We report the case of a 31-year-old man who had mild traumatic brain injury as a result of an accident at the age of 24 years. Seven years after the trauma, at the age of 31 years, he had a lower verbal intelligence quotient than performance intelligence quotient by the Wechsler Adult Intelligence Scale – Revised, and frontal lobe dysfunction, for example, difficulty in maintaining or changing the set as revealed by the Wisconsin Card Sorting Test Keio Version. Conventional brain magnetic resonance imaging had not shown any abnormalities. Abnormal brain areas were detected on magnetic resonance diffusion tensor imaging. On tractography, some fibres from the corpus callosum towards the frontal cortex were noted to be lacking in the left hemisphere compared with the right. The tractography results may explain the patient’s lowered verbal intelligence quotient and focal left frontal lobe dysfunction. Diffusion tensor imaging is therefore helpful in detecting lesions in mild traumatic brain injury with diffuse axonal injury.
Key words: mild traumatic brain injury, diffuse axonal injury, neuropsychological dysfunction, magnetic resonance imaging, diffusion tensor imaging.
J Rehabil Med 2007; 39: 418–420
Correspondence address: Keiji Hashimoto, MD, PhD, Department of Rehabilitation Medicine, The Jikei University School of Medicine, 3-25-8 Nishishinbashi, Minato, 105-8461, Tokyo, Japan. E-mail: keiman@jikei.ac.jp
Submitted May 15, 2006; accepted December 28, 2006
INTRODUCTION
The diagnosis and management of mild traumatic brain injury (MTBI) continues to be the subject of debate, with varying opinions regarding the extent to which organically based impairments vs the impact of other stressors cause ongoing disability (1). Detecting areas of the brain with abnormalities that can explain symptoms and behaviour in patients with MTBI is important in order to confirm the diagnosis of MTBI. We report here a case in which magnetic resonance (MR) diffusion tensor imaging (DTI) could explain a patient’s lowered verbal intelligence quotient (VIQ) and focal left frontal lobe dysfunction.
CASE REPORT
The patient is a 31-year-old man who was involved in a motorcycle collision with a car when he was 24 years of age. He was taken to a hospital 30 minutes after the crash. His consciousness level was E4V3M6 (total 13) on the Glasgow Coma Scale. He recalls talking with one of his friends before arriving at the hospital. Computerized tomography scan findings in the acute phase were negative.
Three years after the accident, he was assigned the diagnosis of MTBI according to the operational criteria of the World Health Organization Collaborating Centre Task Force on MTBI for clinical identification (2).
Prior to the accident, the patient had been healthy and had no physical or cognitive dysfunction. Neither he nor his family noticed symptoms due to MTBI at the time of the accident. About a week after the accident, he went back to work. However, he could not continue working at the company that employed him because of neuropsychological dysfunction, such as fatigue, speech and memory problems, and higher executive dysfunction that had developed 3 years after the accident and was evident in his performance of activities of daily living.
Neuropsychological testing
Three years after the accident, when he was 27 years of age, his intelligent quotient by the Wechsler Adult Intelligence Scale – Revised was 95 in VIQ, 114 in performance intelligence quotient (PIQ) and 104 in full intelligence quotient (FIQ). By the time the patient reached the age of 31 years, these scores had improved to 102 in VIQ, 128 in PIQ and 114 in FIQ.
Also at the age of 27 years, his memory quotient by Wechsler Memory Scale – Revised was 76 for verbal memory, 114 for visual memory, 84 for general memory, 105 for attention/concentration and 84 for delayed recall. Then by 31 years of age, these scores improved to 119 for verbal memory, 104 for visual memory, 116 for general memory, 116 for attention/concentration and 109 for delayed recall. Also, the scores on the Wisconsin Card Sorting Test Keio Version in step I was 0 in categories achieved (CA), 8 in preservative errors of Nelson (PEN) and 8 in difficulty maintaining set (DMS). Scores in step II were 5 in PEN, 0 in CA and 0 in DMS. Scores at that time were 18 for animals and 21 for vegetables on the Word Fluency Test.
A greater than 15-point difference between VIQ and PIQ is considered to be significant according to the manual for the Japanese version of the Wechsler Adult Intelligence Scale – Revised. Therefore, we can say that his VIQ by the Wechsler Adult Intelligence Scale – Revisedwas significantly lower than the PIQ. That he had frontal lobe dysfunction was evident, as he displayed difficulty in maintaining or changing sets revealed by the Wisconsin Card Sorting Test Keio Version. We noted that the lowered visual memory score by Wechsler Memory Scale – Revised in the early stage had improved.
Imaging studies
At 27 years of age (3 years after the accident) a 1.5T magnetic resonance imaging (MRI) was performed to detect abnormalities that would cause neuropsychological dysfunction. However, none were revealed by this conventional MRI, including T2*, weighted images.
When the patient was 31 years of age (7 years after the accident) we used MR DTI to detect abnormal brain areas. DTI was performed by a 1.5T Signa MRI system (GE Medical Systems, Milwaukee, WI, USA). We used a spin echo-based echo planar sequence (TR/TE, 10 000/79 ms; slice thickness, 3 mm; field of view, 25 cm × 25 cm; number of acquisitions, 4; pixel matrix, 128 × 128) for diffusion tensor analysis. Diffusion gradients (b = 1000 s/mm2) were always applied on 2 axes simultaneously around the 180 pulses. Diffusion properties were measured along 6 non-collinear directions. Diffusion-weighted magnetic resonance images were transferred to a workstation supplied by the manufacturer (Advantage Workstation, GE Medical Systems); structural distortion induced by large diffusion gradients was corrected on the basis of T2-weighted echo–planar images (b = 0 s/mm2).
MR tractography was made with diffusion tensor visualization software (3). For tractography of the corpus callosum, seed volumes were located from the genus to the splenium through the body on reconstructed mid-sagittal images. For tractography of the fornix, seed volumes were located in the column of the fornix on reconstructed coronal images. The fractional anisotropy value for stop criteria was 0.18. Tractographic results were overlaid on T2-weighted images.
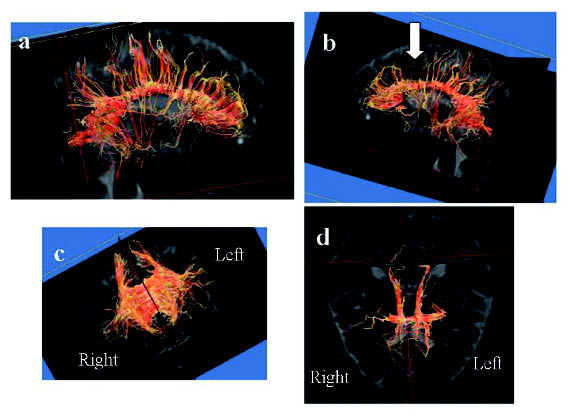
On tractography of the corpus callosum, we could see that some of the fibres from the corpus callosum towards the frontal cortex were lacking in the left hemisphere compared with the right (Fig. 1). Indeed, we could observe a defect of fibres around the cingulate gyrus and prefrontal area.
Fig. 1. Tractography from corpus callosum. (a) Lateral view on the right side. (b) Lateral view on the left side. (c) Anterior posterior view. (d) Top view. We can observe that some fibres from the corpus callosum towards the frontal cortex were lacking in the left hemisphere compared with the right side. The arrow in (b) indicates a defect in the fibres.
Discussion
Although this patient was diagnosed 3 years after his accident as having MTBI, he had rather severe sequelae, which necessitated resignation from his job. However, the presence of a lesion due to TBI could not be demonstrated by diagnostic imaging. Finally, after several years, we were able to detect abnormal areas of the brain using DTI. Neuropsychological dysfunction of brain-injured victims from TBI has hitherto been explained as a result of contusion of the frontal and temporal lobes. However, neuropsychological dysfunction similarly exists in patients with diffuse axonal injury in whom the absence of regional lesions is clearly shown on diagnostic imaging of the brain. The main cause of neuropsychological dysfunction after TBI is consequently considered to be axonal injuries (4). Wallesch et al. (5) performed neuropsychological examinations in the post-traumatic acute phase and reported cognitive dysfunction, particularly functional disorders in the frontal lobe, as sequelae.
Kamikubo et al. (6) reported a patient group whose VIQ level was significant lowered due to TBI sequelae and speculated that the principal factor in the cognitive dysfunction was axonal damage. Actually our case had neuropsychological dysfunction, such as lowered VIQ and executive dysfunction due to frontal lobe dysfunction.
Monchi et al. (7) reported detection by event-related functional MRI of increased activity in the mid-dorsolateral prefrontal cortex (area 9/46) and the mid-ventrolateral prefrontal cortex (area 47/12) while subjects underwent the Wisconsin Card Sorting Test Keio Version.
Nakayama et al. (8) reported a decline in fractional anisotropy around the genu, stem and splenium of the corpus callosum, which was shown by voxel-based analysis. Fractional anisotropy values of the genu (0.47), stem (0.48) and splenium of the corpus callosum (0.52), and the column of the fornix (0.51) were lower in patients with non-missile TBI than in healthy controls (0.58, 0.61, 0.62 and 0.61, respectively) according to the confirmatory study of the regions of interest. The white matter architecture in the corpus callosum and fornix of patients with non-missile TBI were seen to be coarser than in the controls by individual magnetic resonance tractography.
Abo et al. (9) reported that fractional anisotropy values were higher and apparent diffusion coefficient values were lower bilaterally in patients with chronic-stage DAI than in control subjects. Results shown by Abo et al. (9) conflict slightly with those of other reports, therefore, we do not consider that we can diagnose the severity of brain injury only by measuring fractional anisotropy or apparent diffusion coefficient values. In our study, we did not measure fractional anisotropy and addarent diffusion coefficient values, which have the advantage of providing numerical values. However, an advantage of tractography is that findings can be presented visually. Although it is not possible to find such an abnormality on fractional anisotrpy and apparent diffusion coefficient maps, tractography can reveal such abnormal lesions. Oedema sometimes affects tractography in the acute stage, but in this case, timing was so late that the effect of oedema could be excluded. In our patient, the lowered VIQ was probably a result of axonal damage in the left hemisphere. From results of tractography by DTI, we were able to explain that his lower VIQ and focal left frontal lobe dysfunction was due to damage of the prefrontal cortex. Thus, we conclude that DTI is helpful in persons with MTBI with DAI to detect lesions responsible for axonal damage, such as in our patient.
On the other hand, several limitations of this study should be mentioned. As described above, this patient’s test scores improved after repeated testing, which might be considered as evidence of improvement after possible brain damage. However, further studies of the patient are needed before findings of VIQ < PIQ can be convincingly interpreted as reflecting brain injury.
References
1. Ponsford J. Rehabilitation interventions after mild head injury. Curr Opin Neurol 2005; 18: 692–697.
2. Holn L, Cassidy JD, Carrol LJ, Borg J. Summary of the WHO collaborating centre for neurotrauma task force on mild traumatic brain injury. J Rehabil Med 2005; 37: 137–141.
3. Masutani Y, Uozumi K, Akahane M, Ohtomo K. MR diffusion tensor imaging: recent advance and new techniques for diffusion tensor visualization. Eur J Radiol 2003; 46: 53–66.
4. Meythaler JM, Peduzzi JD, Eleftheriou E, Novack TA. Current concepts: diffuse axonal injury-associated traumatic brain injury. Arch Phys Med Rehabil 2001; 82: 1461–1471.
5. Wallesch CW, Curio N, Galazky I, Jost S, Synowitz H. The neuropsychology of blunt head injury in the early postacute stage: effect of focal lesion and diffuse axonal injury. J Neurotrauma 2001; 18: 11–20.
6. Kamikubo T, Ohashi M, Hashimoto K, Okamoto T, Miyano S. Lowered VIQ level following traumatic brain injury. No To Shinkei 2004; 56: 952–956.
7. Monchi O, Petrides M, Petre V, Keith W, Dagher A. Wisconsin card sorting revisited: distinct neural circuits participating different stages of the task identified by event related functional magnetic resonance imaging. J Neorosci 2001; 21: 7733–7741.
8. Nakayama N, Okumura A, Shinoda J, Yasokawa Y, Miwa K, Yoshimura S, et al. Evidence for white matter disruption in traumatic brain injury without macroscopic lesions. J Neurol Neurosurg Psychiatry 2006; 77: 850–855.
9. Abo M, Hashimoto K, Okamoto T, Suzuki M, Kikuchi Y, Watanabe S, et al. Correlation between cognitive deficits and tensor magnetic resonance parameters in patients with chronic diffuse axonal injury. J Appl Res 2006; 6: 36–42.w