Jan-Christoph Kattenstroth, PhD1*, Tobias Kalisch, PhD1,2*, Rebecca Kowalewski, MSc1, Martin Tegenthoff, MD2 and Hubert R. Dinse, PhD1
From the 1Neural Plasticity Laboratory, Institute for Neuroinformatics, and 2Department of Neurology, BG-Kliniken Bergmannsheil, Ruhr-University Bochum, Bochum, Germany. *These authors contributed equally.
OBJECTIVE: To assess joint position sense performance in subacute stroke patients using a novel quantitative assessment.
DESIGN: Proof-of-principle pilot study with a group of subacute stroke patients. Assessment at baseline and after 2 weeks of intervention. Additional data for a healthy age-matched control group.
SUBJECTS/PATIENTS: Ten subacute stroke patients (aged 65.41 years (standard deviation 2.5), 4 females, 2.3 weeks (standard deviation 0.2)) post-stroke receiving in-patient standard rehabilitation and repetitive electrical stimulation of the affected hand.
METHODS: Joint position sense was assessed based on the ability of correctly perceiving the opening angles of the finger joints. Patients had to report size differences of polystyrene balls of various sizes, whilst the balls were enclosed simultaneously by the affected and the non-affected hands. A total of 21 pairwise size comparisons was used to quantify joint position performance.
RESULTS: After 2 weeks of therapeutic intervention a significant improvement in joint position sense performance was observed; however, the performance level was still below that of a healthy control group.
CONCLUSIONS: The results indicate high feasibility and sensitivity of the joint position test in subacute stroke patients. Testing allowed quantification of both the deficit and the rehabilitation outcome.
Key words: rehabilitation; intervention; stroke; proprioception; quick test.
J Rehabil Med 2013; 45: 00–00
Correspondence address: Hubert Dinse, Institute for Neuroinformatics, Ruhr-University Bochum, Universitätsstraße 150, Building NB, Room 3/67, DE-44780 Bochum, Germany. E-mail: hubert.dinse@rub.de
Accepted Jun 4, 2013; Epub ahead of print Sep 16, 2013
Introduction
Proprioception refers to the perception of the position and motion of one’s body in space, and is based on afferent sensory information from muscle spindles (1, 2), cutaneous receptors, and joint mechanoreceptors (3). Studies have demonstrated the importance of proprioception for generating coordinated movements (4, 5), such as reaching and grasping, and the maintenance of static balance (6), and locomotion (7). Consequently, without intact proprioception as occurring in deafferented patients, goal-directed movement (8), prehension (9), accurate aiming, reaching, and tracking movement is severely impaired (4, 10–12). Similarly, the amount of damage of neural circuits mediating proprioceptive functions in stroke patients may be of prognostic importance for self-care, likelihood of discharge home, and period of rehabilitation (13, 14). In addition to the sensorimotor deficits, typically occurring after cerebral infarction, proprioceptive abilities are impaired in up to 50% of patients (14–16). It is possible that largely intact somatosensory input is required for sensorimotor learning (17, 18), and thus for sensorimotor rehabilitation. Studies have shown that the degree of proprioceptive impairment predicts motor recovery and long-term rehabilitation outcome (15, 19, 20).
However, the above-mentioned correlation between proprioceptive impairment and sensorimotor recovery of the upper extremities must be interpreted with caution given the limitations of the clinical tools used for the evaluation of proprioceptive performance. As emphasized in 2 studies by Dukelow et al. (15, 21) the clinical tools used for the assessment of proprioceptive performance have a poor inter- and intra-rater reliability, and a poor or absent normal value criteria (22). In a clinical surrounding, proprioceptive performance is typically assessed by evaluating the patients’ ability accurately to discriminate upward or downward positioning of a passively moved limb or finger (22), as in the thumb localization test (23). To this aim, the limb is manually guided to test positions that are typically not quantified, and the response is given verbally by the patient, making these assessments relatively insensitive (13). Thus, often used clinical measures of proprioceptive functions are not quantitative, lack objectivity, sensitivity, and are often confounded by other impairments (13, 22, 24, 25).
New technological approaches have enabled the development of robotic-aided assessments of proprioception and motor function (15, 26–28). However, these assessments typically require sophisticated technical equipment, qualified experimenters, and, most importantly, they are not mobile and not suitable for a quick administration, which limits their usage in a clinical surrounding. Therefore, new methods are needed that allow rapid, but reliable, assessment even in clinical settings. First, the test should be carried out quickly, which is necessary for patients who are not able to participate in longer assessments. Secondly, the test should be movable for repeated use at different locations without sophisticated equipment, and thirdly; the test should be related to sensorimotor everyday life performance. Finally, the test should be able not only to quantify the impairment of proprioceptive performance, but should be sensitive enough also to determine the efficacy of rehabilitation measures, thus helping to develop targeted individual rehabilitation strategies.
Recently, Kalisch and co-workers (29) introduced a novel quick test on joint position sense of the human hand. Thereby the subjects’ ability to rate volume differences between 2 spherical objects enclosed with their hands was utilized to assess indirectly the sensitivity of the joint position sense. By varying the sizes of the used objects in this contralateral concurrent matching task they were able to quantify this proprioceptive performance in subjects across their lifespan.
The present study applies the previously presented joint position sense assessment (29) to subacute stroke patients for the first time. The aim of the study was to prove the usability of this test for the quantification and documentation of changes over time, elicited either by therapeutic intervention or spontaneous recovery.
Methods
Clinical scales
We used 3 different clinical scales, closely related to everyday life performance, to quantify the subjects’ upper extremity movement disability.
The Medical Research Council Scale (MRC) (30) is often used in clinical practice to describe the muscle strength of the hand muscles. In this scale, muscle strength is graded on a scale ranging from 0 to 5, where 5 indicates full active range of motion and normal muscle resistance, 4 indicates full active range of motion and reduced muscle resistance, 3 indicates full active range of motion and no muscle resistance, 2 indicates reduced active range of motion and no muscle resistance, 1 indicates no active range of motion and palpable muscle contraction only, and 0 indicates no active range of motion and no palpable muscle contraction. Scoring depends on the judgement of the examiner.
The Frenchay Arm Test (FAT) is a measure of upper extremity proximal motor control and dexterity during activities of daily life (ADL) performance and is designed to measure activity limitation (31). The FAT consists of 5 pass/fail tasks, i.e. combing hair, bringing a waterglass to the mouth, picking up a wooden cylinder and manipulating small objects (ruler and clothespin). The subject scores 1 for each task that is completed successfully. Patients scoring 5 out of 5 are likely to use their affected upper limp, even if they feel it is not normal.
The Wolf Motor Function Test (WMFT) (32) quantifies upper extremity movement ability through timed single- or multiple-joint motions and functional tasks. The tasks are arranged in order of complexity, progress from proximal to distal joint involvement, test total extremity movement and movement speed, and require few tools and minimal training for test execution. Tasks 1–6 of the WMFT involve timed joint-segment movements, and tasks 7–15 consisted of timed integrative functional movements (see (32) for details). All tasks are performed as quickly as possible and are truncated at 120 s.
Testing of joint position sense in a clinical population
Joint position sense performance was assessed in subacute stroke patients (n = 10, mean age 65.41 years (standard deviation (SD) 2.5), 4 females, 2.3 weeks (SD 0.2) post-stroke) characterized by a MRC score of 3.7 (SD 0.12) (affected hand) and 4.9 (SD 0.1) (non-affected hand) (p ≤ 0.001), a FAT score of 3.15 (SD 0.31) (affected hand) and 4.9 (SD 0.1) (non-affected hand) (p = 0.005) and a WMTF score of 10.71 (SD 0.86) (affected hand) and 14.8 (SD 0.2) (non-affected hand) (p = 0.01) using a spherical hand grasp-matching task (29). Patients had a left or right cerebral artery thromboembolic infarction affecting the upper extremity and were receiving in-patient standard rehabilitation of physio- and ergo-therapy and repetitive electric stimulation (33, 34) of the affected hand (45 min/day, 5 days/week for a period of 2 weeks). Recovery of joint position sense was assessed once at baseline and again after 2 weeks of intervention in a hospital-based rehabilitation unit. The local ethics committee approved the study, and all subjects provided written informed consent before participating. In addition, we added data (n = 10) from healthy age- and gender-matched subjects (mean 65.6 years (SD 1.35), 5 females) who participated in a previous study conducted by our group (29).
Joint position sense assessment using a matching task (Bochum Joint Position Sense Assessment)
Joint position sense assessment was conducted using the Bochum Joint Position Sense Assessment (BJPSA), as reported previously (29). All patients were asked to compare lightweight polystyrene balls with different diameters with a reference ball in their non-dominant hand without visual information. The patients had to place their hands with palms upwards on a cushion covered by a screen. Each set of polystyrene balls (test and reference ball) was placed in the patients’ opened hands by the experimenter. The patients were instructed to enclose the balls with their fingers. Exploring the balls by repeated opening and closing of the fingers was not permitted. The patients were asked to indicate within 5 s whether they perceived the tested object (located in the affected hand) to be larger, smaller, or equal in volume compared with the reference ball. In 3 consecutive subtests, the complete set of polystyrene balls (126; 210; 252; 294; 337; 421; and 505 cm3) was compared with a small reference (210 cm3), a mid-sized reference (294 cm3), and a large reference (505 cm3) (Figs 1 and 2). The test Bochum Joint Position Sense Assessment was conducted using a custom-made software, which randomizes the order of the references and the test balls. After each trial, the experimenter enters “bigger”, “smaller”, or “equal” with a single mouse-click according to the patients’ answer. The software calculates the number of errors (NE, total of 21 decisions), the weight of errors (WE, volume difference of reference and test object), and the direction of errors (DE), while the DE was calculated by summing up all errors where the size of the reference ball was overestimated (+1 for every error) or underestimated (–1 for every error). The entire test took approximately 5 min. The described test has a high test-retest reliability in healthy subjects, which was shown for NE (Cronbach’s alpha, 0.787, n = 45), WE (Cronbach’s alpha, 0.761, n = 45), and DE (Cronbach’s alpha, 0.766, n = 45) (29).
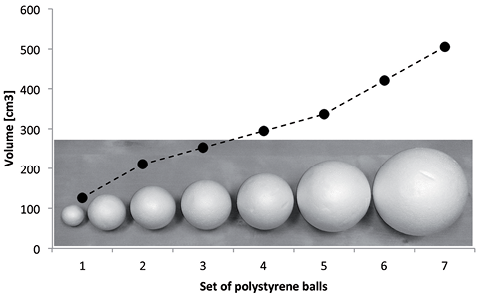
Fig. 1. Set of polystyrene balls used for the assessment of joint position sense. A set of 7 polystyrene balls (volumes 126, 210, 252, 294, 337, 421 and 505 cm3) was used to quantify the performance of the joint position sense of the subjects. Two balls had to be enclosed with both hands simultaneously to detect size differences. As the “enclosure” is a typical and highly stereotypical explorative procedure to gather information about an object’s volume (37–39), all calculations were carried out in units of cubic centimetres (cm3). (Photograph reproduced with permission from (29)).
Fig. 2. Assessment of joint position sense. A complete set of polystyrene test balls (T) (volumes 126, 210, 252, 294, 337, 421 and 505 cm3) was compared with a (A) large reference (505 cm3) (R), (B) mid-sized reference (294 cm3) (R), and (C) small reference (210 cm3) (R). The patients were instructed to enclose the balls with their fingers. Exploring the balls by repeated opening and closing of the fingers was not permitted. The patients had to indicate within 5 s whether they perceived the tested object to be larger, smaller, or equal in volume compared with the reference.

Statistical analysis
In all cases, we report the data as mean and standard error of the mean (SEM). Normality of data was investigated using Kolmogorov-Smirnov test (NE, p = 0.921; WE, p = 0.973, DE, p = 0.617). We used Student’s t-tests to detect differences in the joint position sense performance and the clinical scores after 2 weeks of intervention. To investigate the baseline dependency of individual gains in performance, correlation analyses were conducted using 2-sided Spearman’s correlations. Statistical analyses were carried out using SPSS statistic software (v.20, IBM Corporation, NY, USA).
Results
Spherical hand grasp-matching task
Joint position sense performance was assessed by calculating the NE, the WE (volume differences of reference to test object) and the DE (summing up all errors where the size of the reference ball was overestimated) (+1 for every error) or underestimated (–1 for every error). After 2 weeks of intervention the NE decreased significantly from 5 (SD 0.7) to 3.2 (SD 0.5) (p = 0.024) (Fig. 3A). Furthermore, the WE decreased significantly from 244.05 (SD 48.1) to 84.18 (SD 30.1) (p = 0.001) (Fig. 3B). However, after the intervention the performance level was still below the control group performance levels for NE (0.8 (SD 0.25), p = 0.001) and WE (12.63 (SD 6.43), p=0.022). Moreover, no changes were found in the DE following the intervention (mean –0.8 (SD 0.6) to –0.2 (SD 1.2), p = 0.733). It is noteworthy that mean DE before and after the intervention was on the same level as in the control group (p ≥ 0.099) (Fig. 3C). Correlation analyses (Spearman’s) between baseline performance and improvement following intervention revealed significant correlations for WE (r = –0.707, p = 0.022), while no significant correlations were found for NE (r = –0.560, p = 0.092).
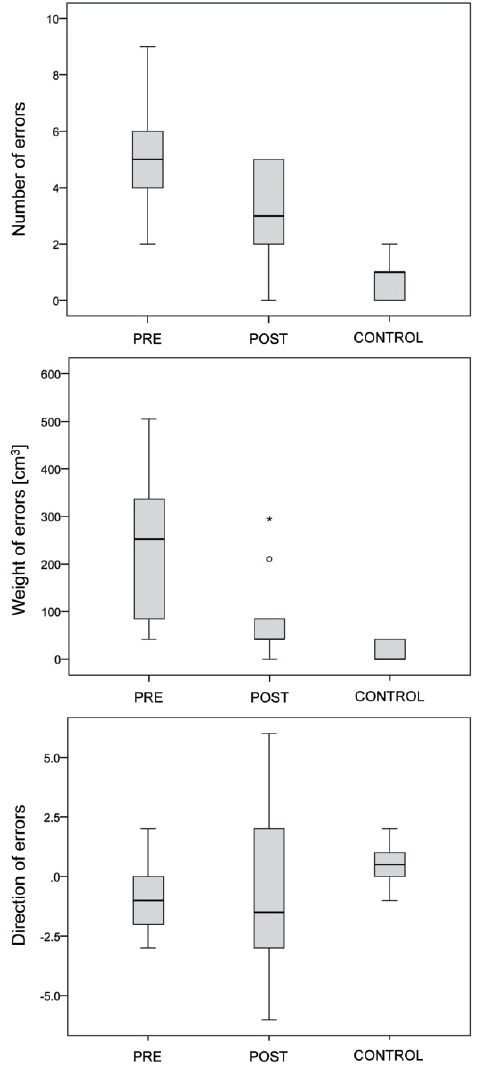
Fig. 3. Joint position sense performance. Joint position sense performance, i.e. (A) the number of errors, (B) the weight of errors, and (C) the direction of errors (DE), before and after 2 weeks of intervention and for age-matched controls (n = 10). Both (A) the number of errors (5 (SD 0.7) to 3.2 (SD 0.5), p = 0.024) and (B) the weight of errors (244.05 (SD 48.1) to 84.18 (SD 30.1), p = 0.001) decreased significantly after intervention. No differences were found for (C) the DE (–0.8 (SD 0.6) to –0.2 (SD 1.2), p = 0.733). For control group data see, text. Horizontal lines within the boxes: medians. Boxes: top and bottom quartiles. Whiskers: minima and maxima within 1.5 interquartile range (IQR). Solid dots: outliers (> 3.0 IQR). *Extreme values.
Joint position sense and clinical scales for motor performance
After the rehabilitation period of 2 weeks we found a significant change in the subjects’ clinical scales. Compared with the initial performance there was a significant increase in the mean MRC (+0.5 (SD 0.1), p = 0.009), the FAT (+1 (SD 0.46), p = 0.045) and the WMFT (+2.4 (SD 0.99), p = 0.037). To investigate the relationship between the observed changes in joint position sense, as assessed with the spherical hand grasp-matching task, and functional changes in motor abilities, as depicted by the used clinical scales, correlation analyses were performed. We found a significant correlation between the individual gain in joint position sense performance, as quantified by the NE (r = –0,756, p = 0.011), the WE (r = –0.740, p = 0.014), and the increase in the MRC scale. There were no significant correlations for changes in joint position sense performance and the FAT (NE, p = 0.065; WE, p = 0.350) or WMFT (NE, p = 0.204; WE, p = 0.655).
Discussion
The present study tested the sensitivity of a previously introduced joint position sense assessment (29) in subacute stroke patients to detect changes in hand performance following a rehabilitation period of 2 weeks.
Joint position sense performance was assessed by calculating the NE, WE, and the DE in a contralateral concurrent matching test, i.e. the detection of size differences in a set of polystyrene balls with both hands simultaneously. The results of this pilot study revealed a significant improvement in joint position sense performance after an intervention period of 2 weeks for the variables NE and WE. Individual changes in performance were significantly correlated with increase in strength of the hand muscles, which was assessed with the MRC scale. As accurate grip strength is a necessary prerequisite for the enclosure of objects, this finding fits into the context. The lack of correlation between changes in FAT and WMFT and joint position sense might be explained by methodological constraints. In the past the FAT has sometimes been criticized for lacking assessment of quality of movement and performance (35). In addition, clients were found to either pass or fail all or most subtests, indicating that the FAT may not be sensitive to change (36). The WMFT, on the other hand, assesses the functional use of the complete upper extremity and not only the hand, as is the case for the proprioception test used.
Performance levels after the intervention were still below the performance of healthy age-matched controls, emphasizing the high discriminative validity of this new assessment. Thus, the novel assessment seems to be suitable for use in clinical settings, since it can be performed quickly, allows repeated measurement over time, reveals a high test-retest reliability (29), and is sensitive to even small changes in impairment.
Assessment of proprioception performance in clinical settings
The ability to sense the static position of a joint or limb segment is the most common assessment of proprioceptive acuity in clinical settings (37, 38). Tests of position sense typically focus on the accuracy by which an individual can identify or match a target joint angle in the absence of vision and are conducted as either ipsilateral remembered matching tasks or contralateral concurrent matching tasks (see (37) for review). In many previous studies a thumb localization test is used, since it represents the clinical reference standard in evaluation of joint position sense (23). However, measures of this type lack objectivity and sensitivity, they are not quantitative (13, 22, 25, 39) since the clinician guides the limb manually, positions are not quantified, and judgement is made subjectively.
The current study employed a recently introduced joint position sense assessment that utilizes enclosure, a highly stereotypical posture of the hand (29). Enclosure belongs to the so-called “exploratory procedures” involved in human haptic processing (39–41) and is the first step in gathering basic information about an object, such as shape and volume (see (42) for details). Therefore, the joint position sense test is highly related to sensorimotor everyday competence, which is the major target of all clinical rehabilitation measures (13, 15). Kalisch and colleagues (29) reported NE to be the main variable describing age-related changes in healthy joint position sense of the hand. The variable WE, which was also affected by age, but to a much lesser extent, showed no significant increase between mid-aged and older adults, pointing towards a saturation of this variable with increasing age.
Our data showed a significant improvement in joint position sense performance in subacute stroke patients after 2 weeks of standard therapeutic intervention. As the number of subjects in this pilot study is too small to draw further conclusions, additional research is needed to clarify the impact of the variables WE and NE for the description of stroke-related proprioception impairment and their importance for the documentation of rehabilitation outcome.
Methodological constraints in assessing joint position sense in patients
There are various different approaches for the assessment of proprioceptive performance, and especially joint position sense, in patients (13, 15, 21, 23, 25, 43, 44). If an assessment is based on the comparison of angle positions it is crucial to deliberate about whether the testing is performed as an ipsilateral remembered matching or a contralateral concurrent matching paradigm. As stroke is an age-associated pathological condition, which is prevalent in adults and older adults, we wanted to rule out memory components that might affect the assessment of proprioception performance. The use of the “unaffected” hand as a reference in a bilateral concurrent matching tasks helps to avoid potential confounding of decreased memory abilities, concentration, and attention.
Nevertheless, it is clear that the “unaffected” hand in hemiparetic stroke patients might also be affected to some extent and therefore bias individual decisions. As reported previously, the assessment applied in the present pilot study reveals high specificity, as there were no significant correlations between joint position sense performance and tactile acuity or dexterity in healthy subjects (29). Since this assessment accounts for whole-hand proprioception and aims at assessing everyday life relevant performance, contrasting other assessments evaluating joint position sensation of single joints, e.g. in one finger (45), is of utmost importance for clinical populations.
Conclusion
The results of this pilot study indicate the impact of the joint position test BJPSA for the assessment of performance in subacute hemiparetic stroke patients. Testing revealed significant changes in performance following 2 weeks of intervention and allows the exact quantification of the deficit compared with healthy subjects’ performance. Further research with a larger number of patients is necessary to corroborate these preliminary findings.
Acknowledgements
This research was supported by Grants from the German Research Foundation (Deutsche Forschungsgemeinschaft, DFG) to HRD (Di 334/19-1, SFB 874) and MT (Te 315/4-1, SFB 874). The authors would like to thank Andreas Korte for programming the BJPSA software.
References
