Tobias Boeselt, PhD1, Christoph Nell, PhD1, Katharina Kehr, MD1, Angélique Holland, MD1, Marc Dresel1, Timm Greulich, MD1, Bjoern Tackenberg, MD2, Klaus Kenn, MD5, Johannes Boeder3, Benjamin Klapdor, MD1, Andreas Kirschbaum, MD4, Claus Vogelmeier, MD1, Peter Alter, MD1 and Andreas Rembert Koczulla, MD1
From the 1Department of Internal Medicine, Division of Pulmonary Diseases, 2Department of Neurology, 3Department of Physical Therapy, 4Department of Thoracic Surgery, Philipps University of Marburg, Marburg and 5Schoen Klinik Berchtesgadener Land, Schönau am Königssee, Germany
BACKGROUND: Admission to the intensive care unit is associated with sustained loss of muscle mass, reduced quality of life and increased mortality. Early rehabilitation measures may counteract this process. New approaches to rehabilitation while the patient remains in bed are whole-body vibration alone and whole-body vibration with a dumbbell. The aims of this study are to determine the safety of whole-body vibration for patients admitted to the intensive care unit, and to compare the effects of these techniques in intensive care unit patients and healthy subjects.
METHODS: Twelve intensive care unit patients and 12 healthy subjects using whole-body vibration for the first time were examined while lying in bed. First both groups performed whole body vibration over 3 min. In a second step whole body vibration with dumbbell was performed. In order to determine the safety of the training intensity, heart rate, oxygen saturation and blood pressure were measured. The study was approved by the Marburg ethics committee.
RESULTS: There were minor reversible and transient increases in diastolic blood pressure (p = 0.005) and heart rate (p = 0.001) in the control group with whole-body vibration with a dumbbell. In intensive care patients receiving whole-body vibration alone, there were increases in diastolic blood pressure (p = 0.011) and heart rate (p < 0.001).
CONCLUSION: This study demonstrates the feasibility of using whole-body vibration and whole-body vibration with a dumbbell for intensive care unit in-bed patients. No clinically significant safety problems were found. Whole-body vibration and whole-body vibration with a dumbbell might therefore be alternative methods for use in early in-bed rehabilitation, not only for hospitalized patients.
Key words: early rehabilitation; exercise; whole-body vibration; intensive care unit; muscle; heart rate; blood pressure; oxygen saturation.
J Rehabil Med 2016; 00: 00–00
Correspondence address: Tobias Boeselt, Department of Internal Medicine, Division of Pulmonary Diseases, Philipps University of Marburg, Baldingerstr., DE-35033 Marburg/Lahn, Germany. E-mail: Tobias.boeselt@staff.uni-marburg.de
Accepted Nov 10, 2015; Epub ahead of print Jan 21, 2016
INTRODUCTION
Patients in intensive care largely require resting conditions and thus are immobilized. Current physiotherapeutic approaches are restricted to passive mobilization. However, deprivation of appropriate physical activity is associated with insufficient muscle stimulation, leading to weakness and muscular atrophy. In particular, the lower extremities are prone rapid loss of a marked proportion of muscle mass (1).
Critical illnesses can lead to a mean weight loss of 17% within the first 10 days of stay in the intensive care unit (ICU) (2) and up to 40% within 4–6 weeks (3). Combined occurrence of immobilization and critical illness is associated with increased loss of muscle mass compared with resting conditions alone (4). Adverse consequences potentially persist over several years. In addition to physical limitations, anxiety disorders, depression, post-traumatic stress disorders (5–8), decreased quality of life (9, 10) and increased long-term mortality (11) may occur.
Early mobilization has a variety of beneficial effects, including improvement in muscle strength, mobility, quality of life (12–14), reduced risk of venous thromboembolism, decubital ulcers, pneumonia (15, 16) and mortality (17). The duration of mechanical ventilation and the total length of hospital stay are reduced. In addition to individual health-related benefits of early mobilization, there are also socioeconomic advantages.
While passive mobilization is feasible in senseless, analgose-dated and mechanically ventilated patients, more effective active rehabilitation usually requires a conscious patient who is willing to participate. In-bed treadmill ergometers, bed-side leg presses, neuromuscular stimulation methods, and assisted walking have been established for this purpose.
The safety and feasibility of early ICU rehabilitation has been proven in several studies (18). However, the strict prerequisites mentioned above markedly prevent the widespread use of classical active rehabilitation in ICU patients.
Whole-body vibration (WBV) is a platform that involves cyclic sinus-wave oscillations of various frequencies and amplitudes transmitted to the body. The frequencies range from 5 to 30 Hz. While low frequencies primarily address relaxation of previously trained muscles and proprioception training, middle frequencies (12–20 Hz) affect the contraction and relaxation cycles and thus train reflex muscle function. At high frequencies (20–30 Hz) the relaxation time will be shorter than the contraction time, increasing the effect of muscle performance, with an expected training effect.
Usually, a stationary platform is used for vertically standing subjects. The method is frequently used for workout purposes to improve muscle strength and endurance. In patients, beneficial effects have been described in several diseases, e.g. osteoporosis (19), anti-synthetase syndrome (20), multiple sclerosis (21), diabetic neuropathy (14), chronic arthritis (22) and type 2 diabetes mellitus (23). WBV has also been used in both, stable (23, 24) and exacerbated chronic obstructive pulmonary disease (COPD) (25), leading to improved 6-min walk distance and quality of life (24–26). Other studies have shown increased muscle force (27, 28). A prospective study on 20 healthy males examined the effects of 56 days of bed rest. Ten participants were randomized to resistive vibration exercise twice daily and others served as controls. The intervention group showed increased plantar flexion force, while a 17.1% decrease was observed in controls (29).
Taking into account the advantages of WBV therapy, the aim of the present study was to examine the feasibility and safety of its use in ICU patients.
MATERIAL AND METHODS
The study was performed in a medical ICU with predominantly pulmonary and cardiac patients. Twelve hospitalized ICU patients and 12 healthy subjects were included in the study. Routinely recorded patient data were analysed retrospectively. The mean duration of ICU stay was 15 (standard deviation (SD) 6) days (Table I). Written informed consent for the application of WBV was provided by the patient, relatives or legal guardian. Blood pressure, heart rate (HR) and oxygen saturation (SaO2) were measured. The study aimed to determine whether WBV training is feasible in ICU patients with a RASS of –1 and 0.
|
Table I. Cause and duration of hospitalization of patients (mean 15 days, standard deviation 6 days) |
||
|
Patient |
Leading cause of hospitalization |
Duration of hospital stay (days), mean |
|
1 |
Coronary heart disease due to myocardial infarction |
14 |
|
2 |
Chronic obstructive pulmonary disease (GOLD IV/D) |
31 |
|
3 |
Heart failure due to myocarditis |
16 |
|
4 |
Intracranial haemorrhage |
18 |
|
5 |
Severe pancreatitis |
12 |
|
6 |
Cardiac decompensation due to myocardial infarction |
11 |
|
7 |
Pulmonary thromboembolism |
14 |
|
8 |
Chronic obstructive pulmonary disease (GOLD IV/D) |
12 |
|
9 |
Respiratory insufficiency global due to restrictive ventilatory disorder |
17 |
|
10 |
Cardiac decompensation due to myocardial infarction |
8 |
|
11 |
Chronic obstructive pulmonary disease (GOLD IV/D) |
10 |
|
12 |
Acute renal failure |
19 |
Training method: whole-body vibration
WBV was performed with the patient lying in bed in the supine position. During the study, a training device (Fig. 1b), type GalileoTM (Novotec Medical, Pforzheim, Germany), was used. The device was attached to the foot of the bed. In order to transmit a sufficient load to the patient’s lower limbs, the bed was inclined by 25°.
The training was performed in 2 stages. First, the vibrating foot plate was used alone. Training was performed shoeless for 3 min, with a frequency of 24 Hz and a mean intensity of foot position 11.

1The foot position is divided into 3 positions (1–3). The foot position 1 describes a 30 cm bride foot stand – the wider the food distance, the more intense the training effort – meaning 3 has the highest training effort.
During training, the knees were slightly bent. As described previously, this results in a decrease in transmission of vibrations to the cranium through increased tension (24).
In a second step, after 1 min of rest, whole-body vibration with a dumbbell (WBVD) was added, with a vibrating dumbbell mounted on the bed trapeze (Fig. 1a).
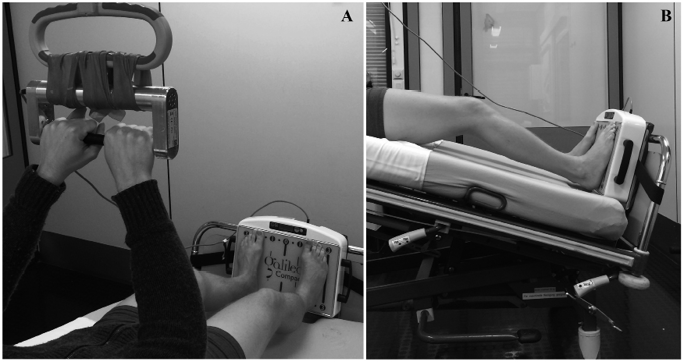
Recorded parameters
Arterial blood pressure, HR (using a 12-lead standard electrocardiogram (ECG)) and peripheral oxygen saturation (SaO2) were assessed. In the patient group, systolic and diastolic blood pressure were determined via the existing invasive blood pressure measurement using a Statham pressure transducer. In healthy controls, blood pressure was determined non-invasively using standardized arm cuffs (Riva Rocci).
Vital signs were recorded 1 min prior to training, every minute during exercise and 1 min after training.
In one healthy subject, electromyography (EMG EmblettaTM gold, San Diego, CA, USA) of the quadriceps femoris muscle (vastus lateralis) was performed in order to record electrical muscle activity during training (see Fig. 5). The measurement was preceded by a minute rest measurement. Following, the thigh was tense isometric for 3 min and charged following for another 3 min on the WBV.
The study was approved by the ethics committee of the University of Marburg (19/2015).
Statistical evaluation
Friedman’s test was used for comparison of several sequential measurements. Differences in measurements obtained from successive time-points were calculated and compared using Mann-Whitney U test. Values were expressed as mean ± standard deviation. All tests are 2-sided and statistical significance was assumed at p < 0.05.
RESULTS
Twelve ICU patients (5 males, 7 females), mean age 41.8 (SD 19.7) years and 12 healthy male volunteers, mean age 31.3 (SD 6.6) years, were examined. Details of the reasons for the patients’ admission to the ICU are shown in Table I.
Vital signs in controls
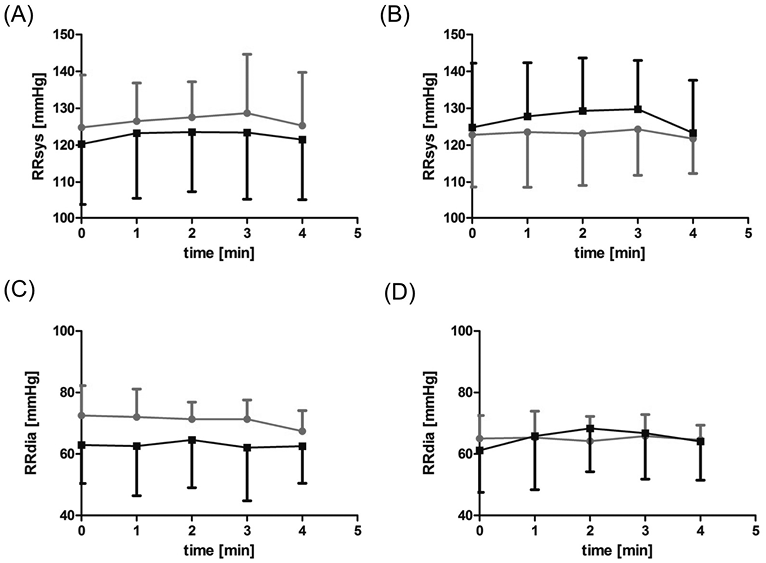
WBV and WBVD caused a mild and completely reversible increase in HR (p = 0.005, p = 0.001; Fig. 2a), while a mild transient increase in systolic blood pressure was observed only during WBVD (p < 0.001; Fig. 3b). Diastolic blood pressure showed a slight reverse trend (decrease) (Fig. 3d). No changes in SaO2 were observed during all phases of treatment.
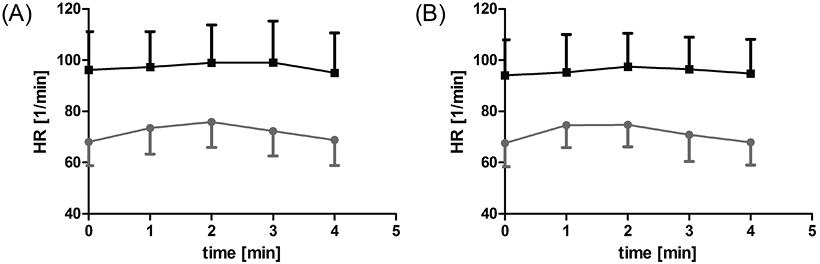
Vital signs in ICU patients
A slight increase in HR (p = 0.011) during WBV was observed in ICU patients (Fig. 2a). The blood pressure showed a slight temporary increase during WBVD (Fig. 2b). In contrast to the controls, there was a transient increase in diastolic blood pressure (p < 0.001; Fig. 3d). There were no changes in SaO2 in the patient group (Fig. 4).
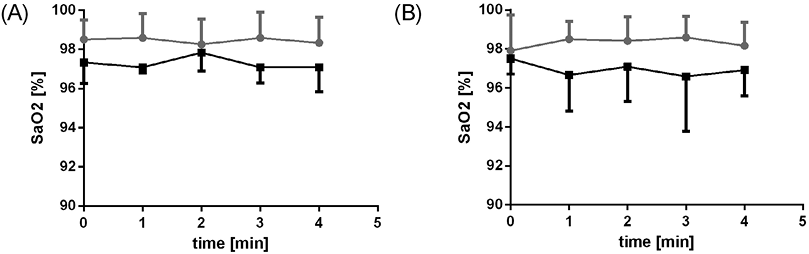
Group differences in vital signs
During WBV training in ICU patients, there was a significant increase in SaO2 in the second period (between the first and second minute) (p = 0.006). For WBVD, significant differences were found only for SaO2 and diastolic blood pressure in the first period (between the initial value and the first minute) (p = 0.003). The differences for these time periods are shown in Table II.
|
Table II. Differences in measurements between time-points for whole-body vibration (WBV) and whole-body vibration with a dumbbell (WBVD) training. Initially, measurements for the first minute (1) of training were compared with the resting condition (0). Subsequent training minutes were compared with previous ones |
||||
|
Characteristics |
Differences |
|||
|
∆1–0 Mean (SD) |
∆2–1 Mean (SD) |
∆3–2 Mean (SD) |
∆4–3 Mean (SD) |
|
|
WBV |
||||
|
Controls |
||||
|
HR (1/min) |
5.42 (5.7) |
2.42 (4.34) |
–3.58 (6.36) |
–3.5 (3.42) |
|
SaO2 (%) |
0.08 (0.67) |
–0.33 (0.65) |
0.33 (0.78) |
–0.25 (1.06) |
|
Sys (mmHg) |
1.67 (8.25) |
1.08 (5.74)** |
5.25 (15.06) |
–7.5 (17.96) |
|
Dia (mmHg) |
–0.5 (12.01) |
–0.67 (10.6) |
0.01 (5.17) |
–3.92 (6.02) |
|
Patients |
||||
|
HR (1/min) |
1.08 (4.25) |
1.67 (3.68) |
0.08 (4.12) |
–4 (4.31) |
|
SaO2 (%) |
–0.25 (0.97) |
0.75 (0.97)** |
–0.75 (1.14) |
0.01 (0.85) |
|
Sys (mmHg) |
2.92 (4.19) |
0.33 (3.31) |
–0.17 (3.21) |
–1.92 (3.55) |
|
Dia (mmHg) |
–0.33 (6.01) |
1.58 (1) |
–2.08 (7.83) |
0.42 (10.77) |
|
WBVD |
||||
|
Controls |
||||
|
HR (1/min) |
6.92 (3.15)*** |
0.17 (3.3) |
–3.92 (8.11) |
–2.92 (6.32) |
|
SaO2 (%) |
0.58 (1.08) |
–0.08 (0.67) |
0.17 (0.58) |
–0.42 (0.9) |
|
Sys (mmHg) |
0.75 (5.38) |
–0.42 (2.35) |
1.17 (6.04) |
–2.58 (9.54) |
|
Dia (mmHg) |
0.33 (4.25)* |
–1.17 (2.17) |
1.67 (5.85) |
–1.25 (7.17) |
|
Patients |
||||
|
HR (1/min) |
1.17 (4.28)*** |
2.25 (4.22) |
–1.08 (2.68) |
–1.67 (3.08) |
|
SaO2 (%) |
–0.83 (2.17) |
0.42 (0.67) |
–0.5 (1.45) |
0.33 (2.27) |
|
Sys (mmHg) |
3 (5.98) |
1.5 (3.68) |
0.42 (3.73) |
–6.42 (3.63) |
|
Dia (mmHg) |
4.67 (7.09)* |
2.5 (5.57) |
–1.58 (4.85) |
–2.67 (8.78) |
|
*p = 0.025; **p = 0.006; ***p = 0.003. SD: standard deviation; HR: heart rate; Sys: systolic blood pressure; Dia: diastolic blood pressure; SaO2: oxygen saturation. |
||||
Electromyography
Exemplary use of electromyography (EMG) in one healthy subject showed significantly increased electrical activity of the quadriceps femoris muscle (vastus lateralis) during training compared with resting conditions (Fig. 5).
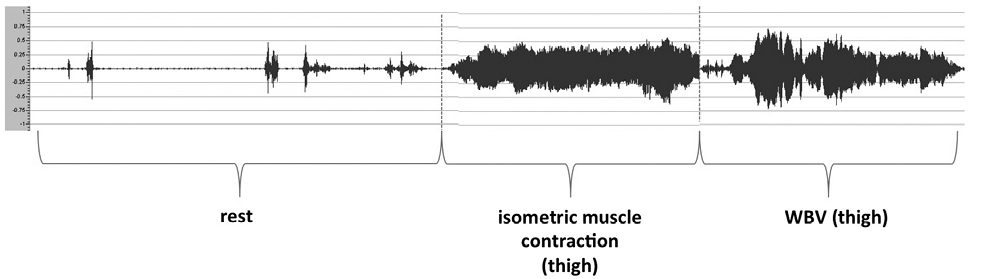
Fig. 5. Surface electromyography of the quadriceps (vastus lateralis) in 1 healthy control subject at rest, under isometric contraction, and under whole-body vibration (WBV).
DISCUSSION
This study examined the novel technique of using WBV and WBVD therapy in ICU patients. No evidence of any harm to patients through application of WBV or WBVD was found; the method appears to be safe.
Transient mild increases in HR during WBV and blood pressure during WBVD were observed; however, all potentially procedure-related effects were minor and completely reversible within minutes. No changes remained after completion of the intervention. The initial increase in HR was lower in patients compared with controls, which may be due to the influence of drugs that lower HR and blood pressure, such as β-blockers and angiotensin-converting-enzyme (ACE) inhibitors. For both of these drugs an influence on HR variability has been described (30).
The WBV plate was attached directly to the patient’s bed and induced vibrations of the bed frame, which were felt by the patients. However, none of the patients complained about the vibrations. To verify the training effects, EMG of the quadriceps femoris muscle (vastus lateralis) was performed in 1 healthy subject. Significantly increased electrical activity was recorded during training compared with at rest (Fig. 5). This suggests an effectiveness of the intervention with regard to the training effect. The training duration we used was too short to achieve a training effect. However, this study was not designed to evaluate such an effect, but to test the safety of the training.
WBVD was used for vibration exercise. This was well tolerated both by the patients and by the control group. However, compared with WBV, WBVD training requires a higher level of patient cooperation. It remains unclear whether a training effect can be achieved with this technique, and further studies are warranted. A study by McBride et al. found that vibration therapy during isometric contraction resulted in more efficient and effective recruitment of high threshold motor units (31).
Based on the maximum bed inclination, we used a 25° inclination, which was usually sufficient put the subject’s weight onto the WBV plate. The WBV device is constructed so as to reduce the frequency of WBV if the patient does not put enough weight on the plate.
The novel approach of using WBV and WBVD in ICU patients was favourably tolerated in the current study. Alternative strategies include electrical muscle stimulation; however, this is only moderately tolerated by the majority of patients (32).
Over a 16-day period, Meesen et al. used neuronal muscle stimulation in critically ill patients, stimulating 1 leg while the second leg was not trained (33). Significant changes were observed after 7 days. While the untrained leg showed reduced muscle size, the muscle mass in the trained leg increased. In this study, no change in vital signs occurred during or after exercise (33). Gerovasili et al. reported decelerated muscle loss concerning the diameter of the rectus femoris muscle in critically ill patients by neuronal muscle stimulation over 9 days (34). Gruther et al. showed long-term effects of neuromuscular training on the diameter of the quadriceps femoris muscle during more than 2 weeks (35). Taken together, there is evidence of a positive effect of electric muscle stimulation on muscle mass. Further research is required to determine whether WBV and WBVD have the potential for a similar effect.
Various studies have reported a positive effect of early rehabilitation in the ICU, resulting in increased muscle strength, mobility and quality of life (36), as well as a reduction in the duration of artificial ventilation (37), length of hospital stay, or time in the ICU (38). It is also known that exercise training can result in physiological left ventricular hypertrophy with beneficial effects in patients with COPD (39).
There is strong evidence for the positive effects and safety of early rehabilitation in the ICU setting; however, no clear definition or recommendations have yet been established (13). Future studies should describe the precise intervention and its validity.
This study has some limitations. The patients were older than controls and the controls were all male. In addition, the ICU patients were taking several medications and had a number of comorbidities, which may have influenced the results. However, these limitations do not affect the findings regarding feasibility and safety.
In conclusion, the present study examined feasibility and safety aspects of using WBV and WBVD therapy in ICU patients, and demonstrated no obvious harm to the patient. WBV and WBVD therapy should be considered for use during ICU treatment. Further prospective randomized trials are required to evaluate the potential beneficial effects of WBV and WBVD therapy on early rehabilitation in ICU patients.
ACKNOWLEDGEMENTS
This study was supported by the German Center for Lung Research (COPD II, 82DZL00502) and by a Research Grant of the University Medical Center Giessen and Marburg. The authors declare that they have no competing interests.
The authors thank NOVOtec for providing the prototype Galileo vibration platform with dumbbell.
REFERENCES
