OBJECTIVE: To investigate the effects of total-contact fitted carbon-composite knee-ankle-foot orthoses (KAFOs) on energy cost of walking in patients with former polio who normally wear a conventional leather/metal KAFO or plastic/metal KAFO.
DESIGN: A prospective uncontrolled study with a multiple baseline and follow-up design. Follow-up measurements continued until 26 weeks after intervention.
PARTICIPANTS: Twenty adults with polio residuals (mean age 55 years).
INTERVENTION: Each participant received a new carbon-composite KAFO, fitted according to a total-contact principle, which resulted in a rigid, lightweight and well-fitting KAFO.
OUTCOME MEASURES: Energy cost of walking, walking speed, biomechanics of gait, physical functioning and patient satisfaction.
RESULTS: The energy cost decreased significantly, by 8%, compared with the original KAFO. Furthermore, the increment in energy cost during walking with the carbon-composite KAFO was reduced by 18% towards normative values. An improvement in knee flexion, forward excursion of the centre of pressure, peak ankle moment, and timing of peak ankle power were significantly associated with the decrease in energy cost. Walking speed and physical functioning remained unchanged.
CONCLUSION: In patients with former polio, carbon-composite KAFOs are superior to conventional leather/metal and plastic/metal KAFOs with respect to improving walking efficiency and gait, and are therefore important in reducing overuse and maintaining functional abilities in polio survivors.
Key words: biomechanics of gait, carbon-composite KAFO, orthosis, post-poliomyelitis syndrome, rehabilitation, walking efficiency.
J Rehabil Med 2007; 39: 651–657
Correspondence address: Merel-Anne Brehm, PhD, Department of Rehabilitation Medicine, VU University Medical Center, De Boelelaan 1117, INL-1081 HV Amsterdam, The Netherlands. E-mail: m.brehm@vumc.nl
Submitted December 4, 2006; accepted May 24, 2007.
Introduction
Persons with a history of poliomyelitis often exhibit gait abnormalities due to residual lower extremity pareses and joint deformities. Gait abnormalities in patients with former polio have been shown to result in an increased energy cost (EC) of walking, being positively associated with lower extremity muscle weakness (1, 2). Many polio survivors experience increased muscle weakness later on in life, together with increased fatigue, pain and reduced endurance. These new neuromuscular symptoms are referred to as the ‘post-poliomyelitis syndrome’ (PPS) (3). PPS often leads to a decline in functional abilities, with a decrease in walking ability as the most prominent problem (4–6). Moreover, persons with PPS are predisposed to premature fatigue in carrying out physical activities of daily life, due to overuse of reduced muscle capacity (7–10), in combination with an increased EC of walking (2). Both the gait abnormalities and the decline in physical performance can be expected to increase over time, due to PPS, ageing, and progressive joint deformities (11).
Lower extremity orthoses are often prescribed for former polio patients to reduce gait abnormalities, to enable standing and walking, and to maintain or improve physical performance. A population-based study reported the need for lower extremity orthoses in approximately 16% of polio survivors (9). A much higher percentage, 48%, has been reported among patients visiting a post-polio clinic (5). A substantial number of the patients wore knee-ankle-foot orthoses (KAFOs), often since their recovery from acute polio in childhood. In general, these KAFOs are rather heavy, and the construction methods for fabricating them have not been changed for decades. Yet, it has been recognized that innovations are needed (12), and that orthoses should be made of the lightest possible, yet durable, materials to make walking energy-efficient (12). This would suggest the use of materials other than leather and metal.
Orthoses based on plastic, such as polypropylene, usually in combination with metal parts, are frequently manufactured to reduce weight. A disadvantage of such devices, however, is the limited rigidity of plastic materials. Therefore, carbon seems to be more promising, since it is both light and rigid. Carbon can be used to reinforce KAFOs made of resin-hardened acrylics or thermoplastics. Such carbon KAFOs have been reported to be more corrective, and to be appreciated more by their users (13, 14). Carbon-composite, consisting of woven carbon pre-impregnated with resin, may be even more promising, as it can be used to make full carbon KAFOs that are lightweight, rigid and strong, and enable total-contact fitting. The effectiveness of such carbon-composite KAFOs has not yet been objectively quantified with regard to the energy efficiency and biomechanics of gait, which are especially relevant in view of the late functional decline due to PPS.
This prospective study investigated the effects of total- contact fitted carbon-composite KAFOs on the EC of walking in patients with former polio who normally wear a conventional leather/metal (LM) KAFO or plastic/metal (PM) KAFO. The study also investigated whether changes in the biomechanics of gait were associated with a change in EC of walking.
Methods
Study population
Adults with a history of poliomyelitis participated in the study. They were recruited from the outpatient clinics of rehabilitation departments in (university) hospitals and rehabilitation centres, from the Dutch Neuromuscular Diseases Association, and from the manufacturers of orthoses. The inclusion criteria were: (i) ability to walk for at least 4 minutes at a comfortable speed; (ii) age between 18 and 70 years; and (iii) using a conventional locked knee-joint KAFO made of LM or PM, with no technical deficits, for at least 2 years.
Intervention
Each participant received a new custom-made carbon-composite KAFO with a locked knee-joint, fitted according to a total-contact principle (15). These design properties resulted in a very rigid, lightweight and well-fitting KAFO, thus enabling the improvement in orthotic control options in comparison with the existing KAFO. Achievable improvements included 3 control options that applied to the whole group: (i) postural correction, (ii) reduction in deformity during weight-bearing, and (iii) application of a dorsiflexion stop in the ankle-joint to improve the ankle rocker. A fourth control option, (iv) ischial weight-bearing, applied to half of the group (Table I). Furthermore, reduction in KAFO weight was an improvement option that also applied to half of the group, and in general to those who wore a LM KAFO. Noppe Orthopedie BV (Noordwijkerhout, The Netherlands) manufactured the new KAFOs. Prior to the manufacture, the configuration of the KAFO was carefully determined on the basis of the results of physical examination and clinical movement analysis, for which the multi-media Sybar® system (16) was used.
| Table I. Biomechanical gait parameters |
| New KAFO control options | Improvement objectives on biomechanics of gait |
| Improved postural correction (total-contact fitting) | Decrease in mean sagittal knee angle in midstance |
| | Decrease in mean frontal knee angle in midstance |
| Reduction in deformity during weight-bearing (stiffness) | Minimization of change in sagittal knee angle in loading response |
| Minimization of change in frontal knee angle in loading response |
| Minimization of mean sagittal knee moment in midstance |
| Minimization of mean frontal knee moment in midstance |
| Minimization of change in hip joint centre vertical motion and trochantor major vertical motion during stance |
| Improved ankle rocker function by applying a dorsiflexion stop at the ankle hinge (biomechanical action) | Increase in forward centre of pressure excursion in midstance |
| Minimization of change in dorsiflexion angle in terminal stance |
| Increase in peak sagittal ankle moment in terminal stance |
| Optimisation of timing peak ankle moment in terminal stance |
| Increase in peak ankle power in terminal stance |
| Optimisation of timing peak sagittal ankle power in terminal stance |
| Improved ischial weight-bearing (biomechanical action) | Minimization of mean sagittal hip moment in stance |
| | Minimization of mean lateral trunk sway in stance |
| Stance (0–100%): loading response (0–30%); midstance (30–70%); terminal stance (70–90%) (based on gait cycle phases according to Perry (24)). |
Study design and procedures
In order to evaluate differences between the new and the old KAFO, the following assessments were made: (i) EC of walking, (ii) biomechanics of gait, (iii) physical functioning, and (iv) patient satisfaction. To determine any learning effects with regard to these assessments, the study design contained 3 baseline measurements with the old KAFO, each separated by 2 weeks (B1, B2, B3), and 3 follow-up measurements with the new KAFO that were performed at 4, 12, and 26 weeks after the intervention (I1, I2, I3). The follow-up period started when the patients were wearing their new KAFO permanent, without any complaints. Biomechanics of gait was assessed at B1 and I3. Furthermore, technical deficits were recorded as they occurred, and once more they were listed with a questionnaire one year after delivery.
Measurements
EC of walking. A lightweight, portable gas-analysis system (VmaxST, Sensormedics, Bilthoven, The Netherlands), which has been shown to be accurate and reproducible in former polio patients (2, 17), was used to determine EC of walking. The system consists of a face mask, a triple volume transducer, a gas-sample line, and a battery-operated unit (650 g) that is worn on the shoulders. All the measurements and calibration procedures were conducted in accordance with the manufacturer’s instructions, including specific instructions not to eat or drink anything during a 1.5-h period prior to testing (17). Each measurement included a resting test and a walking test. The resting test consisted of sitting quietly for 10 min, and was followed by a 5-min walking test on a marked indoor oval track (50 m). During the walking test the subjects were asked to walk at their own, self-preferred, comfortable speed, using their customary walking aids. Throughout both tests breath-by-breath oxygen uptake (VO2) and carbon dioxide production (VCO2) values were registered. The distance covered during the last 2 min of the walking test was also registered in order to calculate the walking speed.
Biomechanics of gait. For the biomechanical analysis of gait, the subject, wearing the KAFO and shoes and using customary walking aids, walked at a comfortable speed along a 10 m walkway. A three-dimensional-movement analysis system (OPTOTRAK, Northern Digital, Inc., Waterloo, Canada) was used to measure the trajectories of active markers placed on the body (100 Hz). An 8-segment model of the human body was applied: left and right foot, lower legs, upper legs, pelvis and trunk. Rigid clusters of 3 markers were tightly attached to each segment. Simultaneously, the ground reaction force (GRF) was recorded at 1000 Hz from a force plate (AMTI, Inc., Watertown, USA) that was sunken into the floor. A total of 3 walking trials with the KAFO-leg stepping on the force plate were captured for each subject. Previous to these walking trials, the marker clusters were calibrated by probing 28 anatomical landmarks during a static trial, in order to define the co-ordinate systems for the joints involved (18, 19).
Physical functioning and patient satisfaction. Physical functioning (PF) was assessed with the Short Form Health Survey-36 (SF36), a multi-dimensional generic measure of health status, which contains 36 items organized into 8 multi-item scales (20). Only the PF scale of the SF36 was used for analysis. PF is summarized from 10 items, each assessing limitations in physical activity due to health problems.
An individualized satisfaction evaluation was made to quantify patient-specific improvements on various aspects of KAFO-use. At baseline, the patients were asked to name 5 self-chosen improvement items, and to rate these items for satisfaction with regard to the old KAFO on a 10-point Likert scale (1 = extremely unsatisfied, 10 = extremely satisfied). At follow-up, the patients were asked to rate the items again with regard to the new KAFO.
Data analysis
EC of walking. For the resting and walking test, respiratory exchange ratios (RERs) were calculated for each breath as the ratio of VCO2 to VO2. Breath-by-breath RER and VO2 values in min 5–7 for resting and min 3–5 for walking were then used to calculate the average steady-state energy consumption (ECS) values. Here, the equation of Garby & Astrup was used ((4.960*RER+16.040)*VO2) (21). A standard mass normalization procedure was applied, and the following parameters were calculated: resting ECS, gross ECS, and net ECS (net = gross – resting), expressed in J/kg/min. Finally, the primary outcomes, i.e. gross and net EC of walking, were calculated, expressed in J/kg/m. Gross and net EC were also expressed as increments above norm values, which were drawn from an able-bodied reference group (2). Walking speed was calculated as the mean speed during the last 3-5 min of the walking test.
Biomechanics of gait. For the synchronization and reduction of the biomechanical gait data, a MATLAB-based software programme was used (BodyMech, Matlab®, The Mathworks (www.bodymech.nl)). First, three-dimensional (3D) joint angles were calculated from the raw marker data. The joint co-ordinate systems were anatomically calibrated according to the CAMARC convention. (19) After synchronization, the GRF signals were used to calculate the centre of pressure (COP) of the force under the foot. 3D net moments around the hip, knee and ankle were calculated as the cross-product of the GRF and the distance from the joint centres to that vector (22, 23). Finally, for each trial, this analysis resulted in individual data on trunk movements, joint angles and net moments of hip, knee and ankle of one gait cycle, i.e. the gait cycle during which GRF signals were present. Subsequently, for each trial the data were time-normalized to the stance phase of that gait cycle (presented from 0% to 100%) and those data were then averaged for the 3 trials per subject. These individual data sets were used to compare the biomechanical output during gait with the old and the new KAFO with respect to 15 gait parameters (based on the gait phases according to Perry (24)) that were considered to be relevant (Table I, column 2).
Statistical analyses
From pilot data and a previous study (2), the EC of walking for patients with polio with a conventional KAFO was 25–35% higher than for healthy controls. As a clinically significant change a reduction of 25% in the extra energy expenditure was chosen, which means a reduction of 7.5% in total energy expenditure. It was calculated that a minimum sample size of 20 participants was required to detect such a change with a power of 80% and p < 0.05.
To study the effect of the intervention, a repeated measurement analysis, i.e. Generalized Estimated Equations (GEE), was applied (25). GEE is a linear regression analysis that takes into account the dependency of the observations within one patient, and that can handle unequal time-intervals. It allows all longitudinal data to be used, not only the data of complete cases. GEE was used to test for changes in gross EC, net EC, walking speed, and PF. Furthermore, GEE analysis was used to investigate whether changes in gross and net EC were associated with changes in biomechanical gait parameters and KAFO weight. GEE analysis was performed in STATA (version 7). Paired t-tests were used to assess the differences between the old and the new KAFO for the biomechanical gait parameters and patient satisfaction. Paired t-tests were performed in SPSS for Windows (version 11.5), with significance level of p < 0.05.
Results
Between October 2002 and October 2004, 23 patients with former polio were included in the study (14 men, 9 women); 14 of them were diagnosed with PPS according to the Halstead criteria (3). During the course of the study, one person was lost to follow-up, and 2 withdrew from the study because they found the burden of participation too high. A total of 20 people completed the study (13 men, 7 women, 13 PPS, 7 non-PPS). Their mean age was 55 years (SD 9.2), their mean body-mass was 72 kg (SD 11.8), and their mean body-mass index was 25.9 (SD 4.1), 25.8 for men and 26 for woman. The mean weight of the old KAFO and the new KAFO was 2.1 kg (SD 0.8) and 1.4 kg (SD 0.3), respectively. Table II presents the clinical condition of this group. The muscle strength sum (MSS) presented is a lower extremity strength sum score (based on manual muscle -testing of the left and right hip flexors, hip extensors, hip abductors, hip adductors, knee flexors, knee extensors, dorsal flexors and plantar flexors) that was calculated according to the method described by Nollet et al. (6).
| Table II. Clinical condition of subjects on inclusion |
| | Paretic body parts | Lower extremity strength (MSS*) | Walking devices | Material, weight (kg) old KAFO | Weight (kg) new carbon KAFO |
| 1 | L | 16.0 | KAFO L | LM, 1.8 | 1.3 |
| 2 | L, R | 8.0 | KAFO R + cane | LM, 4.1 | 1.7 |
| 3 | L, R | 5.0 | KAFO R + cane | LM, 3.1 | 1.4 |
| 4 | L, R | 13.0 | KAFO R + cane | LM, 1.8 | 1.6 |
| 5 | R | 14.0 | KAFO R | PM, 2.3 | 1.5 |
| 6 | L, R, T | 5.0 | KAFO R + 2 canes | LM, 2.9 | 2.1 |
| 7 | R | 18.0 | KAFO R | PM, 1.1 | 1.2 |
| 8 | R | 8.5 | KAFO R | LM, 2.3 | 1.4 |
| 9 | L | 17.5 | KAFO L | LM, 2.6 | 2.0 |
| 10 | L, R | 8.5 | KAFO L + cane | LM, 1.7 | 1.2 |
| 11 | L, R | 5.5 | KAFO R | LM, 2.2 | 1.3 |
| 12 | L, R, T | 6.0 | KAFO R + 2 canes | LM, 2.2 | 1.3 |
| 13 | L | 16.5 | KAFO L | LM, 2.6 | 1.9 |
| 14 | L, R | 23.0 | KAFO R | LM, 2.2 | 1.2 |
| 15 | L, R | 10.0 | KAFO R | LM, 2.4 | 1.5 |
| 16 | L, R | 15.0 | KAFO L + cane | PM, 1.4 | 1.3 |
| 17 | L | 11.0 | KAFO L + cane | PM, 1.6 | 1.3 |
| 18 | R | 16.0 | KAFO R + cane | LM, 1.2 | 1.2 |
| 19 | R | 17.0 | KAFO R + cane | LM, 1.5 | 0.9 |
| 20 | R | 11.5 | KAFO R | PM, 1.0 | 1.1 |
| L: left leg; R: right leg; T: trunk; MSS: muscle strength sum; KAFO: knee-ankle-foot orthosis; LM: leather/metal; PM: plastic/metal. *MSS range: (0–32), calculation described in detail elsewhere (6). |
Technical deficits
In general, the new KAFO was very well appreciated. The main issue that was reported by 7 patients included a technical deficit regarding the hinge at the ankle or knee (i.e. a crack of the clinch-nail). However, this deficit could easily be repaired. One non-technical issue that 7 patients reported included wear to the cloth upholstery inside the KAFO. One patient needed a replacement of the orthosis, due to a break of the KAFO.
Outcomes
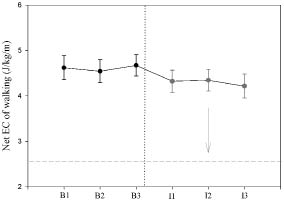
EC of walking. For all parameters data of complete cases were used, except for net EC of walking that was missing for 2 cases at B1. No significant learning effects were found for the EC parameters (Fig. 1) or for walking speed (Table III). The gross and net EC of walking were significantly lower for the new KAFO, than for the old KAFO: 7% (p < 0.001) and 8% (p < 0.001), respectively. The increments in gross and net EC above norm values with the old KAFO were both reduced by 18% during walking with the new KAFO. Walking speed itself remained unchanged (Table III) .
Fig. 1. Averaged data and standard errors over all subjects for net energy cost (EC) of walking on 6 different study visits. The visit numbers represent: B1 (week –6); B2 (week –4); B3 (week –2); I1 (week +4); I2 (week +12); I3 (week +26). Dotted line represents the moment of carbon composite KAFO intervention in the group (n = 20). The dashed line represents the normative net EC value (2.4 J/kg/m) of healthy subjects. The arrow represents the 18% reduction in net EC of walking towards normative.
| Table III. Results for energy cost (EC) of walking and walking speed; the means (SD) are given for the old and the new knee-ankle-foot ortgosis (KAFO) |
| | Old KAFO | New KAFO | Mean changea | 95% CIb |
| Speed (m/min) | 51.0 (12.4) | 52.8 (12.4) | 1.8 (3 %) | (–4.35, 0.57) |
| Gross EC J/kg/m) | 6.11 (1.4) | 5.70 (1.3)* | –0.42 (–7%) | (–0.63, –0.21) |
| Net EC (J/kg/m) | 4.66 (1.0) | 4.30 (1.0)* | –0.36 (–8%) | (–0.54, –0.18) |
| Gross EC, above normc | 2.69 (1.4) | 2.42 (1.3)* | –0.47 (–18%) | (–0.27, –0.67) |
| Net EC, above normc | 2.24 (1.0) | 1.87 (1.0)* | –0.37 (–18%) | (–0.19, –0.55) |
| aMean differences are presented in absolute units and as a percentage (%); % are calculated as ([new-old]/[new +old]/2)*100%. b95% confidence interval of the difference. cNorm values used: 3.2 J/kg/m for gross EC and 2.4 J/kg/m for net EC (see reference 2). *Significantly different (p < 0.000) from the old KAFO. SD: standard deviation. |
Biomechanics of gait. The results of the paired t-tests showed significant improvements in change of knee angle during the loading response (p = 0.003), forward excursion of the COP during midstance (p = 0.020), change in dorsiflexion angle during terminal stance (p = 0.003), and peak ankle moment during terminal stance (p = 0.003) (Table IV).
| Table IV. Results for gait parameters; the means (SD) are given for the old and the new knee-ankle-foot orthosis (KAFO) |
| Biomechanical gait parameters (part of stance phase in %) | Old KAFO | New KAFO | p-value | Association with decrease in net EC Yes/No (p-value) |
| Improved postural correction |
| Sagittal knee angle (40–60%) | –10.3º (8.1) | –8.0º (4.8) | 0.187 | Yes (0.040) |
| Frontal knee angle (40–60%) | 8.2º (9.4) | 11.4º (9.0) | 0.279 | No (0.678) |
| Reduction in deformity |
| Min/max sagittal knee angle (0–30%) | –8.4º (4.4) | –6.2º (3.7) | 0.003* | No (0.491) |
| Min/max frontal knee angle (0–30%) | –5.1º (2.8) | –5.6º (3.0) | 0.369 | No (0.931) |
| Sagittal knee moment (40–60%) | 0.21 Nm/kg (0.17) | 0.16 Nm/kg (0.12) | 0.128 | No (0.572) |
| Frontal knee moment (40–60%) | 0.18 Nm/kg (0.08) | 0.19 Nm/kg (0.15) | 0.731 | No (0.429) |
| Difference HJC and TM vertical motion (0–100%) | 33 mm (9) | 31 mm (13) | 0.457 | No (0.169) |
| Improved ankle rocker function |
| Forward COP excursion (30–70%) | 73 mm (39) | 86 mm (27) | 0.020* | Yes (0.037) |
| Min/max sagittal ankle angle (70–90%) | –6.5º (2.6) | –4.6º (2.4) | 0.003* | No (0.546) |
| Sagittal ankle moment (70–90%) | 0.59 Nm/kg (0.34) | 0.76 Nm/kg (0.24) | 0.003* | Yes (0.011) |
| Timing sagittal ankle moment (70–90%) | 84.2% (3.2) | 82.7% (3.8) | 0.145 | Yes (0.047) |
| Sagittal ankle power (70–90%) | 21.0 (14.9) | 15.7 (9.2) | 0.129 | No (0.181) |
| Timing sagittal ankle power (70–90%) | 83.5% (6.0) | 81.4% (6.2) | 0.291 | Yes (0.003) |
| Improved ischial weight-bearing |
| Sagittal hip moment (0–100%) | 0.20 Nm/kg (0.13) | 0.18 Nm/kg (0.16) | 0.580 | No (0.068) |
| Lateral trunk sway (0–100%) | 84 mm (41) | 83 mm (36) | 0.791 | No (0.703) |
| *Significantly different from the old KAFO. Percentages between brackets refer to the time normalized part of the stance phase. Stance = 0–100%; loading response = 0–30%; midstance = 30–70%, mid midstance = 40–60%; terminal stance = 70–90%. HJC: hip joint centre; TM: trochantor major; COP: centre of pressure; SD: standard deviation; EC:energy cost. |
GEE analysis showed that a decrease in the knee flexion angle during midstance was found to be significantly associated with the decrease in EC (p = 0.030 and p = 0.040 for gross and net EC, respectively). Furthermore, an increase in forward excursion of the COP (p = 0.001 and p = 0.020 for gross and net EC, respectively), peak ankle moment during terminal stance (p = 0.012 and p = 0.011 for gross and net EC, respectively), and timing of peak ankle power during terminal stance (p < 0.001 and p < 0.001 for gross and net EC, respectively) were significantly associated with the decrease in EC (Fig. 2, Table IV). The reduction in KAFO weight was not significantly associated with the decrease in EC.
Physical functioning and patient satisfaction. The results of the SF36 showed no significant difference in PF between the old and the new KAFO. With regard to patient satisfaction, a total of 26 items was mentioned. On average, mean subject satisfaction scores were 48% higher for the new KAFO, compared with the old KAFO (p < 0.000). Items that were most frequently indicated for improvement were weight, fitting, and cosmesis of the KAFO, stability, and walking performance.
Discussion
This study evaluated the effect of carbon-composite KAFOs on walking efficiency and gait in persons with polio residuals who wore either a conventional LM KAFO or PM KAFO. The major finding was that carbon-composite KAFOs reduced the physical effort of walking with clinical significance by decreasing the increment in net EC above norm values by 18%. In view of the lack of causal therapy for PPS (26), the optimization of KAFOs deserves serious consideration in order to reduce overuse and to improve or retain walking ability. A comparison of our EC results with the findings of previous studies is not possible, because this was the first study to focus on the EC of walking. So far, the effectiveness of carbon KAFOs in polio has been investigated only by means of structured questionnaires (13, 14).
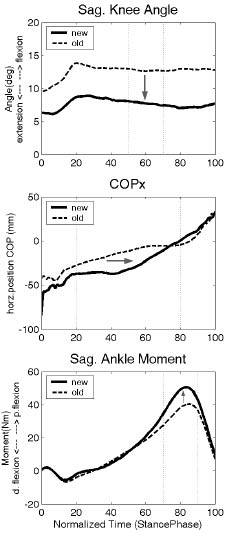
In order to investigate possible causes of improvement in EC of walking, 15 biomechanical gait parameters were analysed. The choice of these parameters was based on expected outcomes in relation to improved orthotic control options (Table I). First, the application of a dorsiflexion stop at the ankle-joint, in combination with a footplate that was stiffened until the metatarsophalangeal joints resulted in a significantly larger peak ankle moment (Fig. 2) and an improved timing of peak ankle power, compared with the conventional KAFO. Its mechanism is based on improving the forward COP excursion in the 2nd (i.e. ankle) rocker of stance phase, thereby enhancing the 3rd (i.e. foot) rocker. The result is a more effective push-off. Bennett et al. (27) described a similar line of reasoning with regard to the impact of ankle-foot orthoses (AFOs) on the forward COP excursion in children with cerebral palsy. They observed a trend of increased ankle moments during AFO walking, which was explained by a change in the COP motion, such that the relative phasing of potential and kinetic energies improved. The result was a more pendulum-like gait. Within an inverted pendulum model (28), this is expected to reduce the EC of walking. Although Bennett et al. (27) have not yet quantified such a relationship, the present study supports this assumption.
Apart from changes at the foot and ankle, significant changes also occurred at the knee. The change in knee flexion during the loading response decreased with the new KAFO, compared with the old KAFO. This was obtained by the combination of the total-contact fitting of the new KAFO and its stiffness, resulting in less postural deformity in response to weight-bearing. Yet, this change did not contribute to the improvement in walking efficiency. A possible explanation for this is that during loading response the function of the knee is more important in providing adequate shock absorption (24), than in controlling the EC of walking. Although the improvement in walking efficiency was not related to less postural deformity, adequate postural correction appeared to be of energetic benefit. As expected, the total-contact fitting and the rigidity of the construction enabled the subjects to remain in a more upright position, demonstrated by a decrease in the knee flexion angle during mid-stance (Fig. 2).
Fig. 2. Averaged data over all subjects for the gait parameters that are associated with the reduction in net EC (energy cost) of walking: (upper) Sagittal knee angle during mid-stance; (middle) Forward excursion of the centre of pressure; (lower) Sagittal ankle moment during terminal stance. Data are shown for the old KAFO (dashed lines) and new KAFO (solid lines). Arrows represent the direction of improvement.
KAFO: knee-ankle-foot orthoses; horz: horizontal; p: plantar; d:dorsal.
No significant changes were found at the hip and trunk. However, we did observe a trend of a reduced EC of walking in association with a reduced hip flexion moment (p = 0.068). The finding that this association did not reach significance might be due to the fact that the control option related to the aimed hip improvement, i.e. ischial weight-bearing, was applied in only 10 cases. The same holds for the reduction in KAFO weight, established in 9 cases, which was not a significant contributor to the reduction in EC.
The results described above clearly indicate that improved orthotic management can influence the gait pattern, which, in turn, improves walking efficiency. This relationship might support arguments proposed in a previous study, i.e. that EC of walking in individuals with PPS is strongly related to the extent of lower extremity muscle weakness, explaining 71% of the variance in EC (2). It was argued that orthotic and walking devices might have contributed to the unexplained variance, and that this will, in part, depend on the quality of the device, i.e. if the orthotic quality is insufficient, EC will be increased, and visa versa. The latter was supported by the present study; improved orthotic quality increased the efficiency of walking. However, this observation of improved walking efficiency was not reflected by a reduction in perceived limitations in PF, which remained unchanged. This is suggested to be due to the fact that the SF36-PF can only detect large changes in clinical status that might not be adequately responsive to intervention effects in polio and PPS (6, 29, 30). As pointed out by Horemans et al. (31), it might also indicate that patients with former polio do not necessarily adapt their daily-life behaviour to their physical capacities, because this is also determined by environmental and personal factors. Ambulatory monitoring of daily activities may be helpful in future outcome studies, to quantify changes in actual daily-life physical performance.
Satisfaction with the new KAFO increased impressively. All of the patients were satisfied with the new orthosis, and averaged satisfaction scores for patient-specific improvement items increased by almost 50%, compared with the old KAFO. Improvement items that were most frequently indicated were weight, fitting, and cosmesis of the KAFO, stability, and walking performance. Even though these items were individually chosen, similar items were mentioned and similar results were found in 2 previous studies (13, 14).
Some limitations of this study should be considered. First, it is difficult to decide to what extent the improvement in energy efficiency of walking is due to the application of carbon-composite. With conventional materials such as LM or PM, optimization of the orthosis might also have been possible to a certain extent. However, we believe that this can be achieved better with carbon-composite, because of its unique combination of properties: rigid, strong, lightweight and well mouldable. Another limitation is that for the 2 types of existing devices studied, weight and rigidity were related; that is, the PM KAFO was light and not rigid while the LM KAFO was rigid and heavy. Together with the limited number of subjects, we were not able to determine the precise contribution of the alteration of these factors to the improved energy efficiency of walking.
In conclusion, this study demonstrates that, for patients with former polio, carbon-composite KAFOs were superior to conventional LM and PM KAFOs with regard to walking efficiency, i.e. the increment in net EC above norm values was decreased by 18%. Improvements in the knee flexion angle, forward excursion of the COP, peak ankle moment, and timing of peak ankle power were significantly associated with the decrease in net EC. These improvements could be achieved due to the properties of carbon-composite.
The optimization of KAFOs to decrease the EC of walking, by using carbon-composite designs and improving their biomechanical action, results in an important option to reduce overuse and to maintain functional abilities in polio survivors, especially in those with PPS.
Acknowledgements
This study was supported by a grant from the Anna Fonds (02/07) and ZonMw (014-32-031), The Netherlands.
We are grateful to all the patients for their dedicated and enthusiastic participation in this study, and also to Tanneke Vogelaar (Department of Rehabilitation Medicine, VU University Medical Center, Amsterdam) for her assistance with data-collection.
References
1. Dean E, Ross J. Movement energetics of individuals with a history of poliomyelitis. Arch Phys Med Rehabil 1993; 74: 478–483.
2. Brehm MA, Nollet F, Harlaar J. The energy demands of walking in individuals with postpoliomyelitis syndrome: relationship with muscle strength and reproducibility. Arch Phys Med Rehabil 2006; 87: 136–140.
3. Halstead LS. Assessment and differential diagnosis for post-polio syndrome. Orthopedics 1991; 14: 1209–1217.
4. Nollet F, Beelen A, Sargeant AJ, de Visser M, Lankhorst GJ. Submaximal exercise capacity and maximal power output in polio patients. Arch Phys Med Rehabil 2001; 82: 1678–1685.
5. Waring WP, Maynard F, Grady W, Grady R, Boyles C. Influence of appropriate lower extremity orthotic management on ambulation, pain, and fatigue in a postpolio population. Arch Phys Med Rehabil 1989; 79: 915–919.
6. Nollet F, Beelen A, Prins MH, de Visser M, Sargeant AJ, Lankhorst GJ, de Jong BA. Disability and functional assessment in former polio patients with and without postpolio syndrome. Arch Phys Med Rehabil 1999; 80: 136–143.
7. Perry J, Barnes G, Gronley JK. The postpolio syndrome. An overuse phenomenon. Clin Orthop Relat Res 1988; 233: 145–162.
8. Perry J, Fontaine JD, Mulroy S. Findings in post-poliomyelitis syndrome. Weakness of muscles of the calf as a source of late pain and fatigue of muscles of the thigh after poliomyelitis. J Bone Joint Surg Am 1995; 77: 1148–1153.
9. Ivanyi B, Nollet F, Redekop WK, de Haan R, Wohlgemuht M, van Wijngaarden JK, de Visser M. Late onset polio sequelae: disabilities and handicaps in a population-based cohort of the 1956 poliomyelitis outbreak in the Netherlands. Arch Phys Med Rehabil 1999; 80: 687–690.
10. Thorén-Jönsson AL, Grimby G. Ability and perceived difficulty in daily activities in people with poliomyelitis sequelae. J Rehabil Med 2001; 33: 4–11.
11. Nollet F, Beelen A, Twisk JW, Lankhorst GJ, De Visser M. Perceived health and physical functioning in postpoliomyelitis syndrome: a 6-year prospective follow-up study. Arch Phys Med Rehabil 2003; 84: 1048–1056.
12. Huber SR. Therapeutic application of orthotics. In: Umphred DA, editor. Neurological rehabilitation. St. Louis: Mosby; 1995, p. 893–910.
13. Steinfeldt F, Selfert W, Günther KP. Moderne Karbonfaser-Orthetik fr Polio-Patienten Kritische Auswertung nach funktionellen Aspekten Z Orthop Ihre Grenzgeb 2003; 141: 357–361.
14. Heim M, Yaacobi E, Azaria M. A pilot study to determine the efficiency of lightweight carbon fibre orthoses in the management of patients suffering from post-poliomyelitis syndrome. Clin Rehabil 1997; 11: 302–305.
15. Chambers RB, Elftman N. Orthotic management of the neuropathic and dysvascular patient. In: Goldberg B, Hsu JD, editors. Atlas of orthoses and assistive devices. St. Louis: Mosby; 1997, p. 427–453.
16. Harlaar J, Redmeijer RA, Tump P, Peters R, Hautus E. The SYBAR system: integrated recording and display of video, EMG, and force plate data. Behav Res Methods Instrum Comput 2000; 32: 11–16.
17. Brehm MA, Harlaar J, Groepenhoff H. The validation of the portable VmaxST system for oxygen-uptake measurement. Gait Posture 2004; 20: 67–73.
18. Wu G, Siegler S, Allard P, Kirtley C, Leardini A, Rosenbaum D, et al. Standardization and Terminology Committee of the International Society of Biomechanics. ISB recommendation on definitions of joint coordinate system of various joints for the reporting of human joint motion-part I: ankle, hip, and spine. International Society of Biomechanics. J Biomech 2002; 35: 543–548.
19. Cappozzo A, Catani F, Della Croce U, Leardini A. Position and orientation in space of bones during movement: anatomical frame definition and determination. Clin Biomech 1995; 10: 171–178.
20. McHorney CA, Ware JE, Raczek AE. The Moss 36-item Short Form Health Survey (SF-36): II. Psychometric and clinical tests of validity in measuring physical and mental health constructs. Med Care 1993; 31: 247–263.
21. Garby L, Astrup A. The relationship between the respiratory quotient and the energy equivalent of oxygen during simultaneous glucose and lipid oxidation and lipogenesis. Acta Physiol Scand 1987; 129: 443–444.
22. Hof AL. An explicit expression for the moment in multibody systems. Technical note. J Biomech 1992; 25: 1209–1211.
23. Wells RP. The projection of the ground reaction force as a predictor of internal joint moments. Bull Prosthet Res 1981; 10–35: 15–19.
24. Perry J, editor. Gait analysis: normal and pathological function. Thorofare (NJ): SLACK Inc.; 1992: p. 100.
25. Zeger SL, Liang KY. Longitudinal data analysis for discrete and continuous outcomes. Biometrics 1986; 42: 121–130.
26. Farbu E, Gilhus NE, Barnes MP, Borg K, de Visser M, Driessen A, et al. EFNS guideline on diagnosis and management of post-polio syndrome. Report of an EFNS task force. Eur J Neurol 2006; 13: 795–801.
27. Bennet BC, Abel MF, Wolovich A, Fromklin F, Kerrigan C, Allaire PE. Abstract of 10th GCMAS meeting, Apr 6–9 2005, Portland (OR), USA.
28. Cavagna GA, Willems PA, Heglund NC. The role of gravity in human walking: pendular energy exchange, external work and optimal speed. J Physiol 2000; 528: 657–668.
29. Freeman AJ, Hobart JC, Langdon DW, Thompson AJ. Clinical appropriateness: a key factor in outcome measure selection: the 36 item short form health survey in multiple sclerosis. J Neurol Neurosurg Psychiatry 2000; 68: 150–156.
30. Nollet F, Horemans H, Beelen A, Trojan DA, Collet J-P, Shapiro S, et al. A multicenter, randomized, double-blinded trial of pyridostigmine in postpolio syndrome Neurology 2000; 55: 899–901.
31. Horemans HL, Bussmann JB, Beelen A, Stam HJ, Nollet F. Walking in postpoliomyelitis syndrome: the relationships between time-scored tests, walking in daily life and perceived mobility problems. J Rehabil Med 2005; 37: 142–146.