoms
The sociodemographic characteristics of patients recruited into this study are summarized in Table I.
| Table I. Patient characteristics (n = 366) |
| | Prevalence (%) |
| Sociodemographic |
| Age (years) | 63.6 (SD 10.7) |
| Employment | 128 (34.9) |
| Unmarried/divorced/widowed | 105 (28.7) |
| Sex (female) | 88 (24.0) |
| Current smoking status | 21 (5.7) |
| Cardiac factors and diagnoses |
| Myocardial infarction | 206 (56.2) |
| PCI | 175 (47.8) |
| Hypertension | 170 (46.4) |
| CABG | 136 (37.2) |
| Valvular heart disease | 30 (8.2) |
| Angina pectoris | 29 (7.9) |
| Conduction deficit | 29 (7.9) |
| Congestive heart failure | 23 (6.3) |
| Cardiac arrest | 14 (3.8) |
| Pacemaker | 9 (2.4) |
| Medical co-morbidities |
| Diabetes | 103 (28.1) |
| COPD | 27 (7.4) |
| Cancer | 22 (6.0) |
| Renal disease | 18 (4.9) |
| Stroke | 15 (4.1) |
| Peripheral vascular disease | 14 (3.8) |
| Concomitant medications |
| Statins | 335 (91.5) |
| ß-blockers | 301 (82.2) |
| Nitroglycerin | 151 (41.3) |
| Anti-diabetic agents | 77 (21.0) |
| Ca2+-channel antagonists | 68 (18.6) |
| Anti-asthmatic agents | 29 (7.9) |
| Psychotropic medications |
| Anxiolytics | 34 (9.3) |
| Antidepressants | 24 (6.3) |
| Other classes | 5 (1.4) |
| PCI: percutaneous coronary intervention; CABG: coronary artery bypass graft; COPD: chronic obstructive pulmonary disease. |
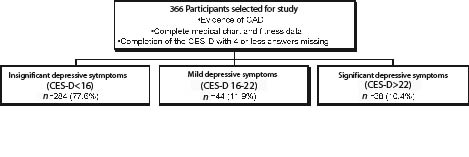
Between September 2004 and June 2005, there were 366 patients enrolled. Seventy-six percent were male and 71% were married. The mean age of patients was 63.6 (SD 10.7) years. Of all patients, 22.3% (n = 82) showed at least mild (CES-D ≥ 16) and 10.4% (n = 38) showed significant (CES-D ≥ 23) depressive symptoms (Fig. 1).
Fig. 1. Definition of cohort with coronary artery disease (CAD). CES-D: Center for Epidemiological Studies Depression Scale.
Only 6.3% of patients (n = 23) were using antidepressant medication and 9.3% (n = 34) were using an anxiolytic. The most common medications included selective serotonin reuptake inhibitor (SSRI) antidepressants, the serotonin and noradrenaline reuptake inhibitor (SNRI) venlafaxine, and benzodiazepine anxiolytics. Of patients showing significant depressive symptoms (CES-D ≥ 23, n = 38), 31 (82%) were not using an antidepressant and of patients showing at least mild depressive symptoms (CES-D ≥ 16, n = 82), 71 (87%) were not using an antidepressant.
Physiological and cardiopulmonary characteristics
Patient physiological and exercise characteristics are summarized in Table II. The mean VO2 Peak was 17.1 (SD 4.7) ml/kg/min, which is approximately 65% of an age-matched norm and considered very poor for patients of this age. The mean BMI was found to be 28.0 (SD 4.7) kg/m2, which is associated with an increased risk of cardiovascular complications. Of all patients, 45.1% were classified as overweight (BMI between 25 and 30 kg/m2) and 27.3% were classified as obese (BMI ≥ 30 kg/m2). The mean WHtR was found to be 0.569 (SD 0.074), which is classified as overweight and characterizes the risk of cardiovascular complications to be significant.
| Table II. Patient physiological characteristics |
| | Mean (SD) |
| Anthropometric |
| BMI, kg/m2 | 28.0 (4.7) |
| Percentage body fat, % | 28.8 (9.1) |
| Waist-to-height ratio | 0.569 (0.074) |
| Resting physiology |
| Resting heart rate, bpm | 66.9 (12.7) |
| Resting systolic BP, mmHg | 131.8 (20.2) |
| Resting diastolic BP, mmHg | 72.9 (9.6) |
| Fitness parameters |
| VO2 peak, ml/kg/min | 17.1 (4.7) |
| Anaerobic threshold, ml/kg | 13.2 (3.0) |
| Maximum heart rate, bpm | 113.1 (22.3) |
| Maximum systolic BP, mmHg | 176.6 (28.2) |
| Maximum diastolic BP, mmHg | 79.1 (11.9) |
| BMI: body mass index; VO2 peak: peak oxygen consumption; BP: blood pressure. |
Predictors of depressive symptoms
When sociodemographic variables, cardiac factors, medical co-morbidities, concomitant medications and fitness parameters previously associated with increased depressive symptoms were entered into a backward linear regression, 6 variables remained in the model accounting for 14.7% of the variance (F = 8.713, p < .001). Table III contains coefficients obtained in the final regression model. This model suggests that a 1 ml/kg/min decrease in VO2 peak is associated with a 0.424-point increase in CES-D score, that a one year decrease in age is associated with a 0.17 point increase in CES-D score, and that a 1 mmHg decrease in maximum diastolic blood pressure is associated with a 0.105-point increase in CES-D score. Those with angina pectoris had a 5.8-point higher CES-D score on average than those without angina. Females had a 2.8-point higher CES-D score on average than males. Patients using an antidepressant had a 5.6-point higher CES-D score on average than those not using an antidepressant.
| Table III. Coefficients of linear regression model predicting CES-D scores* |
| Independent variables | Coefficient (B) | Standard error | p |
| VO2 peak | –0.424 | 0.128 | 0.001 |
| Age | –0.170 | 0.053 | 0.001 |
| Angina pectoris | 5.848 | 1.854 | 0.002 |
| Sex | 2.795 | 1.329 | 0.036 |
| Maximum diastolic BP | –0.105 | 0.047 | 0.026 |
| Antidepressant use | 5.607 | 2.172 | 0.010 |
| *R = 0.408, R-square = 0.166, adjusted R-square = 0.147, Standard error of the estimate = 8.302, p < 0.001. VO2 peak: peak oxygen consumption; BP: blood pressure; CES-D: Center for Epidemiological Studies Depression Scale |
DISCUSSION
A significant correlation between VO2 peak and CES-D scores was identified in a population of patients with CAD entering rehabilitation. This finding is consistent with the few available reports of correlations between reduced cardiopulmonary fitness and depressive symptoms in patients with CAD (4, 18). A correlation between VO2 peak and depressive symptoms has also been identified in women showing no clinical evidence of cardiovascular disease (16) suggesting that the relationship between depressive symptoms and fitness may be of broad clinical importance. The present study demonstrates the importance of the VO2 peak, as it emerged as the strongest predictor of CES-D scores, independent of age, sex, medical complications and concurrent medications. This finding may have implications for treatment. It is possible that exercise prescriptions aimed at improving cardiopulmonary fitness may also be beneficial for the improvement of depressive symptoms in persons with CAD. Longitudinal studies show that depressive symptoms tend to decrease over the course of cardiac rehabilitation (22), though it remains unclear which aspects of rehabilitation may be beneficial and whether this improvement is related to an increase in VO2 peak. It is also possible that depression and cardiovascular disease share aetiological underpinnings, such that, rather than treating sequelae, treatment impacting underlying causal factors will be necessary to improve both cardiac outcomes and depressive symptoms.
It has been speculated that psychological features of depression, such as reduced motivation, anhedonia and introversion, could result in an ongoing reduction in physical activity levels and contribute to reduced cardiopulmonary fitness in depressed subjects (16). Sustained inactivity could lead to loss or weakening of muscle tissue or affect neuronal pathways that regulate heart rate and cardiac output. As a limitation of this study, psychological variables, such as the roles of psychosocial wellbeing and social readjustment, were not formally assessed. Cardiac rehabilitation may help to improve VO2 peak by interacting with psychological features of depression, reducing motivational barriers and modifying behaviours not conducive to maintaining both cardiac health and positive affect.
In addition to psychological factors, physiological processes associated with depressive symptoms might interact with physical fitness. Recently, plasma levels of interleukin-6, a circulating pro-inflammatory cytokine, have been inversely correlated with the VO2 peak in asymptomatic men (23). Elevated levels of these cytokines, indicating activation of the inflammatory system, have also been observed in major depressive disorder where they could directly elicit depressive symptoms and thus contribute to reduced physical activity (24). As atherosclerosis and poor CAD prognoses have been associated with inflammatory activation (25), this hypothesis may be particularly relevant. In this report, increased depressive symptoms were found in patients with angina pectoris, which has been associated with increased plasma levels of pro-inflammatory cytokines (26). Elevated levels of pro-inflammatory cytokines have also been associated with feelings of exhaustion (27), which may be related to cardiopulmonary fitness and contribute to depressive symptoms. These potential mechanisms require further investigation.
Only 24% of subjects included in this study were female, in agreement with previous reports that female patients are less likely to enrol in cardiac rehabilitation following acute coronary events (28). This suggests the need to investigate possible gender-biases in patient attitudes towards rehabilitation or in physician referrals to rehabilitation. A significant association between female sex and increased CES-D scores was found, indicating that female sex independently predicted increased depressive symptoms. This is in accord with other reports that females are at increased risk of depressive symptoms among cardiac rehabilitation participants (17). This may be of particular importance, since female patients and those with higher depressive symptoms are less likely to complete cardiac rehabilitation. However, when female patients and those with significant depressive symptoms complete rehabilitation, they show significant improvements in lipid profile, BMI and exercise capacity (2). This suggests that developing methods to improve adherence to rehabilitation in women and depressed patients may be of substantial clinical benefit. Behaviour change theory has been suggested as a guiding principle in the design of novel individualized physical activity interventions for women, which could be integrated into cardiac rehabilitation. In one small trial, women who were randomized to a self-efficacy based intervention showed improvements in physical activity levels and cardiopulmonary fitness, whereas women randomized to a more traditionally structured exercise programme did not (29). Follow-up intervention has also been shown to protect against symptoms of anxiety and depression in women, while improving long-term risk-factor management (30). Cardiac rehabilitation may be an ideal setting to implement additional female gender-focused support.
Depressive symptoms could also be predicted by younger age. This is consistent with reports of increased psychological distress in younger patients with CAD (31). It is possible that depressive symptoms and CAD share common aetiopathological factors that mediate the association between depression and poorer CAD prognosis. Patients presenting at a younger age with complications of CAD therefore may be more severely affected by a process that also predisposes them to depressive symptoms. This hypothesis is supported by the observation of increased coronary risk factors in younger patients with CAD (31).
Reduced maximum diastolic blood pressure during fitness assessment was independently correlated with CES-D scores, although a correlation with resting diastolic blood pressure was not observed. In studies of older populations, low resting diastolic blood pressure has been associated with depressive symptoms (20), inciting considerable debate concerning possible consequences of aggressive antihypertensive treatment, which may include depressive symptoms and increased risks of cardiac and all-cause mortality (32). However, in this population, the correlation of lower maximum diastolic blood pressure with higher CES-D scores could not be accounted for by controlling for hypertension or the use of antihypertensive medications, and the use of antihypertensives was not associated with low blood pressure. This is consistent with another report associating low resting blood pressure with anxiety and depression independently of antihypertensive treatment (20). Reduced maximal blood pressure during stress testing may be related to depressive symptoms through dysregulation of central neuroendocrine systems. For example, chronic activation of the hypothalamic-pituitary-adrenocortical axis in depressed patients could result in abnormal glucocorticoid feedback regulation of this axis, or desensitization of pressor responses by down-regulation of adrenergic receptors. This could result in the blunted physiological responses to physical exertion observed in CAD patients with depressive symptoms, which is reminiscent of the blunted hypothalamic-pituitary-adrenocortical responses documented in patients with depression secondary to other medical illness (33). Lower maximum diastolic blood pressure was correlated with CES-D scores even when controlling for age and the use of antidepressant medications.
The rate of antidepressant use in this population was comparable to that reported in other studies of cardiac rehabilitation patients (34). Of patients showing significant depressive symptoms only 18% were using an antidepressant. This suggests that antidepressant medication may be underutilized in this population and that systematic screening for depression at entry into cardiac rehabilitation could help to fulfil an unmet need. It is possible that treatment of depressive symptoms may improve cardiac rehabilitation outcomes. Antidepressant use has been associated with reduced rates of mortality and recurrent MI in post-MI patients with depression (35). Similarly, depression has been associated with a reduced likelihood of completing rehabilitation (22) and treatment of depressive symptoms may interact with adherence to rehabilitation exercise prescriptions. Conversely, adverse risk profiles have been associated with the use of some antidepressants in certain patients with CAD (35), cautioning physicians to choose safe and effective treatments.
Limitations of this study include lack of a diagnostic interview for depression and a lack of recorded psychiatric history. In these patients, it is not known whether psychiatric symptoms preceded or followed acute coronary events. In future studies, it would be interesting to determine if the clinical characteristics of patients with a pre-morbid psychiatric condition differ from those who suffer depressive symptoms for the first time following an acute coronary event. A further limitation was the absence of an assessment of CAD severity. However, available indicators such as cardiac risk factors and medical/surgical histories were not correlated with CES-D scores and did not differ significantly between those who used antidepressants and those who did not, suggesting that differences in the severity of CAD do not account for the observed associations between depressive symptoms and cardiopulmonary fitness.
In summary, depressive symptoms are prevalent among patients with stable CAD entering cardiac rehabilitation and they are frequently untreated. Of the investigated parameters, the most significant independent predictor of depressive symptoms was the VO2 Peak during cardiopulmonary fitness assessment. Depressive symptoms were also predicted by younger age and female sex. The correlation between depressive symptoms and reduced physiological responses to exercise suggests the need to investigate possible psychological and physiological mechanisms mediating this correlation. An increased focus on psychological/psychiatric screening and intervention may improve the effectiveness of cardiac rehabilitation.
ACKNOWLEDGEMENTS
We acknowledge support from the Heart and Stroke Foundation and from the Toronto Rehabilitation Institute who receive funding under the Provincial Rehabilitation Research Program from the Ministry of Health and Long-Term Care in Ontario. The views expressed in this paper do not necessarily reflect those of the Ministry.
REFERENCES
1. Frasure-Smith N, Lesperance F, Juneau M, Talajic M, Bourassa MG. Gender, depression, and one-year prognosis after myocardial infarction. Psychosom Med 1999; 61: 26–37.
2. Caulin-Glaser T, Maciejewski PK, Snow R, LaLonde M, Mazure C. Depressive symptoms and sex affect completion rates and clinical outcomes in cardiac rehabilitation. Prev Cardiol 2007; 10: 15–21.
3. Penninx BW, Guralnik JM, Ferrucci L, Simonsick EM, Deeg DJ, Wallace RB. Depressive symptoms and physical decline in community-dwelling older persons. JAMA 1998; 279: 1720–1726.
4. Lavoie KL, Fleet RP, Lesperance F, Arsenault A, Laurin C, Frasure-Smith N, Bacon SL. Are exercise stress tests appropriate for assessing myocardial ischemia in patients with major depressive disorder? Am Heart J 2004; 148: 621–627.
5. Cankurtaran M, Halil M, Yavuz BB, Dagli N, Cankurtaran ES, Ariogul S. Depression and concomitant diseases in a Turkish geriatric outpatient setting. Arch Gerontol Geriatr 2005; 40: 307–315.
6. Almeida OP, Flicker L, Norman P, Hankey GJ, Vasikaran S, van Bockxmeer FM, Jamrozik K. Association of cardiovascular risk factors and disease with depression in later life. Am J Geriatr Psychiatry 2007; 15: 506–513
7. Norwood R, Balkissoon R. Current perspectives on management of co-morbid depression in COPD. COPD 2005; 2: 185–193.
8. Ruo B, Rumsfeld JS, Hlatky MA, Liu H, Browner WS, Whooley MA. Depressive symptoms and health-related quality of life: the Heart and Soul Study. JAMA 2003; 290: 215–221.
9. Ried LD, Tueth MJ, Taylor MD, Sauer BC, Lopez LM, Pepine CJ. Depressive symptoms in coronary artery disease patients after hypertension treatment. Ann Pharmacother 2006; 40: 597–604.
10. Morales K, Wittink M, Datto C, DiFilippo S, Cary M, TenHave T, Katz IR. Simvastatin causes changes in affective processes in elderly volunteers. J Am Geriatr Soc 2006; 54: 70–76.
11. Radloff L. The CES-D scale: A self-report depression scale for research in the general population. Appl Psychol Meas 1977; 1: 385–401.
12. Beekman AT, Deeg DJ, Van Limbeek J, Braam AW, De Vries MZ, Van Tilburg W. Criterion validity of the Center for Epidemiologic Studies Depression scale (CES-D): results from a community-based sample of older subjects in The Netherlands. Psychol Med 1997; 27: 231–235.
13. Roberts RE, Vernon SW. The Center for Epidemiologic Studies Depression Scale: its use in a community sample. Am J Psychiatry 1983; 140: 41–46.
14. Hamm LF, Kavanagh T. The Toronto Cardiac Rehabilitation and Secondary Prevention Program: 1968 into the new millennium. J Cardiopulm Rehabil 2000; 20: 16–22.
15. Milani RV, Lavie CJ, Mehra MR, Ventura HO. Understanding the basics of cardiopulmonary exercise testing. Mayo Clin Proc 2006; 81: 1603–1611.
16. Hollenberg M, Haight T, Tager IB. Depression decreases cardiorespiratory fitness in older women. J Clin Epidemiol 2003; 56: 1111–1117.
17. Josephson EA, Casey EC, Waechter D, Rosneck J, Hughes JW. Gender and depression symptoms in cardiac rehabilitation: women initially exhibit higher depression scores but experience more improvement. J Cardiopulm Rehabil 2006; 26: 160–163.
18. Marchionni N, Fattirolli F, Fumagalli S, Oldridge NB, Del Lungo F, Bonechi F, et al. Determinants of exercise tolerance after acute myocardial infarction in older persons. J Am Geriatr Soc 2000; 48: 146–153.
19. Weber-Hamann B, Werner M, Hentschel F, Bindeballe N, Lederbogen F, Deuschle M, Heuser I. Metabolic changes in elderly patients with major depression: evidence for increased accumulation of visceral fat at follow-up. Psychoneuroendocrinology 2006; 31: 347–354.
20. Hildrum B, Mykletun A, Stordal E, Bjelland I, Dahl AA, Holmen J. Association of low blood pressure with anxiety and depression: the Nord-Trondelag Health Study. J Epidemiol Community Health 2007; 61: 53–58.
21. Hofer S, Doering S, Rumpold G, Oldridge N, Benzer W. Determinants of health-related quality of life in patients with coronary artery disease. Eur J Cardiovasc Prev Rehabil 2006; 13: 398–406.
22. Turner SC, Bethell HJ, Evans JA, Goddard JR, Mullee MA. Patient characteristics and outcomes of cardiac rehabilitation. J Cardiopulm Rehabil 2002; 22: 253–260.
23. Kullo IJ, Khaleghi M, Hensrud D. Markers of inflammation are inversely associated with VO2 max in asymptomatic men. J Appl Physiol 2007;102: 1374–1379
24. Miller GE, Stetler CA, Carney RM, Freedland KE, Banks WA. Clinical depression and inflammatory risk markers for coronary heart disease. Am J Cardiol 2002; 90: 1279–1283.
25. Frasure-Smith N, Lesperance F, Irwin MR, Sauve C, Lesperance J, Theroux P. Depression, C-reactive protein and two-year major adverse cardiac events in men after acute coronary syndromes. Biol Psychiatry 2007; 62: 302–308
26. Hashmi S, Zeng QT. Role of interleukin-17 and interleukin-17-induced cytokines interleukin-6 and interleukin-8 in unstable coronary artery disease. Coron Artery Dis 2006; 17: 699–706.
27. Appels A, Bar FW, Bar J, Bruggeman C, de Baets M. Inflammation, depressive symptomatology, and coronary artery disease. Psychosom Med 2000; 62: 601–605.
28. Grace SL, Abbey SE, Shnek ZM, Irvine J, Franche RL, Stewart DE. Cardiac rehabilitation II: referral and participation. Gen Hosp Psychiatry 2002; 24: 127–134.
29. Dallow CB, Anderson J. Using self-efficacy and a transtheoretical model to develop a physical activity intervention for obese women. Am J Health Promot 2003; 17: 373–381.
30. Mittag O, China C, Hoberg E, Juers E, Kolenda KD, Richardt G, et al. Outcomes of cardiac rehabilitation with versus without a follow-up intervention rendered by telephone (Luebeck follow-up trial): overall and gender-specific effects. Int J Rehabil Res 2006; 29: 295–302.
31. Lavie CJ, Milani RV. Adverse psychological and coronary risk profiles in young patients with coronary artery disease and benefits of formal cardiac rehabilitation. Arch Intern Med 2006; 166: 1878–1883.
32. Messerli FH, Mancia G, Conti CR, Hewkin AC, Kupfer S, Champion A, et al. Dogma disputed: can aggressively lowering blood pressure in hypertensive patients with coronary artery disease be dangerous? Ann Intern Med 2006; 144: 884–893.
33. Giese-Davis J, Wilhelm FH, Conrad A, Abercrombie HC, Sephton S, Yutsis M, et al. Depression and stress reactivity in metastatic breast cancer. Psychosom Med 2006; 68: 675–683.
34. Grace SL, Abbey SE, Pinto R, Shnek ZM, Irvine J, Stewart DE. Longitudinal course of depressive symptomatology after a cardiac event: effects of gender and cardiac rehabilitation. Psychosom Med 2005; 67: 52–58.
35. Roose SP, Miyazaki M. Pharmacologic treatment of depression in patients with heart disease. Psychosom Med 2005; 67 Suppl 1: S54–S57.