Anders Björkman, MD, PhD1, Ulrika Wijk, MSc1, Christian Antfolk, PhD2, Isabella Björkman-Burtscher, PhD3,4 and Birgitta Rosén, PhD1
From the 1Department of Hand Surgery, Skåne University Hospital, Malmö, 2Department of Biomedical Engineering, 3Department of Radiology, Clinical Sciences and 4Lund University BioImaging Center, Lund University, Lund, Sweden
OBJECTIVE: Most amputees experience referred sensations, known as a phantom hand map, on the residual forearm, where touch on specific areas is perceived as touch on the amputated hand. The aim of this study was to evaluate the sensory qualities of the phantom hand map.
METHODS: In 10 traumatic forearm-amputees touch thresholds and discriminative touch of the phantom hand map were assessed and compared with corresponding areas on the contralateral forearm. The study assessed the localization of touch on the phantom hand map, and how distinct and similar to normal touch the referred feeling was.
RESULTS: Similar touch thresholds were seen in the phantom hand map and the control site. Tactile discrimination, requiring both detection of stimulus and interpretation, was significantly better in the phantom hand map.
CONCLUSION: This explorative study suggests that the phantom hand map and the superior tactile discrimination seen in the phantom hand map are based on adaptations within the brain. Further studies investigating the neural basis for the phantom hand map are needed.
Key words: sensory feedback; prosthesis; forearm amputees; referred sensations.
J Rehabil Med 2016; 48: 365–370
Correspondence address: Birgitta Rosén, Department of Hand Surgery, Skåne University Hospital, SE-205 02 Malmö, Sweden. E-mail: birgitta.rosen@med.lu.se
Accepted Jan 27, 2016; Epub ahead of print Mar 17, 2016
INTRODUCTION
Most forearm amputees experience some sort of phantom phenomena after amputation (1, 2). A general awareness of the missing forearm and hand, i.e. phantom limb awareness, is common. Furthermore, forearm amputees often experience different non-painful somatic sensations, such as tingling, itching or pressure in the lost hand, known as phantom limb sensations (1). Painful sensations are also frequently reported, both as pain in the residual forearm and pain in the missing hand, known as phantom limb pain (PLP) (1). Another reported phenomenon is “telescoping”, manifesting itself as “shrinkage” or shortening of the phantom limb, thus the amputee experiences that the lost hand has moved proximally towards or inside the residual forearm (1, 3). Furthermore, amputees may experience “referred sensations”, described as sensations from the phantom fingers, elicited by stimulation of specific skin areas on, for example, the residual limb, known as a phantom hand map (PHM) (4). Some amputees have a very fine PHM, with mapping of all fingers and the hand, while others have a more “simple” map containing 1 or 2 fingers, and others do not have a map at all. The anatomical and physiological substrate behind the PHM is not completely understood. During forearm amputation the major nerves in the forearm, the median, ulnar and radial nerves, are identified and cut under gentle tension, allowing them to retract into the remaining muscles (5). Thus, the PHM could be based on sprouting and neuroma formation from these severed nerves in the remaining forearm (6). Another possibility is that the PHM is based on the extensive cerebral reorganization known to occur after amputation (3, 6–8).
It has been demonstrated, using functional magnetic resonance imaging (fMRI), that stimulation of individual “fingers” in the PHM activates the same areas in the primary somatosensory cortex as stimulation of normal fingers in non-amputated healthy volunteers (8). However, stimulation of the skin just outside the PHM areas on the residual forearm did not result in activation of the cortical finger areas. This indicates a cross activation of areas representing the amputated fingers in the primary somatosensory cortex by afferents from the residual forearm (8). Incidence numbers of PHM in forearm amputees are not known, but a previous study has shown that 12 out of 18 amputees had a clear PHM (9). Earlier studies on sensory qualities in amputees have not focused on specific areas of the residual forearm, and results vary, showing either poorer or similar sensory qualities of skin areas on the residual forearm compared with the intact extremity (10, 11).
Our hypothesis is that phantom phenomena, such as the PHM, in the residual forearm in amputees are based on local nerve regeneration, cerebral reorganization, or a combination of both.
The aim of this study was to evaluate sensory qualities of PHMs in patients with unilateral forearm amputation.
METHODS
Participants
Ten adult forearm amputees experiencing a PHM participated in the study. They were identified in patient registers covering follow-up and technical support visits during the year prior to recruitment at 2 prosthesis centres in Sweden (Skåne University Hospital, Malmö and Red Cross Hospital, Stockholm). All participants had been amputated due to a trauma and they were all otherwise healthy. For demographic details and data about their phantom limb awareness/phantom limb sensations prior to the study, see Table I. Some participants used medications to relieve pain and they were allowed to continue this medication during the assessments. No patients had surgically implanted devices to relieve pain.
The local ethics review board (Dnr2012/778) approved the study and all participants gave their informed written consent. The study was conducted in accordance with the Declaration of Helsinki.
Pre-assessment procedure
Prior to the assessments all participants underwent an interview, and thereafter the “PHM” was mapped on the residual forearm.
Interview. The interview was conducted in order to document medical history, their phantom limb awareness/phantom limb sensations, and whether participants currently experienced pain at rest (Table I).
|
Table I. Study subjects, and data about their phantom limb awareness /phantom limb sensations prior to the study. Phantom limb awareness/phantom limb sensation characteristics; ameasured with visual analogue scales (0–10); bexperience of pain varies depending on time of day and/or medication |
|||||||||
|
Subject number |
Age, years |
Length (cm) of residual forearm (elbow to tip) |
Time (years) since amputation |
Dominant side amputated? |
Phantom limb sensation |
Telescoping |
Phantom limb pain at resta |
Prosthesis type |
Prosthesis usage |
|
1 |
40 |
12 |
28 |
Yes |
Yes |
Yes |
0 |
Cosmetic |
Outside home |
|
2 |
42 |
12 |
16 |
No |
Yes |
Yes |
5 |
Cosmetic |
All day |
|
3 |
49 |
18 |
26 |
No |
Yes |
No |
3 |
Cosmetic |
Outside home |
|
4 |
40 |
9 |
16 |
No |
Yes |
No |
2–7b |
Myoelectric |
All day |
|
5 |
36 |
16 |
6 |
Yes |
Yes |
Yes |
4–6b |
Myoelectric |
Half day |
|
6 |
56 |
26 |
1 |
Yes |
Yes |
Yes |
0 |
Cosmetic |
Sporadic |
|
7 |
23 |
22 |
1 |
Yes |
Yes |
Yes |
2–7b |
Myoelectric |
All day |
|
8 |
40 |
14 |
11 |
Yes |
Yes |
No |
3 |
Myoelectric |
All day |
|
9 |
44 |
10 |
28 |
No |
Yes |
No |
0 |
Myoelectric |
All day |
|
10 |
37 |
14 |
28 |
No |
Yes |
Yes |
1 |
None |
None |
Mapping of the phantom hand on the residual forearm. For each participant we mapped the PHM on the residual forearm. This procedure included application of systematic touches to the distal part of the stump to determine the spots that, when touched, gave rise to referred sensations in specific parts of the phantom hand and phantom digits. To facilitate the mapping, participants were shown a drawn hand template, which was divided into numbered sections that corresponded to each of the individual fingers as well as the volar and dorsal central parts of the hand. Initially the residual forearm was touched by the examiner’s index finger and when the first area was identified the participants were asked to touch the residual forearm themselves. The examiner double-checked that all areas of the distal parts of the stump were touched and the identified spots were touched repeatedly, and verified by the participants. The identified areas on the residual forearm were then numbered (Fig. 1).
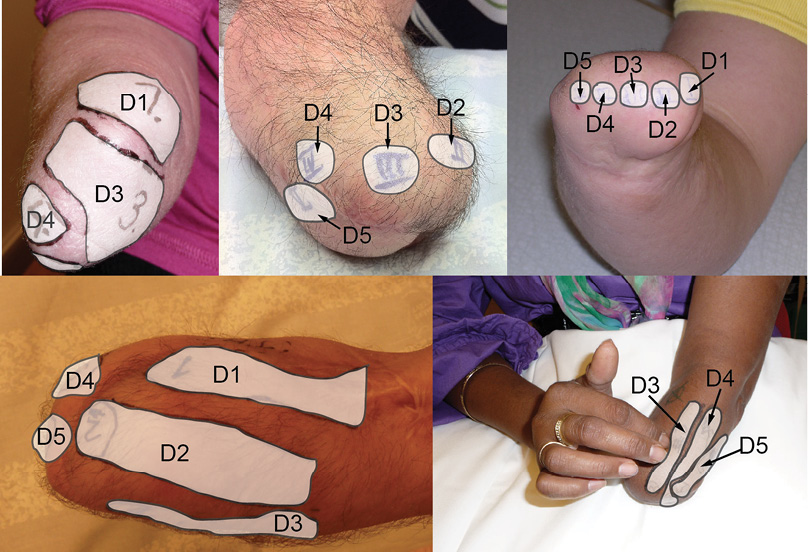
In addition, the participants were asked to point out the area with the strongest referred sensations.
All PHMs were documented with photographs of the residual forearm in the neutral position between pronation and supination and with the elbow at 90° flexion. The mapping procedure took 15–30 min.
As a subjective measure for how distinct the referred touch was, the participants were asked to rate how distinct/strong they experienced touch in different PHM areas, using a visual analogue scale (0–10), with the extremes of the scale defined as “no experience of touch” and “very distinct experience of touch”. Furthermore, as a subjective measure for the quality of the referred touch, the participants were asked how the touch was experienced in the PHM areas compared with normal touch of the specific area of the contralateral intact hand.
Again, a visual analogue scale was used (0–10), with the extremes of the scale defined as “not at all” and “fully comparable to normal touch experience”. The exact wording of the questions is given in Table II.
Assessment of sensory qualities
Performed assessments target the evaluation of integrity of peripheral nervous components as well as discriminative touch and localization of touch, which are more complex functions requiring both the detection and interpretation of a stimulus. Three different tests were carried out by an occupational therapist experienced in sensory testing to assess the sensory qualities and properties of the PHM. In each of the tests the participant was seated comfortably with their residual forearm on a table in a resting position and with their vision occluded with an eye-mask.
Touch thresholds. To examine the integrity of peripheral nervous components touch thresholds were investigated using a full set of Semmes Weinstein monofilaments (SWM) (North Coast Medical, Gilroy, CA, USA), which is a standardized test instrument (12). The monofilaments are calibrated to provide a specified force, ranging from 8 mg to 300 g (12). The test was performed on the PHM area with the strongest phantom feeling and on the corresponding area on the intact forearm. The assessment started with SWM #4.31 (equivalent to a pressure of 2 g, representing some protective sensibility) and thereafter in an ascending or descending order depending on the answer to the filament first tested. Each filament was applied 3 times according to standard procedure (12, 13).
Discriminative touch. The 2-point discrimination test (2PD) was used to assess discriminative touch. The test was performed on 1 of the PHM areas that corresponded to a finger pulp, and on the corresponding area on the contralateral, intact forearm. The test was performed strictly according to the “Moberg method”, as described below (14). Participant and examiner were seated comfortably, with the participant’s residual forearm and examiner’s hand stabilized against a firm support. All disturbing movements were avoided. Test application force was approximately 10 g, which, according to Moberg, corresponds to the force producing the very first small blanching around the prongs, and the test instrument was applied to the skin perpendicular to the skin. In his original articles Moberg suggests using an opened paper clip as test instrument, with a blunt wire with a diameter of 0.8–0.9 mm. Here the expected distances were several cm, and the test instrument used was a digital calliper modified with 2 blunt prongs of 1 mm diameter. The distance between the prongs could be set with high accuracy.
The response alternatives of the participants were predefined as “1 point”, “2 points” or “I can’t tell”. Ten touches, with an equal number of 1 or 2 prongs, were performed in random order, and 7 correct answers out of 10 were accepted as validation for a correct answer and to proceed to a smaller distance between the prongs. The application of touches was performed in a descending or ascending order of distance between the prongs, starting with 15 mm. The distance was decreased or increased by 5 mm each time it was changed. During repeated testing a learning effect is always a risk, and to control for that testing was started on the control site (on the intact forearm) in every other test person. The participant was familiarized with the test on the uninjured hand prior to the test.
Localization of touch. The participant’s ability to localize touch between different areas of the PHM was assessed (15). Areas of the PHM were randomly touched with a supra-threshold monofilament (SWM #6.65, equivalent to a pressure of 300 g). This was repeated 6 times for each skin area, resulting in a total of 30 stimulations for participants having a map with 5 phantom fingers.
Magnetic resonance imaging of the residual forearm
Magnetic resonance imaging (MRI) was performed in 2 participants (#1 and #9) on a 1.5T MRI scanner (Philips Achieva, Philips Healthcare, The Netherlands) to evaluate the occurrence of large neuromas in relation to the PHM. MRI data were acquired with transverse and oblique sagittal T1 turbo spin echo (TSE), transverse T2 TSE and transverse short T1 inversion recovery (STIR) sequences with spatial resolution of 4 × 0.4 × 0.4 mm (T1 TSE and T2 TSE) and 4 × 0.5 × 0.5 mm (STIR) and covering the residual forearm including the elbow. Magnetic resonance (MR) visible vitamin E markers were applied on the skin on the residual forearm corresponding to the different parts of the PHM. MR images were evaluated by an experienced musculoskeletal radiologist (> 20 years of experience) with focus on the visibility of the main (median, ulnar and radial) forearm nerves and large neuromas from these nerves, in relation to the PHM, with detection limitations defined by the spatial resolution of the scans.
Analysis
Data were analysed (SPSS®Version22, IBM®) with a paired comparison between touch threshold and discriminative touch on PHM areas and the contralateral forearm using the Wilcoxon signed-rank test.
RESULTS
Five men and 5 women participated in the study. The presence of a PHM was one of the inclusion criteria. Interestingly, some participants had not experienced or been aware of the details of the phantom hand until they were screened for this study. In addition to a PHM, 6 participants also experienced a telescoping phenomena, whereby the phantom hand had moved closer to or inside the residual forearm. Regarding phantom limb sensations, the experiences varied widely, from a clenched position always or sometimes, to an experience in one participant of being able to move the phantom hand freely. In 1 patient some movements in the phantom hand could be performed under concentration. Itching from an area distal to the residual forearm was also present in one participant. Details regarding individual PHM areas are given in Table II and illustrated in Fig. 1. To investigate the subjective experience of quantity as well as quality of the PHM at touch, 2 questions were asked: (i) “How distinct is the feeling of a finger at touch of the PHM?”; and (ii) “How close to normal touch of a finger (compared with the contralateral side) is the feeling at touch of the PHM?”. The strength of touch experience for PHM areas, as described by the participants, was mean 6.4 (range 3–10) on a visual analogue scale (0–10) (Table I). The quality of touch in PHM areas compared with normal touch experience was rated as mean 7.4 (range 3–10) on a visual analogue scale (0–10) (Table II).
|
Table II. Phantom hand map (PHM) characteristics (D = digit; ameasured with visual analogue scales (0–10); bexperience of pain varies depending on time of day and/or medication; cindividual PHM sensations delimited by commas, combined sensations (e.g. 2 fingers felt as 1) denoted by &; dPHM areas similar in size as corresponding contralateral body parts are marked, as in general PHM areas differ in size compared with corresponding body parts) |
|||||
|
Patient number |
Number of individual PHM areas |
Description of PHM areasc |
Pain at touch of PHM areasa |
“How distinct is the feeling of a finger at touch of the PHM”a |
“How close to normal touch of a finger is the feeling at touch of the PHM”a |
|
1 |
5 |
D1; D2; D3; D4; D5 |
0 |
8 |
7 |
|
2 |
6 |
D1 & D2; D3; D4; D5; radial side D1; ulnar side D5d |
6 |
7 |
8 |
|
3 |
3 |
D1; D3; D5 |
0 |
3 |
8 |
|
4 |
5 |
D1; D2; D3; D4; D5 |
1 |
D3: 3 D1; D2; D4; D5: 7 |
6 |
|
5 |
3 |
D1; D3 & D4; D5d |
4 |
D3 & D4: 4 D1; D5: 10 |
D3 & D4: 3 D1; D5: 10 |
|
6 |
4 |
Thenar & D1; D2; D3; Dorsum D1 & D2 & D4 & D5 |
0 |
10 |
7 |
|
7 |
5 |
D1; D2; D5; dorsum of metacarpus; hypothenar |
1 |
6 |
4 |
|
8 |
5 |
D1; D2; D3; D4; D5d |
2–7 |
5 |
5 |
|
9 |
6 |
D1; D2; D3; D4; D5; hypothenarb |
0 |
8 |
8 |
|
10 |
4 |
D1; D2; D4; D5 |
3 |
5 |
6 |
Results of assessment of touch thresholds, discriminative touch and capacity to localize touch in PHM areas are presented with raw data in Table III. Median touch threshold was within normal range for the PHM areas and the corresponding area on the contralateral forearm (8 mg; range 8–1,400 mg) (16). Six of the 10 participants experienced the same touch threshold in PHM areas as in corresponding areas on the contralateral forearm. Three participants had a higher (worse) threshold in the PHM areas, whereas one participant had a lower threshold, indicating a better sense of touch in the PHM compared with corresponding areas on the contralateral forearm. All but one patients had a lower (better) discriminative touch in the PHM area compared with the contralateral intact arm, and discriminative touch was significantly better (p = 0.008) in PHM areas compared with corresponding sites on the contralateral forearm.
Median discriminative touch, expressed as 2PD in PHM areas, was 25 mm (interquartile range (IQR) 19–26 mm), representing values better than expected normal values (16), and better than the median of 45 mm (IQR 26–50 mm) 2PD on corresponding areas on the contralateral forearm.
|
Table III. Results from assessment of touch thresholds, discriminative touch (both measured in the phantom hand map (PHM) area with the most intense phantom sensation) and capacity to localize touch. Normal touch threshold forearm: 0.010–0.18; normal touch threshold index finger: 0.02–0.13 [15]; normal discriminative touch forearm: 40 mm [15]; normal discriminative touch index finger: ≤ 5 mm. SWM Semmes Weinstein monofilament; 2PD: 2-point discrimination. Capacity to localize touch in PHM was tested in areas representing digits |
||||||||
|
Subject number |
Touch threshold (SWM), g |
|
Discriminative touch (2PD), mm |
|
Capacity to localize touch in PHM |
|||
|
PHM area |
Corresponding contralateral area |
|
PHM area |
Corresponding contralateral area |
|
Correct answers, % |
Sites, n |
|
|
1 |
0.008 |
0.008 |
|
25 |
50 |
|
97 |
5 |
|
2 |
1.4 |
0.6 |
|
25 |
50 |
|
71 |
4 |
|
3 |
0.16 |
0.008 |
|
25 |
45 |
|
100 |
3 |
|
4 |
0.008 |
0.008 |
|
25 |
45 |
|
27 |
5 |
|
5 |
0.008 |
0.008 |
|
40 |
60 |
|
100 |
3 |
|
6 |
0.008 |
0.008 |
|
15 |
30 |
|
83 |
3 |
|
7 |
0.04 |
0.008 |
|
20 |
40 |
|
94 |
3 |
|
8 |
0.008 |
0.008 |
|
30 |
50 |
|
90 |
5 |
|
9 |
0.008 |
0.008 |
|
25 |
15 |
|
100 |
5 |
|
10 |
0.008 |
0.02 |
|
10 |
12 |
|
80 |
4 |
|
Median |
0.008 |
0.008 |
|
25 |
45 |
|
95 |
4 |
No statistically significant difference between touch threshold in the PHM areas and the contralateral side (p = 0.14) was found. When assessing the capacity to localize touch between different fingers of the PHM the median correct response rate was 95%.
All but one subject had a lower (better) discriminative touch in the PHM area compared with the contralateral intact arm. When assessing the capacity to localize touch between different areas of the hand of the PHM, the median correct response rate was 95%.
On MRI in 2 patients the median, ulnar and radial nerves were identified at the level of the elbow and could be followed to the level of nerve transection. No neuromas were identified along these nerve structures. Individual cutaneous nerve branches, sprouts or neuromas located close to the PHM areas were not identified within the detection limits given by the spatial resolution of the MR scans.
DISCUSSION
The capacity to detect a cutaneous stimulus, a function based on cutaneous receptor density was the same in PHM areas as on corresponding areas on the contralateral, intact forearm. However, tactile discrimination, a function that requires both the detection and interpretation of a stimulus, i.e. a more complex task, was significantly better in PHM areas compared with corresponding areas on the intact forearm. MRI of the residual forearm, based on the special resolution given, neither detected nor excluded nerve structures that could form an anatomical basis for the PHM. This explorative study suggests that the PHM and the superior tactile discrimination in the PHM may be based on adaptation in brain functions.
Normal tactile discrimination, measured with 2PD, in the forearm, taking sex and handedness into consideration, is known to be approximately 40 mm (16). Our study shows a significant difference between the intact forearms, with median 2PD of 45 mm compared with 25 mm in PHM areas. The difference between right- and left-handedness and sex regarding touch thresholds and discriminative touch with slightly better results in left-handed persons and in women shown by Weinstein (16) could not be analysed due to the composition of the present study population. Age is also a factor of importance, and 2PD is generally better in the young (17, 18); however, no such pattern could be seen in this study.
Individual PHM differs between amputees, some having a very detailed map with all fingers (9, 19) and part of the palm represented, and others showing a simpler PHM or no map at all. This could be attributed to local nerve regeneration in the residual forearm, individual differences in the cerebral reorganization occurring after amputation or a combination of both. Sprouts from the transected nerves in the residual forearm might reinnervate specific areas of the skin more densely than others and thus form the basis for the PHM. Individual variations in this sprouting could explain the differences in PHM appearance between amputees. However, if the PHM is based on sprouting, resulting in skin areas with denser innervation, the perception of touch should be better, with lower touch thresholds, in PHM areas compared with adjacent skin areas. All participants in the present study had normal perception of touch, indicating normal innervation and function in the peripheral receptors inside as well as outside the PHM areas. Furthermore, MRI was performed in 2 out of the 10 amputees and, in these, no connection between the major forearm nerves and the PHM areas could be visualized; however, this does not exclude the existence of such connections below MR spatial resolution capacities.
Studies in animals have shown enlargement of residual limb representation after amputation (20), indicating that the occurrence of PHM might be the normal cortical reaction to loss of a limb. However, even if the PHM is a normal cerebral post-amputation phenomenon it requires that sensory information is sent from receptors in the skin to the brain. In patients with atrophic skin and/or extensive subcutaneous scarring in the residual forearm, the innervation of the skin is poor or damaged, which could be detected as poor touch threshold, and thus afferent brain input from these areas would be decreased. This could be a mechanism explaining poor or absent PHM in some amputees. Future larger studies correlating perception of touch in the residual forearm with the presence of a PHM, could provide an answer to this, if taking into account the time since amputation and the physiological condition of the residual forearm.
Hunter et al. (1) found, studying a group of 11 forearm amputees, that use of a functional prosthesis correlated with stable vividness of phantom limb awareness, whereas amputees who used a cosmetic prosthesis had less vivid phantom awareness. Furthermore, frequent use of a functional prosthesis, especially a myoelectric prosthesis, has been shown to result in less cortical reorganization and less phantom limb pain (11, 21), although it has also been reported that perception of touch in the residual forearm is better in amputees not using a prosthesis than in those who used a prosthesis (21). Based on our study population we could not establish any valid links between discriminative capacity on the PHM and prosthesis type or usage.
The present study has several limitations. The absolute number of participating patients is low. Traumatic amputation of an arm is a relatively uncommon injury today in Europe. Nevertheless, it is an injury that causes great impact on function, on activities of daily life and on quality of life for the individual. In a previous study (9) we showed that 70% of forearm amputees experience a PHM. Given that forearm amputees are rare, the patients with PHM are even rarer. Another limitation is the lack of a control group. We used a corresponding skin area on the contralateral, normal, arm as control to the PHM areas on the residual forearm. If a group of amputees with similar anatomical appearance of the residual forearm, but without a PHM, were available, that would, of course, have been the optimal control. However, the number of available forearm amputees is, as mentioned, limited. To use a control point on the residual forearm outside the PHM was an alternative. However, the residual forearms are anatomically very different, both in terms of length, remaining soft-tissue structures and skin quality. Fig. 1 shows the variety of shapes of the residual forearms as well as differences in the configuration of PHM between patients. In some patients the PHM is just a few small spots and, in others, larger areas correspond to specific parts of the hand. In patients with a short residual forearm with large PHM areas there is no space for control sites on the residual forearm. Skin areas at elbow level or more proximal have normal touch thresholds and discriminative touch that are different from those found in the forearm (16). Therefore we considered the contralateral, uninjured forearm with intact skin and identical internal (personal) and external (environmental) prerequisites, to be the best control site.
Seven of the 10 participants reported phantom hand pain of various intensities during the day at rest. This could be a potentially confounding factor when assessing sensory functions of the stump. However, none of the participants reported discomfort during the assessments.
It was of interest to examine the integrity of peripheral nervous components, as well as discriminative touch and localization of touch, which are more complex functions, that require both the detection and interpretation of a stimulus. To assess the integrity of peripheral nervous components the Semmes-Weinstein monofilaments is one of few assessment instruments for the upper extremity that fulfil criteria for standardization (22). However, the reliability of clinical test instruments for sensory testing is debated. That, in combination with the well-known methodological problems in 2PD testing, calls for a very thorough test procedure (17, 22, 23).
The superior ability to detect and localize sensory stimuli in the PHM, the subjective experience of the “fingers” in the PHM, together with the fact that cutaneous stimulation of the PHM results in activation in the finger areas in the primary somatosensory cortex (8), suggest that the PHM is an optimal target for transferring sensory information from a hand prosthesis to the prosthesis user.
ACKNOWLEDGEMENTS
This study was supported by the Stockholm Brain Institute, the Swedish Research Council, the Crawford Foundation, the Promobilia Foundation and Skåne County Council Research and Development Foundation. The authors thank Associate Professor Mats Geijer for evaluation of MR images.
REFERENCES
