Sung Ho Jang, MD
From the Department of Physical Medicine and Rehabilitation, College of Medicine, Yeungnam University, new primary melanomaDaemyungdong, Namku, Taegu, Republic of Korea
The corticospinal tract, a major neural tract in the human brain for motor function, is concerned mainly with movement of the distal extremities. Preservation or recovery of the corticospinal tract is essential for good recovery of impaired motor function in patients with brain injury. Therefore, thorough and precise knowledge of the corticospinal tract is necessary for successful brain rehabilitation. Many studies have reviewed the corticospinal tract; however, review articles from the rehabilitative viewpoint are lacking. Therefore, the aim of this paper was to review the corticospinal tract from the rehabilitative viewpoint with regard to classification, cerebral origin, collaterals and development.
Key words: corticospinal tract; brain injury; motor function; rehabilitation; pyramidal tract.
J Rehabil Med 2014: 46: 193–199
Correspondence address: Sung Ho Jang, Department of Physical Medicine and Rehabilitation, College of Medicine, Yeungnam University 317-1, Daemyungdong, Namku, Taegu, 705-717, Republic of Korea. E-mail: strokerehab@hanmail.net
Accepted Nov 11, 2013; Epub ahead of print Feb 13, 2014
Introduction
In the human brain, the descending motor pathways are classified as the corticospinal tract (CST or pyramidal tract (PT)) and the non-corticospinal tract (1–3). The CST, a major neural tract in the human brain for motor function, was acquired as the result of long-term evolution (1–7). It is concerned mainly with movement of the distal extremities, particularly fine motor activities of the hand (2–4, 8). In addition, preservation or recovery of the CST is essential for good recovery of impaired motor function in patients with brain injury (8–19). Thus, thorough knowledge of the CST is mandatory for successful brain rehabilitation.
For successful brain rehabilitation, understanding of the exact neurological manifestations related to neural injury, prediction of the recovery course and prognosis of each neurological manifestation, presumption of the possible recovery mechanisms and their clinical significance, and adoption of effective strategies to facilitate a recovery mechanism for achievement of the best outcome are important. For this purpose, in brain rehabilitation of motor function, up-to-date and precise information on the CST is necessary. Many studies have reviewed the CST; however, review articles from the viewpoint of brain rehabilitation are lacking (2, 3, 20, 21). Therefore, the aim of the current study was to review the CST from the rehabilitative viewpoint with regard to classification, cerebral origin, collaterals, and development. This review was limited to the topics described above because other topics relevant to brain rehabilitation, such as recovery mechanisms or prognosis prediction using transcranial magnetic stimulation (TMS) or diffusion tensor imaging (DTI) have been reviewed previously (17, 20, 21).
Classification of the corticospinal tract
There are 3 known separate CSTs: the crossed lateral CST, the uncrossed lateral CST, and the uncrossed anterior (ventral) CST (1–3, 22–25) (Fig. 1A). In addition, Nathan et al. (24) reported the presence of the crossed anterior CST. Although the functions of these CSTs are known to differ, the exact functional role of each CST has not been clearly elucidated. Previous studies on the evolution of the motor system have suggested that the CST is unique to mammals, and that its development is associated with acquisition of dexterous motor skills (4–7).
The largest CST is the crossed lateral CST, which occupies 75–90% of the CST fibres extending caudally to the dorsolateral fasciculus to the last sacral segment after crossing the medulla (1–3, 21, 24, 25). Many previous studies have demonstrated the relationship between the status of the CST and motor function, as follows: (i) distal muscles for the fingers and ankles are controlled mainly by the lateral CST, and proximal muscles are controlled by other neural tracts, such as the cortico-reticulospinal tract and the anterior CST; (ii) there is greater involvement of the lateral CST in motor function of the upper extremities than in the lower extremities; (iii) finger extensors most accurately reflect the function of the lateral CST; and (iv) the lateral CST is mandatory for hand function; in contrast, association of walking with the lateral CST is not as strong as that of hand function (2, 3, 5, 8, 26–30).
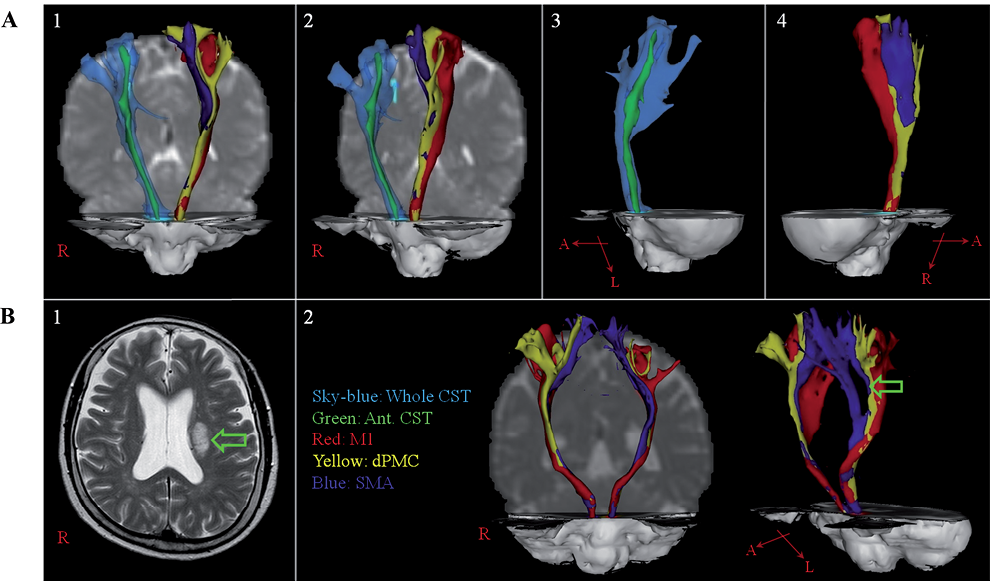
Fig. 1. Diffusion tensor tractographies for the corticospinal tract (CST) in: (A) a normal subject (45-year-old male); and (B) a stroke patient (72-year-old female). (A) The whole CST and the anterior CST were reconstructed in the right hemisphere. The CST fibres were reconstructed in the left hemisphere (red: CST fibres from the primary motor cortex [M1]; yellow: CST fibres from the dorsal premotor cortex [dPMC]; blue: CST fibres from the supplementary motor area [SMA]). (1) antero-posterior view; (2) oblique view; (3) right lateral view; (4) left lateral view. (B) (1) T2-weighted brain magnetic resonance image of the patient showed an infarct (arrow) in the left corona radiata. (2) The CST fibres according to the cerebral origin. The CST fibres from the dPMC in the left hemisphere reveal injury (arrow) by the infarct in the left corona radiata.
The anterior CST, which does not cross the medulla, occupies 5–15% of the entire CST (1, 3, 27, 28). It extends caudally only to the upper thoracic cord and rarely descends below the thoracic spinal cord (1, 3). A study using DTI reported that the fibre number of the anterior CST was 12.4% of the entire CST in the human brain, and the anterior CST has the characteristics of less directionality, compared with the entire CST (31). The function of the anterior CST has not been clearly elucidated. However, some studies have reported that the anterior CST primarily innervates the proximal muscles, such as the musculature of the neck, trunk, and proximal upper extremities (1, 3, 22). On the other hand, it is regarded as one of the potential descending motor pathways that might play a role in walking (2).
Some researchers have proposed the existence of the uncrossed lateral CST (1, 3, 32, 33). The size varies in different individuals: it may consist of only a few isolated fibres. It descends within the lateral funiculus and is located ventrally to the crossed lateral CST. Its function is unknown and it has been reported to reach the lower segments of the spinal cord (1, 3). Nyberg-Hansen (1) reported the existence of the uncrossed lateral CST in 6 (10%) of 60 human brains.
In brain rehabilitation, classification of the CSTs is important from two viewpoints: (i) understanding of the neurological manifestations of patients and prediction of their recovery courses and prognoses. This information allows clinicians to adopt more accurate rehabilitation strategies for brain-injured patients. For example, when a patient has preservation of the lateral CST in the affected hemisphere, the rehabilitation team can focus on the recovery of fine motor activity and strength of the affected hand (2, 3, 9, 12, 14–16, 18–21, 29, 34–38). By contrast, rehabilitation can be focused on the recovery of gait function or functional compensation of the affected upper extremity when the lateral CST in the affected hemisphere is completely injured or a complete injury of the lateral CST is predicted (8, 11, 18, 20, 21, 39–44). (ii) Although the role of the lateral CST has been elucidated relatively well, little is known about other CSTs that may substitute for the function of an injured CST. For example, the anterior CST has been suggested as a motor recovery pathway of the ipsilateral motor pathway from the unaffected motor cortex to the affected extremities along with the cortico-reticulospinal tract following injury of the lateral CST (41, 44–47). However, because identification and visualization of the anterior CST in the live human brain could not be achieved, this could not be confirmed. A study using DTI reported on a method of identification of the anterior CST; therefore, studies of the anterior CST are now possible (31). Further research into the CSTs, in addition to the lateral CST, in terms of their normal functions and roles in motor recovery are required.
cerebral origin of the corticospinal tract
Early in brain development, corticospinal neurons are distributed throughout the frontal, parietal, occipital and temporal lobes (48, 49). However, later in development, as a result of the elimination of axon branches projecting to the spinal cord from other cortical areas, distribution is restricted to the posterior frontal and anterior parietal lobes (48, 49). Therefore, the CST usually originates from the fronto-parietal cortices, including the primary motor cortex (M1), secondary motor area and somatosensory cortex (Fig. 1A). The multiple cerebral origins of the CST appear to be important in terms of various functions of CST fibres and the motor recovery mechanism: perilesional reorganization following M1 injury (7, 50–54). Many studies have reported differences in function of the CSTs according to cerebral origin, as follows: the M1; execution of movements, the supplementary motor area (SMA); planning and coordination of internally-generated movement, the premotor cortex (PMC); planning and coordination of visually guided movements, and the somatosensory cortex; descending control of somatosensory afferent inputs generated by movement (7, 50–53). These different functions appear to have an association with different neurological manifestations, such as weakness, apraxia and impaired somatosensory motor coordination following CST injury (52, 53, 55) (Fig. 1B). The motor representations (praxicons), which are stored in the dominant parietal cortex, help in programming of the premotor area, and the premotor area then helps in implementation of the required movements by selective activation of the M1, which innervates the specific muscle motor neuron pools required for performance of skilled movements (56).
A few studies have reported on the distribution of the CST origin in the cerebral cortex in the human brain (57–59). In 1967, Jane et al. (57) reported that 60% of CST fibres originated from the precentral gyrus and that the remaining fibres originated from the PMC and parietal lobe in a patient with brain injury. Since the introduction of DTI, two studies have reported on the origin of the CST (58, 59). In 2009, in a study of 42 healthy children, Kumar et al. (58) reported that the CST originated from both pre- and post-central gyri (71.4%), the precentral gyrus only (19%), and the post-central gyrus only (7.1%) . A study using DTI reported the distribution of the CST origin in 36 healthy adults (59). In this study, in order to avoid a crossing fibre effect, the M1, the primary somatosensory cortex (S1), and the dorsal premotor cortex were confined to the lateral margin of the precentral knob. The CSTs were found to originate from the M1 (36.9%), the S1 (31.7%), the SMA (24.7%) and the dorsal PMC (6.7%), respectively.
Several studies have reported the anatomical differences between CST fibres according to cerebral origin (59–62). In 1996, using an anterograde transport method, Dum & Strick (60), who investigated the pattern of spinal termination of efferents from the SMA and M1 in the macaque monkey, found that the extent and density of M1 labelling was greater than that of the SMA. Subsequently, in a study using densitometry analysis in the macaque monkey, Maier & Armand reported that projections from the M1 occupied 21–65% and those from the SMA occupied 1–6% (61). In 2002, using the same method in the macaque monkey, the same research group investigated differences in hand representations of the M1 and SMA and reported that corticospinal projections from the M1 showed far greater density and occupied a much greater proportion of hand muscle motor nuclei than those of the SMA (62). A study using DTI reported that CST fibres from the M1 and S1 have similar characteristics, unlike CST fibres from dorsal PMC or SMA (59). This characteristic of the S1, which is the second largest source of CST fibres, appears to be associated with the high incidence of reorganization into the S1 following an M1 infarct (54, 63–65).
In brain rehabilitation, the cerebral origin of CST fibres is important for the following reasons: (i) understanding the neurological manifestations of patients with brain injury in detail; patients with brain injury can show various neurological manifestations, such as motor weakness, limb-kinetic apraxia, or somatosensory-motor incoordination according to the cerebral origin of the injured CST fibres; (ii) CST fibres from a different cerebral origin can compensate for the function of the injured CST fibres. However, further studies into these aspects are required.
collaterals of the corticospinal tract
The CST has various collaterals (1, 27), the most representative of which is the aberrant pyramidal tract (APT). The APT indicates the collateral pathway of the PT, which separates from the original PT at the level of the midbrain and the pons, and descends through the medial lemniscus (66–69). Several studies have reported the incidence and courses of the APT (67–69). In 2001, using the modified Bielschowsky stain, Yamashita & Yamamoto (67) investigated the incidence and details of the course of the APT in 150 consecutive autopsied human brains. According to their findings, the APT was observed in all of the 150 brains examined, with the exception of one brain with holoprosencephaly; they also reported that the course of the APT left the PT within the cerebral peduncle and passed into the medial lemniscus of the pons through the upper medulla (67). In 2009, in a study using DTI, Hong et al. (68) reported the existence of the APT in 6 (21.4%) of 28 hemispheres of normal subjects and that the APT descended through the medial lemniscus from the midbrain to the pons, and then entered into the PT at the upper medulla. In 2011, using DTI, Kwon et al. (69) reported the existence of the APT in 18.3% of 186 hemispheres of the normal human brain. They also found that 26.5% of the APTs originated from the S1 without a M1 origin, although all of the PTs were found to originate from SM1 and the APT has different characteristics, including less directionality and fewer neural fibres than the PT.
Other studies have demonstrated that the APT could function as a motor recovery mechanism in patients with a cerebral infarct (37, 70–72). In 2009, Jang (70) reported on a patient with a pontine infarct whose motor function of the affected hand was recovered by the lateral CST, which descended through the APT instead of the infarcted pons. In 2010, Lindenberg et al. (37), who recruited 35 chronic patients with middle cerebral artery infarct, reported better motor outcome in patients who had alternate motor fibres in the posterior pons. Although they did not identify the exact pathway of the alternate motor fibres in their study, the alternate motor fibres appeared to be the APT, which separates from the original PT at the level of the midbrain and the pons and descends through the medial lemniscus. In a study using DTI, Yeo & Jang (72) reported on a patient with a cerebral peduncle infarct in the mid to lateral portion of the left cerebral peduncle who showed an APT originating from the M1 and descending through the medial lemniscus pathway from the midbrain to the pons with the discontinued PT in the affected hemisphere (72). During the same year, in a study using DTI, Hong & Jang (71) reported on a patient with a corona radiata infarct whose motor function was recovered by an APT, which was bypassed through the medial lemniscus from the midbrain to the lower pons with degeneration of the PT.
A few studies have demonstrated that the APT could function as a motor recovery mechanism; however, so far, it has not been widely accepted as a motor recovery mechanism. Therefore, further studies involving large numbers of patients are required. In addition, further study of the contribution of the APT to motor recovery and its mechanism, as well as rehabilitative strategies for triggering the APT following brain injury, are needed. Conduct of research into other collaterals, in addition to the APT, is also needed.
Development of the corticospinal tract
Martin (49) proposed 3 developmental stages of the CST in the human brain: (i) growth of axons of cortical lamina 5 neurons to the grey matter of the spinal cord during the late prenatal or early postnatal period; (ii) refinement of the grey matter terminations of the spinal cord during postnatal development (1–2 years after birth): elimination of transient termination and local growth in the grey matter of the spinal cord; (iii) motor control development: the role of the CST is expressed in control of distal limbs and other adaptive movements. On the other hand, 2 important time-periods have been suggested with regard to changes in the ipsilateral CST: 2 years – significant change in the ipsilateral CST; and 10 years – disappearance of the ipsilateral CST.
In 2001, using TMS, Eyre et al. (73) demonstrated bilateral innervations of spinal motoneuronal pools. For comparison of development of the ipsilateral and contralateral CSTs, they recruited 9 neonates, who were studied longitudinally for 2 years from birth and 85 healthy subjects (0–55 years). According to their findings, the thresholds of both CSTs were increased within the first 3 months after birth. Differential development was observed from 3 months, so that by 18 months the ipsilateral CST had significantly smaller amplitude, higher thresholds, and longer latencies than the contralateral CSTs. The amplitude of the ipsilateral CSTs diminished, with a parallel increase in amplitude of the contralateral CSTs (49). Consequently, it has been suggested that at around 2 years after birth, the ipsilateral CSTs have smaller amplitudes, longer latencies and higher thresholds than the contralateral CSTs (49, 73–75). The mechanism of this differential development between the ipsilateral and contralateral CSTs has been explained by a greater withdrawal of the ipsilateral CSTs than the contralateral CSTs, and faster growth of axonal diameters in the contralateral CSTs, which is driven by activity-dependent shaping of neuronal connectivity.
In 1997, Müller et al. (76) investigated development of the ipsilateral CST connection in 50 normal children (age range 3–11 years). According to their findings, the incidence of ipsilateral MEPs showed a decrease with ageing and the ipsilateral MEP was not observed in children older than 9 years and 9 months. The time of disappearance of the ipsilateral MEP is known to coincide in general with the time of disappearance of mirror movements, which are a phenomenon of involuntary movements that accompany voluntary movement in homologous muscles of opposite sides of the body and maturation of the corpus callosum region for transcallosal motor fibres at around 10 years after birth (77–81). Mirror movements are normally observed during early childhood, and decrease with development of the brain, and a marked disappearance was observed at the age of approximately 10 years (Cohen et al. (82): 9 years; Connolly & Stratton (77): 5–13 years; Lazarus & Todor (78): 8.5 years) (77, 78, 82)). On the other hand, in 2009, using DTI, Koerte et al. (81) reported the difference in fractional anisotropy on the corpus callosum region for transcallosal motor fibres between 2 age groups: 11 children (mean age 8.4; range 7–11 years) and 10 adolescents (mean age 15.6, range 15–17 years) . The basic mechanism of the disappearance of the ipsilateral CST and mirror movements has been explained by the inhibition hypothesis: maturation of a callosally mediated 2-way inhibitory system through which each hemisphere supresses the ipsilateral CST of the contralateral hemisphere (49, 76–80, 82–85).
For brain rehabilitation, the important question is which ipsilateral CSTs contribute to motor recovery according to the time of brain injury. Many studies have reported on the differences in motor evoked potential (MEP) between patients with congenital or perinatal lesions, and those with acquired adult brain lesions, such as stroke (34, 41, 45, 46, 73, 86–88). In detail, in the case of a congenital brain lesion or perinatal lesion, such as hemiplegic cerebral palsy, the ipsilateral CST from the unaffected motor cortex was similar to that of the contralateral CST (73, 86–88). This is because the ipsilateral CST from the unaffected motor cortex was reinforced for motor recovery. By contrast, the ipsilateral MEPs observed in adult hemiparetic stroke patients had smaller amplitudes with latencies of 5–14 ms longer than those of the contralateral MEPs (34, 45, 46). The ipsilateral motor pathway of adult stroke patients has been explained by the disinhibition hypothesis (46, 89, 90): the normal motor cortices maintain balance through transcallosal inhibition. However, in the case of stroke, decrease in interhemispheric transcallosal inhibition occurs from the affected side toward the unaffected side. Therefore, the unaffected motor cortex is recruited in order to counteract the loss of control induced by the lesion.
Several studies have attempted to elucidate this topic; however, little is known about the exact time of brain injury when the ipsilateral motor pathway could be attributed to reinforcement or disinhibition (91–94). In 1991, Benecke et al. (91) reported on differences in MEP between patients with congenital and acquired brain lesions. Two patients with congenital porencephaly showed MEPs with shorter latencies and larger amplitudes, whereas 6 other patients with later acquired brain damage, such as cerebral infarct or intractable epilepsy that occurred at least 8 years after birth, showed MEPs with long latencies and small amplitudes. Therefore, they suggested that reinforcement of the ipsilateral CST was responsible for residual motor functions in patients with early brain damage, whereas in patients with later acquired brain damage, the cortico-reticulospinal tract might play a dominant role in ipsilateral motor control. In 1997, Maegaki et al. (92) reported that mechanisms of reorganization differed according to the time of brain injury. They recruited 20 hemiplegic patients with cerebral lesions resulting from injuries that occurred at various times with congenital lesions, 1 patient with a birth lesion, 4 with infantile lesions (1–12 months) and 7 with childhood lesions (2–13 years). Findings on TMS using an 8-shaped TMS coil showed that bilateral MEPs of biceps brachii were elicited in patients with a later childhood lesion as well as those with early lesions (congenital or birth lesion). However, those of hand muscles were elicited only in patients with lesions up to 2 years. They suggested that axonal sprouting for the ipsilateral CST occurred not only in early gestation, but also in the early postnatal period, at least up to 2 years. In 1999, Nezu et al. also reported on the different characteristics of ipsilateral CSTs between patients with hemiplegic cerebral palsy and those with acquired lesions (hemiconvulsion-hemiplegia-epilepsy syndrome) (93).
Further studies to elucidate the exact timing of reinforcement of the ipsilateral CST are required. In addition, clinical studies are needed into the differences in clinical significance between reinforcement and disinhibition of ipsilateral motor pathways with regard to motor function, mirror movements, and identification of the motor pathway (34, 45, 46).
Conclusion
This review examined the CST in terms of classification, cerebral origin, collaterals, and development in the viewpoint of brain rehabilitation. The CST is one of the major neural tracts in the human brain. Therefore, precise and thorough knowledge of the CST is necessary for successful brain rehabilitation; however, information about this topic from the viewpoint of brain rehabilitation is lacking. This omission appears to be related to the tools used for evaluation of the CST. In the past, TMS and functional neuroimaging techniques have been used mainly for research into the CST. These evaluation tools have limitations in that they are not capable of identification and visualization of the CST. By contrast, the recently developed DTI has the advantage of overcoming these limitations. In addition, DTI would be useful for studies of the anterior CST, the collaterals of the CST, and the cerebral origins of the CST. Therefore, further studies combining DTI, TMS and functional neuroimaging techniques are needed. In addition, comparative studies of the non-CSTs, including the reticulospinal tract, vestibulospinal tract, and rubrospinal tract are required in order to elucidate motor function in the normal brain and motor recovery following brain injury from the viewpoint of brain rehabilitation.
Acknowledgements
This work was supported by the DGIST R&D Program of the Ministry of Education, Science and Technology of Korea (14-BD-0401).
References
