Selma Eroglu, MD1, Esra Erkol Inal, MD2, Mehmet Eroglu, MD3, Serdar Oruc, MD4,
Alper Murat Ulasli, MD1, Halime Cevik, MD1, Seden Demirci, MD5, Ozlem Solak, MD1,
Umit Dundar, MD1, Hasan Toktas, MD1 and Mehmet Yaman, MD4
From the 1Department of Physical Medicine and Rehabilitation, School of Medicine, Afyon Kocatepe University,
Afyonkarahisar, 2Department of Physical Medicine and Rehabilitation, School of Medicine, Suleyman Demirel
University, Isparta, 3Department of Orthopaedics and Traumatology, 4Department of Neurology, School of Medicine, Afyon Kocatepe University, Afyonkarahisar and 5Department of Neurology, School of Medicine, Suleyman Demirel University, Isparta, Turkey
OBJECTIVE: Early degeneration of the knees might occur in patients with multiple sclerosis secondary to balance and walking impairment and muscle weakness. The aims of this study were to evaluate the knee joints of patients with multiple sclerosis compared with healthy controls, using ultrasonography, and to investigate whether there is any correlation between femoral cartilage degeneration and disease-related parameters.
DESIGN: Study participants were 79 patients with multiple sclerosis and 60 healthy controls. The disease-related parameters, Expanded Disability Status Scale (EDSS), Western Ontario and McMaster Universities (WOMAC) osteoarthritis index, visual analogue scale (VAS) for pain severity, and Berg Balance Scale (BBS) scores were recorded. Femoral cartilage and knee effusion were evaluated using ultrasonography.
RESULTS: Femoral cartilages of patients with multiple sclerosis were more degenerated than those of healthy controls. Moreover, patients with multiple sclerosis had more effusion in their knees than did controls. In the multiple sclerosis group there was no correlation between cartilage degeneration grade, amount of effusion, and VAS-pain, BBS, WOMAC and EDSS scores.
CONCLUSION: Patients with multiple sclerosis may have more rapid degeneration of the knee cartilage and increased effusion compared with healthy controls. Ultrasonography is an effective method to detect these changes. However, cartilage degeneration was not found to be associated with disease-related parameters in multiple sclerosis.
Key words: multiple sclerosis; cartilage degeneration; ultrasonography; EDSS; Berg Balance Scale.
J Rehabil Med 2016; 48: 604–608
Correspondence address: Selma Eroglu, Department of Physical Medicine and Rehabilitation, Faculty of Medicine, Afyon Kocatepe University, Afyonkarahisar, TR-03200, Turkey. E-mail address: seroglu79@gmail.com
Accepted Apr 21, 2016; Epub ahead of print Jun 16, 2016
INTRODUCTION
Multiple sclerosis (MS) is an immune-mediated progressive disease that leads to both demyelination and axonal degeneration within the central nervous system (CNS) (1, 2). These pathological changes may result in various impairments, including motor (muscle weakness, increased tone, fatigue), sensory, autonomic (bladder dysfunction, etc.), cognitive and visual dysfunction (1–3). The cumulative impact of lesions throughout the CNS leads to clinical manifestations. Since nerve dysfunction increases with the length of the pathways, MS predominantly affects the most caudal nerves, i.e. muscles and sensory structures of the lower limbs and bladder. The progressive disability in MS is more prominent in the lower limbs, and MS patients eventually experience walking problems (1) due to a combination of motor and sensory dysfunction and balance disturbance (1, 4).
The hypothesis of this study was that the knee joint would be affected due to balance disturbance and muscle weakness related to MS and earlier degeneration might occur. A few studies on neurologically impaired patients (stroke and spinal cord injury) have reported changes in the femoral cartilage (5–7). However, to our knowledge, no study has evaluated the knee and femoral cartilage in patients with MS.
The aims of this study were therefore to evaluate the knee joints of patients with MS, using ultrasonography (US) to compare the results with those for healthy controls, and to investigate whether there is a correlation between the condition of the femoral cartilage and disease-related parameters.
METHODS
Subjects
The study was conducted at a university-based hospital. A total of 82 patients with MS (60 females, 22 males) being followed up at outpatients’ clinic and 60 healthy age- and sex-matched controls (43 females, 17 males) were enrolled in this cross-sectional study. The study protocol was approved by the local ethics committee. All subjects were informed about the study procedure and provided written consent to participate. Initial inclusion criteria were: (i) patients with MS, 18–50 years of age; (ii) EDSS score of < 6.5 (ambulating patient); and (iii) no history of fracture or surgery around the knee, or accompanying rheumatic or neurological diseases. Three subjects were excluded becaues the ultrasonographic assessment could not be done appropriately due to spasticity around the knee, or they could not walk independently. The final study group included 79 patients with MS (58 females, 21 males). Sample size was not based on a formal power analysis, but on the size of similar published studies (6, 7). The control group comprised subjects with no history of rheumatic, neurological or endocrinological diseases or trauma or surgery around the knee.
Demographic features of the subjects, including age, gender, body weight/height, disease duration, type of MS (secondary/primary progressive, relapse-remitting, etc.), number of attacks, and medications used, were noted. Body mass indices (BMI; mass (kg)/height(m2)) of the patients were calculated at the time of examination.
Outcome measures
The Expanded Disability Status Scale (EDSS) (a 0–10 scale, in which 0 represents no impairment due to MS, 4.0 indicates the onset of significant walking impairment, 7.5 indicates wheelchair dependence, and 10.0 represents death due to MS) was used to assess the degree of progressive disability related to MS (8).
The Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) is a self-administered questionnaire to assess symptoms and physical functional disability in patients with knee OA. It includes 24 questions, grouped into 3 subscales (pain, joint stiffness and physical function). In this study, we used a validated Turkish version of WOMAC Likert scale, with 5 alternative answers to each question (0 = none, 1 = mild, 2 = moderate, 3 = severe, 4 = extreme). On the Likert scale, the minimum score is 0 and the maximum score is 96 (20 points for pain, 8 points for joint stiffness, and 68 points for physical function). Higher scores represent poor health, worse symptoms, and more limitations (9–11). Each subscale of the WOMAC index and their relationship with the clinical parameters of MS were evaluated separately in the study and control groups.
The severity of knee pain at the time of examination was assessed with a visual analogue scale (VAS; 0–10 cm), with 0 referring to “no pain” and 10 to “maximum pain” for each knee.
Balance disturbance, a common symptom in MS, was assessed with the Berg Balance Scale (BBS). The BBS is a widely used clinical test to evaluate balance disturbance. It consists of 14 balance-related tasks or items, such as standing, sitting, transfers and standing on 1 leg. Each item is scored from 0 to 4, with 0 indicating severe balance disturbance and 4 indicating normal balance function. Thus, the score achieved can change between 0 and 56. Higher scores indicate better balance (2, 12). All assessments, except EDSS, were also carried out with the control group.
Ultrasonographic examination
Ultrasonographic evaluation of the patients was performed once in the morning (between 08:00–10:00 a.m.) and the patients were asked not to perform any exhausting activities prior to the examination. Femoral cartilage and knee effusion were evaluated with a multi-frequency linear probe (6–18 MHz, MyLab 70 XVG device (Esaote Biomedica, Genoa, Italy). All measurements were performed bilaterally. Distal femoral cartilage was assessed while the subjects lay in a supine position on the examination bed, with their knees in maximum flexion. The probe was placed transversely to the leg just above the superior margin of the patella and perpendicular to the bone surface (13–15). The distal femoral cartilage was visualized as a strongly anechoic structure between the sharp bony cortex and the suprapatellar fat pad. Cartilage bands of the medial and lateral condyles and trochlea were visualized. The system, previously defined by Lee et al., was used to grade the cartilage. Sharpness, clarity and thickness of the cartilage band were assessed for grading. According to the grading system osteoarthritic femoral condylar cartilage was graded from 1 to 6, in which Grade 1 refers to blurred margin or partial lack of the clarity, without thickness change, and Grade 6 refers to no visualized cartilage band (14). Samples for cartilage grading are shown in Fig. 1. Ultrasonographic grading was performed by the same investigator (AMU), who had 6 years’ experience in musculoskeletal ultrasonography. After the US images were recorded, cartilage grading was carried out twice at different times by evaluating the images and intra-observer reliability was calculated.
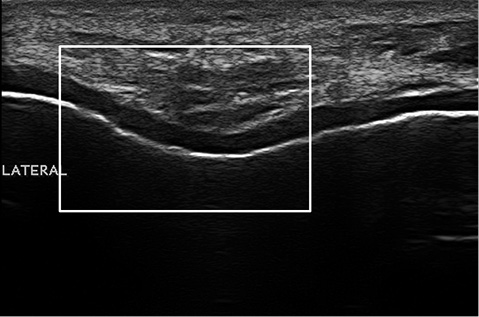
Fig. 1. A cartilage grading sample. Grade 3 femoral cartilage degeneration is detected by a blurred margin and complete lack of clarity.
The presence of effusion was assessed with knees in 30° of flexion. The probe was placed longitudinally over the suprapatellar region and scanning was performed with a fan-like movement of the probe from lateral through medial sides. The effusion was defined as hypoechoic or anechoic area within the suprapatellar bursa and was measured at its greatest thickness both in the medial and lateral aspects (13, 16).
Statistical analysis
Statistical analysis was performed with SPSS 15.0 (IL, USA). Data were expressed as mean (standard deviation (SD)) for continuous variables, and as median (minimum–maximum) for ordinal variables. Mann-Whitney U test was used to compare 2 independent samples. Correlations between patients’ characteristics and femoral cartilage grades were analysed using Spearman’s correlation coefficients. Intra-class correlation coefficient for femoral cartilage grading was calculated for the agreement between assessments performed at 2 different times. Statistical significance was set at p < 0.05.
RESULTS
Measurements regarding 158 knees of 79 MS patients and 120 knees of 60 healthy controls were used for the analysis. The demographic characteristics of the subjects are shown in Table I. The mean age of the patients and controls were not significantly different (p = 0.06). The mean BMI of the patients with MS was lower than that of the control group (p = 0.01). The median femoral cartilage grades and the mean effusion values (mm) in the medial and lateral compartments of the knee and the scores of VAS, EDSS, BBS and WOMAC of the subjects are shown in Table I.
|
Table I. Characteristics and clinical parameters of the patients with multiple sclerosis (MS) and control group |
|||
|
Characteristics and clinical parameters |
MS |
Controls |
p-value |
|
Age, years, mean (SD) |
35.01 (8.2) |
32.4 (8.04) |
0.06 |
|
Height, cm, mean (SD) |
166.5 (9.5) |
164.7 (11.5) |
0.29 |
|
Weight, kg, mean (SD) |
67.8 (13.6) |
71.1 (11.8) |
0.13 |
|
BMI, kg/m2, mean (SD) |
24.4 (4.6) |
26.3 (4.3) |
0.01 |
|
Disease duration, years, mean (SD) |
8.03 (5.5) |
N/A |
N/A |
|
EDSS, median (min–max) |
3 (0.5–7.5) |
N/A |
N/A |
|
Cartilage grade – right knee, median (min–max) |
1 (0–5) |
0 (0–3) |
< 0.001 |
|
Cartilage grade – left knee, median (min–max) |
1 (0–5) |
1 (0–2) |
< 0.001 |
|
Berg Balance Scale, median (min–max) |
51.5 (4–56) |
56 |
< 0.001 |
|
WOMAC Pain, median |
0.5 (0–18) |
0 (0–1) |
< 0.001 |
|
WOMAC Stiffness, median (min–max) |
0 (0–6) |
0 (0–2) |
< 0.001 |
|
WOMAC Function, median (min–max) |
2.5 (0–68) |
1 (0–3) |
< 0.001 |
|
VAS – right knee, mean (SD) |
1.6 (2.4) |
0 |
< 0.001 |
|
VAS – left knee, mean (SD) |
1.6 (2.6) |
0 |
< 0.001 |
|
Effusion, mm, mean (SD) Right knee medial |
0.28 (1.2) |
0 |
0.01 |
|
Right knee lateral |
2.0 (3.0) |
0.78 (1.7) |
0.03 |
|
Left knee medial |
0.34 (1.2) |
0 |
0.01 |
|
Left knee lateral |
2.0 (3.0) |
0.41 (1.6) |
< 0.001 |
|
EDSS: Expanded Disability Status Scale; WOMAC: Western Ontario and McMaster Universities Arthritis Index; VAS: visual analogue pain scale; N/A: not applicable; SD: standard deviation; BMI: body mass index. |
|||
WOMAC pain, stiffness and function scores and VAS scores were significantly worse in the MS group compared with those of the controls, (p < 0.001 for all). BBS scores were lower in the MS group (p < 0.001).
The femoral cartilages of patients with MS were more degenerated compared with healthy controls (p < 0.001). The intra-observer reliability for cartilage grading revealed substantial agreement (intraclass correlation coefficient = 0.87). Moreover, patients with MS had more effusion (in both compartments of bilateral knees) than did controls (p < 0.001) (Table I).
In the MS group; no relationship could be detected between cartilage degeneration grades, amount of effusion, and VAS scores, BBS, WOMAC and EDSS scores (Table II).
|
Table II. Spearman’s correlation coefficients for the relationships between the parameters in patients with multiple sclerosis (MS) |
||||||||||||||
|
|
Effusion Left Lateral |
Effusion Left Medial |
Effusion Right Lateral |
Effusion Right Medial |
Cartilage grade – Left |
Cartilage grade – Right |
VAS Left |
VAS Right |
WOMAC Function |
WOMAC Stiffness |
WOMAC Pain |
BBS |
EDSS |
Disease duration |
|
Body mass index |
0.003 |
0.143 |
0.113 |
0.080 |
–0.038 |
0.144 |
0.267 |
0.220 |
0.166 |
0.138 |
0.241 |
–0.043 |
0.040 |
0.058 |
|
Disease duration |
0.148 |
–0.026 |
0.098 |
–0.043 |
0.120 |
–0.003 |
0.086 |
0.119 |
0.449 |
0.263 |
0.244 |
–0.520 |
0.506 |
|
|
Expanded Disability Status Scale (EDSS) |
0.058 |
–0.060 |
–0.055 |
–0.115 |
–0.133 |
–0.127 |
0.072 |
0.080 |
0.549 |
0.313 |
0.134 |
–0.687 |
|
|
|
Berg Balance Scale (BBS) |
–0.283 |
–0.050 |
–0.164 |
0.069 |
0.103 |
0.053 |
–0.167 |
–0.162 |
–0.646 |
–0.304 |
–0.348 |
|
|
|
|
WOMAC Pain |
0.126 |
0.109 |
0.214 |
0.030 |
0.229 |
0.145 |
0.612 |
0.654 |
0.592 |
0.589 |
|
|
|
|
|
WOMAC Stiffness |
0.077 |
0.058 |
–0.028 |
–0.032 |
0.032 |
0.015 |
0.459 |
0.463 |
0.573 |
|
|
|
|
|
|
WOMAC Function |
0.141 |
0.138 |
0.152 |
0.040 |
0.004 |
0.005 |
0.410 |
0.431 |
|
|
|
|
|
|
|
VAS Right |
0.146 |
0.035 |
0.175 |
0.179 |
0.129 |
–0.050 |
0.788 |
|
|
|
|
|
|
|
|
VAS Left |
0.087 |
0.057 |
0.032 |
0.060 |
0.176 |
0.134 |
|
|
|
|
|
|
|
|
|
Cartilage grade – Right |
–0.036 |
0.051 |
0.025 |
–0.209 |
0.604 |
|
|
|
|
|
|
|
|
|
|
Cartilage grade – Left |
–0.130 |
–0.119 |
0.013 |
–0.215 |
|
|
|
|
|
|
|
|
|
|
|
Effusion Right Medial |
0.350 |
0.430 |
0.328 |
|
|
|
|
|
|
|
|
|
|
|
|
Effusion Right Lateral |
0.659 |
0.148 |
|
|
|
|
|
|
|
|
|
|
|
|
|
Effusion Left Medial |
0.264 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The bold values indicate the ones with statistical significance (p < 0.05). WOMAC: Western Ontario and McMaster Universities Arthritis Index; VAS: visual analogue pain scale. |
||||||||||||||
DISCUSSION
The aim of this study was to evaluate knee joint degeneration and its association with disease-related parameters in patients with MS. The major findings were as follows: (i) femoral cartilage of patients with MS was more degenerated than those of the healthy controls; (ii) however, femoral cartilage degeneration was not associated with disease-related parameters. To our knowledge, this is the first study to evaluate the condition of femoral cartilage in patients with MS.
Multiple sclerosis predominantly affects the motor and sensory structures of the lower limbs. Clinical manifestations of the disease depend on the total impact of the lesions throughout the CNS (1, 2, 4). It can lead to a wide range of motor, sensory and visual symptoms. Most patients have a range of mobility problems and biomechanical changes in the lower limbs, resulting in a great impact on quality of life (1–3, 8). According to the results of this study, patients with poorer balance also had poorer WOMAC pain, stiffness and function scores. In particular, the WOMAC physical function subscale would be important to investigate the association of knee degeneration with physical function.
Musculoskeletal US allows qualitative and quantitative assessment of the articular cartilage in different joints and plays an important role in screening and follow-up. US was used as the imaging method in our study, since it has the advantages of being a rapid, dynamic and cost-effective method (5, 6, 14). It was reported previously that increased joint effusion (detected with US) is associated with worse pain or poorer function in symptomatic knee osteoarthritis (13). Moreover, Yang et al.’s study, comparing the knees of hemiplegic and healthy subjects, found that suprapatellar effusion was more prevalent bilaterally in hemiplegic knees than in those of the otherwise healthy subjects (7). The proposed mechanism for patients with stroke included a biomechanical factor (e.g. incorrect knee joint posture or movement following repeated minor trauma of the paralysed knee), a compensatory behavioural factor (e.g. overuse of the non-paralysed knee), or a combination of both. Likewise, in the current study, suprapatellar effusion was more common in patients with MS. Because limb control is poor and sensory protection is impaired, patients with MS may be more prone to micro-traumas resulting in effusion and cartilage degeneration, and they may even not realize the traumas. However, the amount of effusion and the severity of femoral cartilage degeneration were not correlated in patients with MS.
MS has a progressive course, and higher EDSS scores reveal a more severe clinical involvement of MS (8). It can be suggested that cartilage degeneration would progress with an increase in the EDSS score. However, in the current study, no significant correlation was observed between femoral cartilage degeneration and disease duration. This may be related to the fact that an increased EDSS does not necessarily mean a longer disease duration.
In their study Tunc et al. reported that the femoral cartilage was thinner on the hemiparetic side of stroke patients, and certain disease-related parameters (disease duration and ambulatory status) of the patients correlated with femoral cartilage thickness (5). Kara et al. (6), found that femoral cartilage thickness was changed (significantly thicker bilaterally in the intercondylar areas) in patients with spinal cord injury, and there was a negative correlation with disease duration and severity. Patients with stroke or spinal cord injury may experience an immobile period. In the literature, contradictory results on the effects of immobilization on articular cartilage thickness have been reported. Although some researchers have stated that cartilage thickness decreased, others have reported that it was either increased or remained unchanged (17–23). In our study, the femoral cartilage of patients with MS was more degenerated than those of controls. This finding suggests that immobilization does not affect the cartilage alone, but also that impaired balance and ambulation, muscle weakness, abnormal muscle tone, decreased sensation and fatigue may negatively affect the cartilage (1).
This study has some limitations. Firstly, the strength or spasticity of the muscles and sensory deficits in the lower limbs were not evaluated individually, as those may have affected the stability of the knee joint and resulted in cartilage degeneration. Secondly, assessment of the cartilage and effusion was done with US, not with magnetic resonance imaging (MRI). MRI would allow: (i) visualization of cartilage morphology at high resolution; (ii) assessment of the biochemical composition of cartilage; and (iii) evaluation of all the structures within the knee joint. Moreover, quantitative measurement of cartilage morphology (cartilage thickness, volume, surface area and surface roughness) can also be performed by using MRI (24).
In conclusion, this study demonstrated that, patients with MS may have more rapid cartilage degeneration and increased effusion in their knees compared with healthy controls and that the intra-articular changes may be visualized with US. However, the degeneration of cartilage was not associated with disease-related parameters. Further studies, investigating the effect of spasticity, muscle strength and sensory deficits, are needed to clarify the impact of MS on knee degeneration.
REFERENCES
