OBJECTIVE: To evaluate the effects of a cognitive behavioural training programme and a physical activity programme for patients with stress-related illnesses.
DESIGN: In a randomized controlled study, patients were allocated randomly to 1 of 3 groups, where group 1 participated in a cognitive behavioural training programme, group 2 participated in a physical activity programme, and group 3, the control group, was offered usual care for the course of the study.
SUBJECTS: A total of 75 patients participated in the study. They had been on sick leave for at least 50% of the time for between 1 month and 2 years due to stress-related illnesses.
METHODS: Measurements of autonomic activity, pressure-pain thresholds and subjective ratings of health and behaviour were made before and after a 10-week intervention period, and at 6 and 12 months after the intervention.
RESULTS: Minor differences in autonomic activity and pressure- pain thresholds were found between the groups immediately after the intervention. At the 6- and 12-month follow-up assessments, the differences were no longer present. Patients in the cognitive behavioural training group improved their ratings of general health compared with the physical activity group throughout the study.
CONCLUSION: The study showed little difference in the effect of cognitive behavioural training and physical activity, compared with usual care, for patients with stress-related illnesses.
Key words: autonomic activity, pressure-pain threshold, behaviour, burn-out, sick leave.
J Rehabil Med 2007; 39: 366–373
Correspondence address: Marina Heiden, Centre for Musculoskeletal Research, PO Box 7629, SE-907 12 Umeå, Sweden. E-mail: marina.heiden@hig.se
Submitted February 2, 2006; accepted November 8, 2006
INTRODUCTION
Stress-related illnesses are common, often leading to long-term sick leave (1, 2). Patients with stress-related illnesses generally complain of fatigue, sleep disturbance, concentration difficulty, musculoskeletal pain and depression (2–4). In the present study, stress-related illnesses are defined as illnesses characterized by stress-related symptoms, such as emotional and physical fatigue, cognitive difficulty, sleep disturbance, somatic pain and depression.
Previous studies that have investigated the effects of cognitive behavioural training programmes on patients with chronic pain have shown promising results in helping patients to manage their pain and return to work (5). Studies have also shown positive effects of cognitive behavioural interventions for patients with chronic fatigue syndrome, mainly on physical functioning and ratings of fatigue (6). Since everyday stress may create a sense of time urgency, a need to accomplish more in less time, and increased aggressiveness, which are aspects of type A behaviour (7), it seems reasonable that behavioural approaches to reduce type A behaviour may improve symptoms of stress. Cognitive behavioural training programmes may thus be beneficial to patients with stress- related illnesses, in introducing the patients to positive coping strategies and stimulating reflection and understanding of their behaviour in different situations.
Research also indicates that regular physical activity can reduce stress-related symptoms (8, 9). Thus, for patients with stress-related illnesses, appropriate physical activity might not only improve their fitness, but also diminish their symptoms of anxiety, depression, and sleep disturbances (10). In a review of physical exercise in patients with fibromyalgia, chronic pain and chronic fatigue syndrome, Mannerkorpi & Iversen (11) suggested that pool exercise may alleviate the patients’ symptoms of distress, and that aerobic exercise may improve their physical functioning and reduce tenderness. Due to the similarity in symptoms between these patients and patients with stress-related illnesses, this may also apply to the latter patient group.
Although patients’ own assessments of treatment effects are important for the evaluation of rehabilitation programmes, they are subjective and influenced by a number of factors, such as the patients’ expectations of the treatment (12). Therefore, objective methods of assessment could provide useful information about the effects of rehabilitation programmes on symptom severity. In a previous study on patients with stress-related illnesses (4), we found signs of higher autonomic reactivity to standardized laboratory tasks in patients compared with healthy control subjects. The patients also had lower pressure-pain thresholds in the shoulders and lower back than their healthy counterparts.
The aim of the present study was to evaluate the effects of a cognitive behavioural training programme and a physical activity programme, compared with usual care, for patients with stress-related illnesses. Post-intervention effects on physiological and psychological symptoms and return-to-work were followed up for 12 months.
METHODS
Subjects
A total of 75 patients (15 men and 60 women, aged 44 years (standard deviation (SD) 9)) were included in the study. They were selected on the basis of their illness certificate, issued by their primary care physician, and included in the study if they met the following inclusion criteria: (i) 25–60 years of age; (ii) on sick leave for at least 50% of the time for between 1 month and 2 years; and (iii) stress-related diagnoses stated as the cause of the sick leave. Patients were excluded from the study if the certificate indicated that they had any cardiovascular or neurological disease, thyroid disease, diabetes type 2, major depressive disorder, or if they were suffering from substance abuse (e.g. alcohol). An expert panel, consisting of a physician, a physiotherapist and a psychiatrist, made the decision of including patients in the study group. The most common medications taken by the patients were anti-depressants (21 patients), anti-hypertensives (8 patients) and sleeping pills (5 patients). Patients’ diagnoses usually consisted of a stress-related characterization of the illness, followed by an extended description of the symptoms, for example “Stress-related complaints: sleep disturbances, headache, somatic pain”. The most common symptoms of the 75 patients in the study are summarized in Table I. All patients were given verbal descriptions of the study design and the examination protocol before giving their informed consent to participate. The study was approved by the ethics committee at the University of Umeå (project no. 01–132).
| Table I. Summary of the most common symptoms of the 75 patients. Since each patient may experience more than one symptom, the total frequency of symptoms may exceed the number of patients included in the study. |
| Symptoms | Frequency |
| Functional depression | 25 |
| General fatigue | 17 |
| Sleep disturbances | 12 |
| Somatic pain | 10 |
| Concentration difficulties | 6 |
| Headache | 4 |
| Anxiety | 4 |
Study design
This was a randomized controlled study. Upon inclusion in the study, all patients performed an experimental test procedure (see below). Each patient was then allocated to 1 of 3 groups according to a computer-generated allocation schedule. Group 1 participated in a cognitive behavioural training programme, group 2 participated in a physical activity programme, and group 3, the control group, received usual care throughout the study. After a 10-week intervention period, the test procedure was repeated, and follow-up tests were performed at 6 and 12 months after the intervention. The study was repeated successively so that each group contained approximately 8–10 patients, giving a total of 28 patients in the cognitive behavioural training programme, 23 patients in the physical activity programme and 24 patients in the control condition.
Interventions
Cognitive behavioural training. The cognitive behavioural training programme focused on cognitive restructuring to improve participants’ self-care behaviour (strategies for coping with negative emotions, daily routines with regular relaxation and physical activity, and eating and sleeping habits) and social support (building a support network) (5, 6, 13). It comprised 2 three-hour group sessions per week for 10 weeks, and was based on a manual (14) that was given to all participants. The sessions contained educational elements in the form of seminars, group discussions, and required daily practice of skills. Table II summarizes the content of the cognitive behavioural training programme. During the programme, each participant formulated a life-guiding plan, and actively participated in the supporting network of the group. The sessions were led by a stress-management consultant (K.S.), with experience of rehabilitation of patients with stress-related illnesses. The cognitive behavioural training group assembled at 1.5, 3 and 6 months after the intervention.
| Table II. The cognitive behavioural training programme. Each session contained an educational element, and a task was given as homework. The subsequent session started with a group discussion about the homework, followed by an educational part, then new homework was given to the participants. During the 10-week intervention period, the participants kept a homework diary. |
| Session | Main topics covered |
| 1 | Introduction |
| 2 | Stress, definitions and consequences |
| 3 | Stress and burn-out |
| 4 | Stress and personality (type A, B and D), coping strategies |
| 5 | Self-care behaviour (part a: diet), relaxation techniques |
| 6 | Introduction to Qigong, relaxation techniques |
| 7 | Self-care behaviour (part b: physical activity), relaxation techniques, building a supporting network within the group |
| 8 | Invited speaker with own experience of stress-related illness, relaxation techniques |
| 9 | Introduction to how the brain works regarding emotions, relaxation techniques |
| 10 | Anxiety and depression, relaxation techniques |
| 11 | Anxiety and depression, emotional coping strategies, relaxation techniques |
| 12 | Introduction to Feldenkrais, stress management exercises, relaxation techniques |
| 13 | Existential issues, relaxation techniques |
| 14 | Life-guiding plan, relaxation techniques |
| 15 | How to set goals, stress management exercises, relaxation techniques |
| 16 | Awareness of and coping with emotions, stress management exercises, relaxation techniques |
| 17 | Formulating goals, stress management exercises, relaxation techniques |
| 18 | Life-guiding plan, stress management exercises, relaxation techniques |
| 19 | Completing the life-guiding plan, relaxation techniques |
| 20 | Concluding remarks, focusing on where to go from here, relaxation techniques |
Physical activity. Participants in the physical activity group were offered 2 exercise sessions per week for 10 weeks. One of the sessions followed a rehabilitation programme with low-intensity exercises in a warm water pool (32°C) (11). Prior to this session, the participants met with the group leader to discuss their progress, and the difficulties they were facing during exercise. For the other session, the participants chose an exercise (e.g. strength training, swimming, aerobics or walking) in consultation with the group leader, a physiotherapist with experience of rehabilitation of patients with stress-related illnesses. During the intervention, each participant kept a diary of their physical exercise. The physical activity group assembled at 1.5, 3 and 6 months after the intervention.
Control condition. Participants in the control group were offered usual care provided by the Swedish social insurance system during the course of the study. They were promised treatment after the study was completed.
Experimental test procedure
The test procedure comprised 3 parts, all of which were performed on one occasion in a laboratory. Subjects performed tests of autonomic regulation and algometric tests, and completed questionnaires about physical and mental health and behavioural patterns, in randomized order. Each subject was given the same order of measurements throughout the study, and the experimenters were not informed of the group to which the subjects belonged. A detailed description of the test procedure and measurements has been given previously (4).
Autonomic regulation tests
Subjects performed mental arithmetic (2.0 min), handgrip (1.7 min), and deep breathing at a rate of 6 breaths/min (1.2 min). The tasks were presented in randomized order, with rest periods in between, but each subject was given the same order of tasks throughout the study. During the rest periods and tasks, as well as during an initial rest period (5 min), an electrocardiogram was recorded from the wrist-thorax derivation with the ground electrode on the left elbow, and respiration rate was monitored through a strain gauge wrapped around the chest. Skin blood flow was measured with photoplethysmography in the left index finger. Electrodermal activity (EDA) was assessed by the skin-conductance method where 0.5 V was applied to the skin via electrodes that were placed on the thenar and hypothenar of the left hand. Beat-to-beat blood pressure was measured with finger cuffs that were placed around the subject’s left middle and ring fingers (Portapres Model–2, TNO TPD Biomedical Instrumentation, The Netherlands). All data were sampled simultaneously at 2 kHz using a multi-channel polygraph (LABLINC V System, Coulbourn Instruments, PA, USA).
The recordings were processed in Spike 5 (Cambridge Electronic Design Ltd, Cambridge, UK), and heart rate variability analyses were performed in Matlab 6.5 (Mathworks Inc., Natick, MA, USA). For each experimental stage (rest periods and tasks), intervals of ≥ 90 consecutive heartbeats were analysed. The heart rate was re-sampled at 5 Hz after cubic spline interpolation, and spectral analysis was performed using autoregressive (AR) modelling of order 16–70 after linear trend removal. Model coefficients were estimated by the least squares method (15). From the spectrum, the spectral density of the low frequency (LF, 0.04–0.15 Hz) and high frequency (HF, 0.15–0.40 Hz) components was calculated (16). The respiratory signal was re-sampled at the occurrence of each QRS complex, and spectral analysis was performed as described for heart rate (AR order 19–40). The peak frequency was used as an estimate of respiration rate. Skin blood flow was assessed by the rectified photoplethysmogram signal area. Electrodermal responses were identified throughout the recording as any change in skin conductance higher than 3 times the standard deviation (3 × SD). The area under the rectified signal was also calculated. Due to repeated technical problems during data collection, the blood pressure measurements were excluded from the analysis. For the remaining measures, reactivity indices for the tasks were calculated as the value during the task minus the value during the rest period immediately before the task.
Algometric measurements
Pressure-pain thresholds in the trapezius pars descendens were assessed by applying a pressure algometer (Somedic Production AB, Sollentuna, Sweden) approximately half way between C7 and the acromion, while the subject was sitting in an upright position. For the erector spinae muscle, the algometer was applied on the muscle belly at the level of L3–L4, while the subject was lying in prone position. Pressure was increased at a rate of approximately 50 kPa/cm2/sec, and subjects pressed a button as soon as the pressure sensation became a pain sensation. The pressure value was then registered by the examiner. Measurements were performed bilaterally by alternating between the left and right side. For each measurement location, 5 algometric recordings were obtained. These recordings were sorted by size, and the smallest and largest values were excluded from further analysis. From the remaining 3 recordings, a mean value was computed and used as outcome measure.
Questionnaires
Subjects completed the Short Form 36 Item Health Survey (SF-36) (17, 18) to assess their general physical and mental health, the Shirom-Melamed Burnout Questionnaire (19) for the assessment of different aspects of burn-out, the Coping Resources Inventory (CRI) (20) of perceived resources during stressful situations, a short version of the Jenkins Activity Survey (JAS) (21) containing questions about behavioural patterns, and the Karolinska Sleep Questionnaire (KSQ) (22) to assess sleep quality and sleep complaints. At the time of the first test procedure (i.e. before the intervention), subjects rated their expectations of cognitive behavioural training and physical activity, respectively, as well as their expectations of their health in general on 7-point rating scales.
The SF-36 items were summarized in 8 indices (i.e. physical functioning, physical role limitation, pain, general health, vitality, social functioning, emotional role limitation and mental health), according to the SF-36 manual (SF-36 Health Survey: Manual & Interpretation Guide). For the Shirom-Melamed Burnout Questionnaire, indices for burn-out, tension, listlessness and cognitive difficulties were calculated (23, 24). The CRI items yielded indices for measuring resources in 5 domains: cognitive, social, emotional, spiritual/philosophical, and physical (20), and a mean value of the 11 items in JAS was calculated and used as a JAS index. Finally, 2 KSQ indices were computed by averaging the items of Disturbed sleep and Awakening (25). The constructed indices had Cronbach’s α > 0.7, with the exception of the CRI index "spiritual/philosophical", which had a value of 0.64.
Statistical analysis. Statistical analyses were performed in SPSS 11.0 (SPSS Inc., Chicago, IL, USA). Baseline characteristics were compared between patient groups using parametric or non-parametric analyses of variance, depending on the distribution of data. For the autonomic reactivity indices, repeated-measures analyses of variance with Time and Task as within-subjects variables, and Group and Gender as between-subjects variables were employed to assess changes over time. Similarly, repeated-measures analyses of variance with Time, Group and Gender as variables were performed for the algometric measurements and the questionnaire ratings. Whenever the sphericity assumption was not met, the Huynh-Feldt correction was used. Finally, the number of patients working part-time or full-time throughout the study was compared between groups with χ2 tests. In all tests, p < 0.05 was considered significant. Due to the large number of tests performed, Holm’s sequentially rejective procedure was used to adjust for multiple tests (26).
Power analyses showed that 75 subjects would be sufficient for detecting minimum group differences in reactivity of 5 beats/min in heart rate, 21 seco2 in heart rate variability, 5 responses/min and 4 µS/min in EDA, 19 arbitrary units (a.u.)/min in skin blood flow, 4 breaths/min in respiration rate, as well as 165 kPa in algometric measurements, 26% in SF-36 ratings, 6 units in CRI ratings, 0.5 and 1.0 units in JAS and KSQ ratings, respectively, and 1.5 in Shirom-Melamed Burnout Questionnaire ratings, with statistical power of at least 0.95. Data are presented in the text and tables as mean and SD, and in the graphs as mean and standard error of the mean (SEM).
RESULTS
Of the 75 patients, 15% (2 men and 9 women) decided to withdraw during the course of the study (8 patients withdrew during the intervention period, 2 patients withdrew by the 6-month follow-up assessment, and one patient withdrew by the 12-month follow-up assessment). Eight of the patients who withdrew were from the cognitive behavioural training group, one from the physical activity group, and 2 from the control group. Demographic characteristics of all patients are shown in Table III. No differences were found between those patients who completed the study and those who did not (p > 0.05).
During the intervention period, patients in the cognitive behavioural training group attended 19 (SD = 1) out of 20 sessions. The majority of the patients (74%) also reported having put their knowledge into practice by the end of the programme. In the physical activity group, patients participated in 8 (SD = 2) of 10 pool exercises, and reported a higher level of physical exercise than patients in the cognitive behavioural training group at all follow-up occasions (χ2(2) > 6.2, p < 0.046). There were no differences between the groups in patients’ expectations of the treatment received (χ2(1) = 0.06, p = 0.801). In addition, there were no differences between the 3 patient groups in their expectations of their health in the near future (χ2(2) = 1.03, p = 0.597).
| Table III. Demographic characteristics of the patients in the cognitive behavioural training group, the physical activity group, and the control group. |
| | Cognitive behavioural (n = 28) | Physical activity (n = 23) | Control (n = 24) |
| Age, years (mean (SD)) | 44 (8) | 44 (10) | 44 (9) |
| Height, cm (mean (SD)) | 171 (8) | 170 (8) | 169 (6) |
| Weight, kg (mean (SD)) | 70 (12) | 75 (17) | 76 (11) |
| Smoking, n (%) | | | |
| | Present smoker | 3 (11) | 1 (4) | 1 (4) |
| | Non-smoker | 25 (89) | 22 (96) | 23 (96) |
| Smokeless tobacco use, n (%) | | | |
| | Yes | 7 (25) | 4 (17) | 4 (17) |
| | No | 21 (75) | 19 (83) | 20 (83) |
| Marital status, n (%) | | | |
| | Unmarried | 2 (7) | 2 (8) | 0 (0) |
| | Married/cohabiting | 20 (72) | 19 (83) | 19 (79) |
| | Divorced | 6 (21) | 2 (9) | 5 (21) |
| Sick leave duration, months (mean (SD)) | 9 (7) | 9 (7) | 8 (6) |
Autonomic activity
When the autonomic variables during the initial rest period at the first measurement occasion were compared between the 3 patient groups, no significant differences were found (p > 0.05). For the autonomic reactivity indices, differences between the patient groups were found in heart rate variability (low, high, and total spectral density) in response to deep breathing (LF: F(2,68) = 4.2, p = 0.020; HF: F(2,68) = 3.7, p = 0.031; Total: F(2,68) = 4.4, p = 0.016). Patients who were later allocated to the cognitive behavioural training group had higher reactivity than the control group patients.
At the second measurement occasion, i.e. after the intervention period, the patients showed lower heart rate reactivity to the tasks (HR: F(1,56) = 26.6, p < 0.001; LF: F(1,57) = 8.6, p = 0.005; HF: F(1,57) = 11.2, p = 0.001; Total: F(1,57) = 13.8, p <0.001), and to the mental arithmetic task in particular (HR: F(2,112) = 5.3, p = 0.006), than before the intervention. For patients in the cognitive behavioural training group, a stronger electrodermal response to deep breathing was observed, compared with patients in the physical activity group (F(4,99) = 2.8, p = 0.037). Furthermore, patients in the cognitive behavioural training group exhibited reduced heart rate variability in response to deep breathing than the other patient groups, compared with before the intervention (LF: F(3,96) = 3.2, p = 0.022; HF: F(3,85) = 3.7, p = 0.014; Total: F(3,92) = 3.3, p = 0.022). The previously observed group differences in heart rate variability in response to deep breathing were no longer present (ANOVA, p > 0.05).
The immediate effects of the intervention period remained at 6 and 12 months after the intervention. Overall, the patients exhibited a relative decrease in autonomic reactivity to the tasks (HR: F(3,153) = 17.1, p < 0.001; EDA area: F(3,137) = 6.0, p = 0.001; EDA responses: F(2,124) = 8.2, p < 0.001). For patients in the cognitive behavioural training group, the effect of deep breathing on their heart rate variability was lower than before the intervention (HF: F(8,213) = 2.6, p = 0.009). After correcting for multiple tests, only the overall decrease in autonomic reactivity over time remained significant.
Autonomic activity during the initial rest periods also changed significantly over time. Throughout the study, EDA decreased (EDA area: F(3,152) = 3.1, p = 0.029; EDA responses: F(2,123) = 26.5, p < 0.001), and skin blood flow increased (F(3,156) = 2.7, p = 0.045). No differences between patient groups were found (p > 0.05).
Pressure-pain thresholds
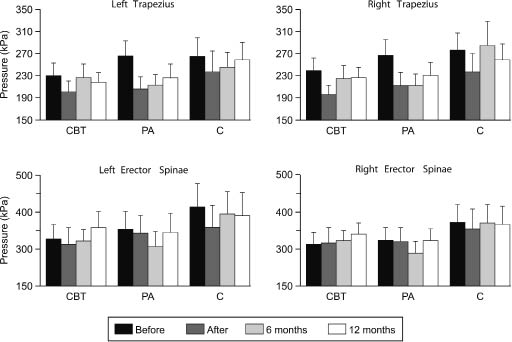
Initial algometric recordings revealed no differences in pressure-pain thresholds between the patient groups (p > 0.05). Immediately after the intervention period, all the groups demonstrated lower pressure-pain thresholds in the right trapezius (F(1,56) = 7.5, p = 0.008) (Fig. 1). Patients in the control group also showed lower pressure-pain thresholds in erector spinae compared with the treatment groups (right: F(2,56) = 3.4, p = 0.041).
Fig. 1. Mean values of pressure-pain thresholds in trapezius and erector spinae for patients in the cognitive behavioural training group (CBT), the physical activity group (PA), and the control group (C). The T bars represent +1 standard error of the mean.
At 6 months after the intervention, the patients’ pressure-pain thresholds in trapezius had increased (right: F(2,97) = 3.5, p = 0.039), and the previously observed group differences were no longer present (F(4,104) = 0.6, p = 0.634). Similar results were found at 12 months after the intervention. When correcting for multiple tests, only overall time effects remained significant.
Subjective ratings
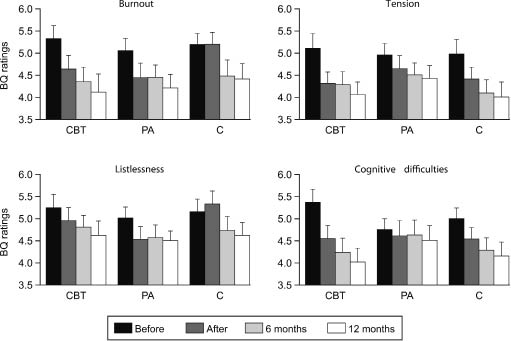
No significant differences in questionnaire ratings were found between the patient groups at the first measurement occasion (p > 0.05). After the intervention period, patients’ SF-36 ratings of physical role limitations, bodily pain, vitality, social functioning and mental health had improved (p < 0.05). Patients also rated lower levels of burn-out (Burnout: F(1,61) = 10.0, p = 0.002; Tension: F(1,61) = 20.3, p < 0.001; Cognitive difficulties: F(1,61) = 6.4, p = 0.014, Fig. 2), and weaker type A behavioural pattern (Before: 2.8 (SD = 0.4); After 2.7 (SD = 0.4); F(1,61) = 7.9, p = 0.007) than before the intervention. Significant group differences were found in SF-36 ratings of general health (F(2,59) = 3.8, p = 0.027), where the cognitive behavioural training group showed an improvement compared with the physical activity group. Furthermore, patients in the cognitive behavioural training group reduced their burn-out ratings compared with the control group (F(2,61) = 3.9, p = 0.024). There were no differences in patients’ CRI ratings, compared with the first measurement occasion (p > 0.05).
Fig. 2. Mean values of ratings in Shirom-Melamed Burnout Questionnaire (BQ) for patients in the cognitive behavioural training group (CBT), the physical activity group (PA), and the control group (C). High values correspond to high levels of burn-out. The T bars represent +1 standard error of the mean.
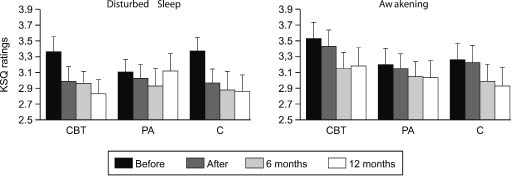
Fig. 3. Mean values of ratings in the Karolinska Sleep Questionnaire (KSQ) for patients in the cognitive behavioural training group (CBT), the physical activity group (PA), and the control group (C). High values correspond to high levels of sleep disturbances. The T bars represent +1 standard error of the mean.
At the 6-month follow-up assessment, patients’ ratings of physical and mental health, burn-out, and behavioural patterns continued to improve. They also reported having fewer sleep disturbances (Disturbed sleep: F(2,116) = 6.4, p = 0.002; Awakening: F(2,116) = 4.0, p = 0.020; Fig. 3). Patient groups no longer differed in ratings of burn-out (F(4,116) = 1.3, p = 0.062), but patients in the cognitive behavioural training group had higher ratings of general health than patients in the physical activity group (F(4,112) = 3.4, p = 0.011). No differences between the patient groups were found in CRI ratings (p > 0.05). At 12 months after the intervention, similar results were found. After correcting for multiple tests, only overall time effects remained significant.
The number of patients working part-time or full-time did not differ between the groups before or after the intervention period (χ2(2) < 2.9, p > 0.230, Table IV). When logistic regression analyses were employed to assess whether any of the initial measures could explain patients’ improvement at 12 months after the intervention, the results showed that patients’ expectations of their health as well as their CRI ratings of perceived resources in the cognitive domain significantly explained their return-to-work (Expectations: p = 0.018; CRI cognitive: p = 0.044). Higher ratings of expectation of health and cognitive resources increased the probability of having returned to work by 12 months after the intervention period (Odds ratio and confidence interval: Expectations 2.54 [1.17 – 5.50]; CRI cognitive: 1.13 [1.00 – 1.28]).
| Table IV. Number of patients working part-time or full-time throughout the study in the cognitive behavioural training group (CBT, n = 28), the physical activity group (PA, n = 23), and the control group (C, n = 24). |
| | Before intervention | After intervention | 6 months after intervention | 12 months after intervention |
| CBT | | 9 | 8 | 7 | 11 |
| PA |
| | 4 | 6 | 9 | 8 |
| C |
| | 4 | 9 | 11 | 12 |
Confirmatory analyses
Throughout the course of the study, some patients engaged in other treatments or changed their medication. To avoid confounding effects of such changes on the outcome of the cognitive behavioural training programme and the physical activity programme, all analyses were performed on a subset of patients (23%) who did not engage in other treatments or changed their medication during the study. The results were largely similar to those found previously. However, some CRI ratings now improved significantly over time (Philosophical: F(3,24) = 3.6, p = 0.028; Physical: F(3,24) = 5.9, p = 0.004), and patients in the cognitive behavioural training group rated a higher level of coping resources than patients in the physical activity group (Cognitive: F(6,24) = 2.9, p = 0.029). They also reported weaker type A behavioural patterns than patients in the physical activity group, compared with before the intervention (F(2,9) = 6.7, p = 0.016). At 6 and 12 months after the intervention, patients in the physical activity group were more likely to be on sick leave than patients in the other groups (χ2(2) > 8.2, p < 0.017).
Additional analyses were performed on patients without prescribed medication (44%). The results were similar to those obtained in the original analyses.
DISCUSSION
When examining the effects of a cognitive behavioural training programme and a physical activity programme, as compared with usual care, for patients with stress-related illnesses, only minor differences were found. The number of patients returning to work did not differ between the groups throughout the study. Additional analyses revealed that the patients’ initial ratings of cognitive resources and expectations of health were informative measures of their return to work at 12 months after the intervention. The result underlines the need to incorporate patients’ expectations of their health in the rehabilitation process.
Autonomic activity
Throughout the study, the patients exhibited a relative decrease in autonomic reactivity to the tasks. This may suggest that the patients reduced their autonomic response to stressors in general, or it may reflect a habituation to the experimental protocol used in the present study. In a previous study, we found that patients with stress-related illnesses reacted somewhat more strongly to the mental arithmetic and the handgrip task than did healthy control subjects (4). The fact that no difference in autonomic reactivity to either mental arithmetic or handgrip was found between the patient groups in the present study might suggest that the outcome measures were not sensitive enough to detect small changes in autonomic reactivity, or that the treatments had no effect on the patients’ autonomic reactivity to these kinds of mentally and physically demanding tasks.
Our result differs from those of Vocks et al. (27), who found that patients with enhanced blood pressure reactivity who participated in a cognitive behavioural stress management programme reduced their blood pressure reactivity to a mentally demanding task, compared with a control group who received progressive muscular relaxation training. Granath et al. (28), on the other hand, found that subjects with self-reported stress-related problems improved regardless of whether they received cognitive behavioural therapy or yoga.
Pressure-pain thresholds
In the present study, we found short-term, but not long-term, effects of cognitive behavioural training and physical activity on pressure-pain thresholds in erector spinae. The results are in agreement with previous studies of treatments for pain patients, where only minor changes in pressure-pain thresholds were reported (29, 30). Schreiber et al. (31) demonstrated higher pressure-pain thresholds in patients with major depression immediately after receiving electroconvulsive treatment. Unfortunately, no control group was included in that study, thus the observed change in pressure-pain thresholds cannot be attributed solely to the treatment.
After the intervention period, patients’ pressure-pain thresholds in the trapezius muscle decreased. One explanation for this difference might be that the patients became more aware of their illness after entering the study. Another explanation might be that they experienced some discomfort following the first measurement, and therefore tended to press the button registering the pressure-pain threshold sooner than the first time.
Subjective ratings
Although the ratings of burn-out differed between the cognitive behavioural training group and the control group after the intervention, but not at the follow-up assessments, patients in the cognitive behavioural training group rated improved general health (SF-36) compared with patients in the physical activity group until 12 months after the intervention period. Jensen et al. (32) showed that female pain patients on sick leave who received cognitive behavioural therapy reported better SF-36 ratings (general health, mental health and social functioning) than patients who received “treatment-as-usual”. However, they found no difference in absenteeism from work between the treatment groups. Similarly, we found no effect of cognitive behavioural training or physical activity on patients’ sick leave.
In general, the results of the study showed an improvement in the control group of about the same magnitude as in the treatment groups over the 12-month follow-up period. It is possible that the attention given to these patients by performing the measurements affected the results obtained from this group. However, it is likely that this effect is equal between the 3 groups, since the same attention was given to all patients. The small differences between the groups in autonomic activity, pressure-pain thresholds and subjective ratings that we found suggest little effect of the cognitive behavioural training programme and the physical activity programme for the patients on these parameters. A reason for this may be that the patients had been on sick leave for a long time (~9 months) before entering the study. Marhold et al. (5) found that pain patients who had been on short-term sick leave (2–6 months) were more likely to return to work after participating in a cognitive behavioural return-to-work programme, compared with control group patients, than patients who had been on long-term sick leave (> 12 months). Furthermore, the short-term sick leave patients rated better health after the programme than the long-term sick leave patients. In this study, we evaluated the effects of a cognitive behavioural training programme and a physical activity programme, respectively. It may be that a combination of the 2 programmes would have yielded better results, although it would have required more time and effort from the patients. It is also possible that by involving the patients’ workplaces to a greater extent in the rehabilitation, the probability of patients returning to work would increase.
There were some limitations to the present study that should be addressed. The selection of patients was based solely on what was specified in their illness certificate, issued by their primary care physician. Thus, patients’ own opinions of their illness were not considered in the screening process. Despite the fact that patients with diagnoses of major depressive disorder or cardiovascular disease were excluded from the study, some patients reported taking anti-depressants and anti-hypertensives, suggesting a diverse management and treatment of these patients. The patients taking these medications, however, were evenly distributed across the groups. Due to the high proportion of females among the patients, no gender differences were investigated. The over-representation of female patients may reflect gender differences in the reporting of stress-related symptoms (33, 34). More patients in the cognitive behavioural training group than in the other groups decided to withdraw from the study (cognitive behavioural training group: 29%; physical activity group: 4%; control group: 8%), which may have introduced bias in the results by having patients that are particularly sympathetic towards the received treatment in the cognitive behavioural training group. One may argue that the 12-month follow-up period was short, when considering the patients’ history of sick leave. However, the inclusion of a control group of patients who were not offered any treatment during the study limited the follow-up assessment period due to ethical considerations.
In conclusion, this study showed little difference in the effect of cognitive behavioural training and physical activity, compared with usual care, for patients on long-term sick leave due to stress-related illnesses. Further studies are needed to determine whether either treatment, or a combination of the treatments, would be more suitable for patients with shorter sick leave or at earlier stages of the illness.
ACKNOWLEDGEMENTS
This study was presented in part at the 2nd ICOH International Conference on Psychosocial Factors at Work, Okayama Japan, August 23–26, 2005.
The authors thank Dr Stefan Backgård at the Swedish Social Insurance Organization for expert advice in patient selection, and Anna-Sofia Magnusson, Christine Gruffman and Eva Tengman at IKSU Sportcenter for organizing the physical activity programme. The authors also thank Dr Tohr Nilsson for critical remarks on the manuscript, and Margaretha Marklund for valuable assistance during data collection and analysis, as well as for graphical work. The study was supported by grants from VINNOVA (project no. 510240).
REFERENCES
1. Hurrell JJ, Murphy LR. Occupational stress intervention. Am J Ind Med 1996; 29: 338–341.
2. van der Klink JJ, van Dijk FJ. Dutch practice guidelines for managing adjustment disorders in occupational and primary health care. Scand J Work Environ Health 2003; 29: 478–487.
3. De Vente W, Olff M, Van Amsterdam JG, Kamphuis JH, Emmelkamp PM. Physiological differences between burnout patients and healthy controls: blood pressure, heart rate, and cortisol responses. Occup Environ Med 2003; 60 Suppl 1: i54–61.
4. Heiden M, Barnekow-Bergkvist M, Nakata M, Lyskov E. Autonomic activity, pain, and perceived health in patients on sick leave due to stress-related illnesses. Integr Physiol Behav Sci 2005; 40: 3–16.
5. Marhold C, Linton SJ, Melin L. A cognitive-behavioral return-to-work program: effects on pain patients with a history of long-term versus short-term sick leave. Pain 2001; 91: 155–163.
6. Deale A, Chalder T, Marks I, Wessely S. Cognitive behavior therapy for chronic fatigue syndrome: a randomized controlled trial. Am J Psychiatry 1997; 154: 408–414.
7. Friedman M, Rosenman RH, editors. Type A behavior and your heart. New York: Fawcett Crest; 1974.
8. Jennen C, Uhlenbruck G. Exercise and life-satisfactory-fitness: complementary strategies in the prevention and rehabilitation of illnesses. Evid Based Complement Alternat Med 2004; 1: 157–165.
9. Carmack CL, Boudreaux E, Amaral-Melendez M, Brantley PJ, de Moor C. Aerobic fitness and leisure physical activity as moderators of the stress-illness relation. Ann Behav Med 1999; 21: 251–257.
10. Fox KR. The influence of physical activity on mental well-being. Public Health Nutr 1999; 2: 411–418.
11. Mannerkorpi K, Iversen MD. Physical exercise in fibromyalgia and related syndromes. Best Pract Res Clin Rheumatol 2003; 17: 629–647.
12. Goossens ME, Vlaeyen JW, Hidding A, Kole-Snijders A, Evers SM. Treatment expectancy affects the outcome of cognitive- behavioral interventions in chronic pain. Clin J Pain 2005; 21: 18–26, 69–72.
13. Burell G, Öhman A, Sundin Ö, Ström G, Ramund B, Cullhed I, et al. Modification of the type A behavior pattern in post-myocardial infarction patients: a route to cardiac rehabilitation. Int J Behav Med 1994; 1: 32–54.
14. Sahlin T, Hofvendahl S, editors. Open your heart: different ways to managing stress. [Öppna ditt hjärta: olika vägar att hantera stress.] Sweden: Novartis; 1998 (in Swedish).
15. Marple SLJ, editors. Digital spectral analysis: with appplications. Englewood Cliffs, NJ: Prentice-Hall; 1987.
16. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Heart rate variability – standards of measurement, physiological interpretation, and clinical use. Circulation 1996; 93: 1043–1065.
17. Ware JEJ, Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care 1992; 30: 473–483.
18. Sullivan M, Karlsson J, Ware JEJ. The Swedish SF-36 Health Survey-I. Evaluation of data quality, scaling assumptions, reliability and construct validity across general populations in Sweden. Soc Sci Med 1995; 41: 1349–1358.
19. Melamed S, Kushnir T, Shirom A. Burnout and risk factors for cardiovascular diseases. Behav Med 1992; 18: 53–60.
20. Hammer AL, Marting MS, editors. Manual for the Coping Resources Inventory. Palo Alto, CA: Consulting Psychologists Press; 1988.
21. Jenkins CD, Rosenman RH, Friedman M. Development of an objective psychological test for the determination of the coronary-prone behavior pattern in employed men. J Chronic Dis 1967; 20: 371–379.
22. Kecklund G, Åkerstedt T. The psychometric properties of the Karolinska sleep questionnaire. J Sleep Res 1992; 1: 113.
23. Melamed S, Ugarten U, Shirom A, Kahana L, Lerman Y, Froom P. Chronic burnout, somatic arousal and elevated salivary cortisol levels. J Psychosom Res 1999; 46: 591–598.
24. Soares JJ, Jablonska B. Psychosocial experiences among primary care patients with and without musculoskeletal pain. Eur J Pain 2004; 8: 79–89.
25. Åkerstedt T, Knutsson A, Westerholm P, Theorell T, Alfredsson L, Kecklund G. Sleep disturbances, work stress and work hours: a cross-sectional study. J Psychosom Res 2002; 53: 741–748.
26. Holm S. A simple sequentially rejective multiple test procedure. Scand J Statist 1979; 6: 65–70.
27. Vocks S, Ockenfels M, Jurgensen R, Mussgay L, Ruddel H. Blood pressure reactivity can be reduced by a cognitive behavioral stress management program. Int J Behav Med 2004; 11: 63–70.
28. Granath J, Ingvarsson S, von Thiele U, Lundberg U. Stress management: a randomized study of cognitive behavioural therapy and yoga. Cogn Behav Ther 2006; 35: 3–10.
29. Waling K, Järvholm B, Sundelin G. Effects of training on female trapezius myalgia: An intervention study with a 3-year follow-up period. Spine 2002; 27: 789–796.
30. Taimela S, Takala EP, Asklöf T, Seppälä K, Parviainen S. Active treatment of chronic neck pain: a prospective randomized intervention. Spine 2000; 25: 1021–1027.
31. Schreiber S, Shmueli D, Grunhaus L, Dolberg OT, Feldinger E, Magora F, et al. The influence of electroconvulsive therapy on pain threshold and pain tolerance in major depression patients before, during and after treatment. Eur J Pain 2003; 7: 419–424.
32. Jensen IB, Bergström G, Ljungquist T, Bodin L, Nygren AL. A randomized controlled component analysis of a behavioral medicine rehabilitation program for chronic spinal pain: are the effects dependent on gender? Pain 2001; 91: 65–78.
33. Nakao M, Fricchione G, Zuttermeister PC, Myers P, Barsky AJ, Benson H. Effects of gender and marital status on somatic symptoms of patients attending a mind/body medicine clinic. Behav Med 2001; 26: 159–168.
34. Collins PA, Gibbs AC. Stress in police officers: a study of the origins, prevalence and severity of stress-related symptoms within a county police force. Occup Med (Lond) 2003; 53: 256–264.