Monica Skarin, MSc1*, Anna Sjöholm, MSc1*, Åsa Lundgren Nilsson, PhD1, Michael Nilsson, PhD1,2, Julie Bernhardt, PhD3,4 and Thomas Lindén, PhD1,3
From the 1Institute of Neuroscience and Physiology, the Sahlgrenska Academy, University of Gothenburg, Sweden, 2Hunter Medical Research institute, Newcastle, 3Stroke Division, the Florey Institute of Neuroscience and Mental Health and 4LaTrobe University, Melbourne, Australia. *The 2 first authors contributed equally to this study.
OBJECTIVE: To establish physical activity level, environment and social interaction in routine stroke rehabilitation practice.
DESIGN: Prospective observational study using behavioural mapping.
METHODS: Patients at 4 hospital stroke rehabilitation units in Sweden at least 7 days post-stroke were observed over 1 week-day at 10 min intervals between 08.00 h and 17.00 h. At each observation, physical activity, location and people present were ascertained.
RESULTS: Patients (n = 104) had a mean age of 70.3 (standard deviation (SD) 14.4) years. Median time since stroke was 19 (range 7–142) days. Patients were alone for 52% of the day, during this time 7% was spent in standing/walking activities. While with a physiotherapist patients were standing/walking for 43% of the time. Using median regression it was found that the median percentage of time spent in standing/walking was associated with gait independence and age. Gait independence increased the median percentage of time spent in standing/walking by 16.5% (95% confidence interval (CI) 12.1–20.9, p < 0.001), while an increase of 1 year of age reduced the median percentage of time spent in standing/walking by 0.16% (95% CI –0.31 to –0.01, p < 0.05).
CONCLUSION: Patients had low levels of physical activity and social interaction. This study suggests that there is a huge potential to increase patients’ activity level and augment social interaction above current levels.
Key words: stroke; rehabilitation; physical activity; observation.
J Rehabil Med 2013; 45: 997–1003
Correspondence address: Thomas Lindén, Institute of Neuroscience and Physiology, University of Gothenburg, Per Dubbsg 14, SE-41345 Göteborg. E-mail: thomas.linden@neuro.gu.se
Accepted May 20, 2013; Epub ahead of print XXX?, 2013
INTRODUCTION
It is well known that organized care in stroke units, compared with general medical wards, increases the chance of survival and physical independence after stroke (1). One factor distinguishing a stroke unit from a general ward appears to be earlier mobilization (2–4).
Several meta-analyses support the hypothesis that higher intensities of treatment and increased exercise therapy time result in improvements in functional outcome, such as activities of daily living (ADL) and gait speed after stroke (5–7). Current practice guidelines (8) do not specify or recommend a certain level of physical activity in rehabilitation. Although studies have shown that patients can tolerate considerably higher levels of physical activity than “standard care” (9), current evidence is insufficient to allow development of clear guidelines about the optimal timing and dose of physical activity in stroke rehabilitation. There is, however, some indicative information about current activity levels of patients undergoing rehabilitation.
A recent systematic review (10) of observational studies examining physical activity of hospitalized stroke patients concluded that it is common to find low levels of physical activity among patients undergoing rehabilitation (10). In an American study (11), time spent with therapists took up, on average, 20% of the day, whereas inactive behaviour constituted nearly 50% of patients’ time (11). Further data from an observational study (12) comparing 4 rehabilitation centres in Europe (Belgium, the UK, Switzerland and Germany); found that patients spent more than half the working day alone and physically inactive. The findings from the study also suggest a great geographical variation in the quantity of therapy stroke patients receive, with time spent with a therapist ranging from 7% in the UK to 22% in the Swiss unit. Self-directed exercise also differed between centres, ranging from less than 1% in the Swiss unit to 6% in the German unit (12).
In view of the evidence that improved functional outcome can be achieved with more intensive rehabilitation, and the knowledge that significant variation exists between sites or countries, we sought to determine the current activity patterns of stroke patients managed in Swedish rehabilitation units. This study could provide baseline data for a randomized controlled trial, which is necessary to build the evidence-base for optimal timing and dosing.
The study was designed for measuring care processes that could be observed openly and had a sound evidence-base; physical activity levels together with levels of contact with multi-disciplinary team members (1, 2).
The specific aims of this study were: (i) to describe the observed levels of physical activity, specifically when patients were alone or with others (e.g. nurses, doctors, therapists); (ii) to describe the observed levels of interaction between patients and members of the multidisciplinary team; and (iii) to examine factors (e.g. age, stroke severity) that may be associated with high levels of physical activity.
MethodS
Subjects and settings
The study was undertaken in 4 rehabilitation units in the Swedish West Gothia region at Södra Älvsborg Hospital, Borås, Skaraborg Hospital Skövde, Sahlgrenska University Hospital, Gothenburg, and NU Hospital Uddevalla. The centres were physical medicine and rehabilitation-, internal medicine-, geriatric- or combined units. Patients (> 18 years) with confirmed first or recurrent stroke (as defined by the World Health Organization; WHO) (13), at least 7 days post-stroke were eligible for inclusion. Those receiving palliative care were excluded. Approval for this study was obtained from the Regional Ethical Review Board of Gothenburg, registration number 421-09, and informed consent was obtained prior to inclusion.
Study design and duration
This was a prospective, observational study using behavioural mapping. Data were acquired over a 10-month period between November 2009 and September 2010.
Observational technique
Each patient was observed on a week-day from 08.00 h to 17.00 h, i.e. the most active part of a patients’ day. Standardized behavioural mapping was employed, with observations every 10 min, with the exception of a 30-min lunch break randomly scheduled between 11.20 h and 13.40 h. At each time-point a trained observer recorded patient activity, the person attending the patient and their location on mapping forms. If feasible, patients were followed off-ward, but not intruded on if in private situations (e.g. visiting the bathroom). However, once patients were again visible, the patient or caregiver was questioned to determine the activity during the unobserved period. This observational method has been detailed previously (14). Inter-observer agreement has been verified in previous behavioural mapping studies, showing a close association for activity, location and people present (14, 15). In addition, the method has been shown to be valid when compared with an activity monitor (16).
Procedure
Patients and staff were informed that patients’ activity throughout the day would be monitored, and it was emphasized that they should do nothing different. Eligible patients were informed and recruited to the study by a member of the local staff. To avoid selection biases in those cases where more than 7 patients met inclusion criteria, staff were given the instruction to recruit those patients who were born on earlier dates each month (e.g. if one patient was born on 8 January, one on 11 September and one on 25 November, the first two would be recruited). Observations were made at each unit on 4–6 separate occasions. The unit was screened prior to the planned day of observation, to determine whether observation was feasible. Mapping was considered feasible if a minimum of 2 and maximum of 7 patients were available on the day.
Categories of physical activity
Eleven whole-body physical activities (e.g. lying, sitting, standing) could be recorded at each observation. These were grouped into 5 pre-specified activity categories (ACs); to reflect the level of physical work during these activities. The categories have been classified by experienced clinicians and used in previous behavioural mapping studies (14, 17). The 5 categories were: AC0: no activity (asleep, inactive – no motor activity); AC1: non-therapeutic activity (eat, talk, read in bed); AC2: minimal therapeutic activity (sit in chair); AC3: moderate therapeutic activity (sit unsupported, transfer feet on floor); and AC4: high therapeutic activity (standing, walking, stairs).
People present and location of activity
The 11 categories for social interaction included nursing and medical staff, therapists (e.g. occupational therapists (OT), physiotherapists (PT), speech therapists (ST), family (immediate and other visitors), other patients, etc. The 6 categories for location were bedroom, hall, bathroom, therapy area, patients’ lounge and off-ward.
Pre-specified data
Patient demographics were extracted from the medical records. Demographics that helped describe the patient and their stroke were recorded, e.g. gender, age, time since stroke, first ever stroke, side of lesion, living arrangements and mobility prior to stroke, and whether Swedish was the patient’s first language. Stroke type was classified according to the Oxfordshire Community Stroke Project classification (18). We recorded neurological impairment at time of admission to the acute hospital by extracting a National Institutes of Health Stroke Scale (NIHSS) (19) score from the medical records (20). Patients were grouped into mild (NIHSS < 8), moderate (NIHSS 8–16), and severe (NIHSS > 16) stroke categories (21). To estimate cognitive impairment the COG-4 (22) subscale was used. The COG-4 is a proposed tool for establishing stroke patients’ cognitive performance and consists of 4 extracted items corresponding to the NIHSS items on orientation, executive function, language and inattention. Disability prior to the current stroke event was estimated using the Modified Rankin Scale (23). The Barthel Index (24) was used to determine ADL capacity on the day of observation. Patients’ level of motor function at the time of observation was also recorded by the observer (with assistance from treating staff if not evident), using the Mobility Scale for Acute Stroke (MSAS) (25). Performance on 5 activities (bridging, supine to sit, sitting, standing up, and walking) was examined; an MSAS score of 1 demonstrating that the patient is not capable of carrying out the activity, and a score of 6 demonstrating that they are unassisted and safe.
Analyses
The highest level of activity (5 ACs) in every 10-min interval was recorded in the database (Microsoft Access 2000). Results are expressed in relative numbers as a function of time, 100% being equal to a behaviour lasting a full 9-h day (08.00–17.00 h). When averages are reported, observed data were pooled across sites and over days. Where the duration (min) of a specific behaviour is reported, the number of consecutive observations in which the behaviour occurred were noted and the assumption made that the behaviour between 2 observations did not alter. Therefore, when minutes are reported these should be considered approximate. When reporting nursing data, nurses and nurse assistants were combined. To analyse activity levels and social interaction, descriptive statistics were used. To analyse factors associated to high physical activity (e.g. standing and walking; AC4) median regression was used. Included variables were: age, stroke severity (NIHSS), cognitive function (COG-4) and walking ability (MSAS walking item was re-coded into gait independent, i.e. a score of 6 on the walking item, and gait dependent, i.e. a score of 1–5 on the walking item). Patients who scored > 0 points on any of the 4 items on the COG-4 were considered cognitively impaired. All statistical analyses were performed with IBM SPSS statistics version 19.0.
Results
A total of 5,376 observations were performed on 104 patients; 26 patients in each hospital. On 338 occasions (6.3% of observations) it was not possible to ascertain the type of motor activity, primarily when patients were in private situations (2.5%), off-ward (1.7%), or when a patient treatment, for any reason, could not be directly observed (1.9%). Patient characteristics are shown in Table I.
|
Table I. Patient characteristics (n = 104) |
|
|
n (%) |
|
|
Age Mean (SD) [range] |
70.3 (14.4) [34–93] |
|
Men, n (%) |
55 (53) |
|
First-ever stroke , n (%) |
84 (81) |
|
Time since stroke, days, median (IQR) [range] |
19 (12–34) [7–142] |
|
Stroke type, n (%) |
|
|
Infarct |
75 (72) |
|
Haemorrhage |
29 (28) |
|
NIHSS |
|
|
Median (IQR) [range] |
8 (4–18) [0–34] |
|
Mild stroke, NIHSS < 8, n (%) |
48 (47) |
|
Moderate stroke, NIHSS 8–16, n (%) |
26 (25) |
|
Severe stroke, NIHSS > 16, n (%) |
29 (28) |
|
COG-4, n (%) |
|
|
Any cognitive impairmenta |
68 (66) |
|
> 1 cognitive domain impairedb |
51 (50) |
|
Barthel |
|
|
Median (IQR) [range] |
65 (35–95) [0–100] |
|
MSAS |
|
|
Median (IQR) [range] |
28 (16–30) [5–30] |
|
OCSP infarct classification |
|
|
LACI |
6 (6) |
|
PACI |
30 (29) |
|
POCI |
14 (14) |
|
TACI |
23 (23) |
|
Pre-stroke mobility, n (%) |
|
|
Independent no aids |
85 (82) |
|
Independent with aid |
16 (15) |
|
Dependent |
3 (3) |
|
aCOG-4 score >0; bCOG-4 score >1 SD: standard deviation; IQR: interquartile range; NIHSS: National Institute of Health Stroke Care; COG-4: Cognitive Function Subscale; MSAS: Mobility Scale for Acute Care; OCSP: Oxford Community Stroke Project Classification; LACI: lacunar infarct; PACI: partial anterior circulation infarct; POCI: posterior circulation infarct ; TACI: total anterior circulation infarct. |
|
Unit characteristics
The 4 rehabilitation facilities had between 18 and 21 dedicated stroke beds and their mean length of stay ranged from 11 to 38 days. All units had a therapy room on-site; however, this was sometimes located on a different floor. Staff patient ratios were from 1:1.5 to 1:3 for nurses and nurse assistants, from 1:4 to 1:8 for PTs, from 1:4 to 1:8 for OTs, from 1:14 to 1:90 for STs and from 1:5 to 1:7 for doctors. Our observations were made between 08.00 h and 17.00 h. In all units, therapists worked a 5-day week only.
Time in physical activity
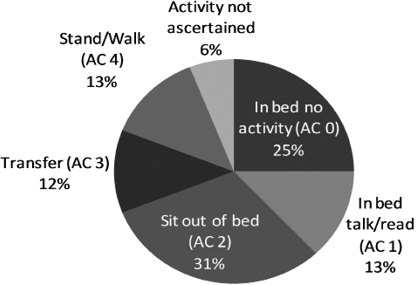
Observations of activity are shown in Fig. 1. Patients spent only 13% (approximately 70 min) in standing and walking activities during the day, while in comparison, 38% (nearly 3.5 h) was spent in bed, primarily (> 2 h) with no motor activity.
Fig. 1. Physical activity observed between 08.00 h and 17.00 h, averaged across all cases.
Time in physical activity by type of company
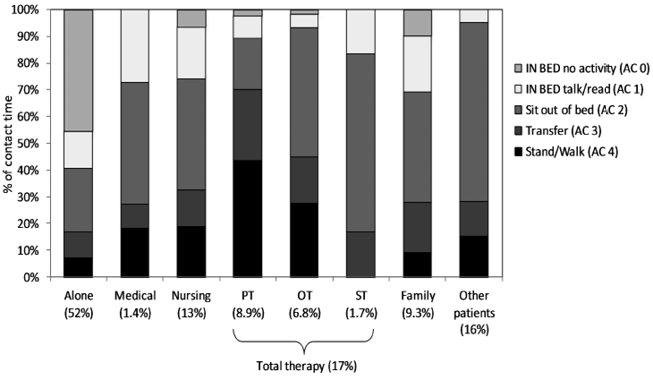
Fig. 2 shows the level of activity (AC 0–4) patients engaged in post-stroke when they were alone (52% of the day, i.e. > 4.5 h), and with therapists, doctors, nurses, family and other patients. As expected, patients were least physically active when they were alone with 59% (approximately 3 h) of their time alone spent in bed. For only 7% of their time alone were they standing or walking. We expected those patients who were independent in walking (46%, n = 48) to stand and walk when alone and, accordingly, found that they accounted for 97% of the observed standing and walking time when alone. Patients were, not surprisingly, most physically active while with a PT (this was true both expressed as a percentage of time and time in min); during 43% (approximately 21 min) of the time patients spend with a PT they were engaged in standing and walking activities. When with an OT patients spent 28% (approximately 11 min) in standing and walking activities. Patients were not standing or walking at all whilst with a ST. Two-thirds of the time patients were together with a ST they were sitting out of bed. Most of the time patients spent with each other, they were sitting out of bed. When family and/or friends were present, patients spent one-third of the time in bed, but 9% of the time they were observed standing or walking.
Fig. 2. Proportion of the day in different activity levels divided by category of accompanying person(s). PT: physiotherapist; OT: occupational therapist; ST: speech therapist.
Time in social interaction
Fig. 3 shows time together with others. The categories are not mutually exclusive, e.g. a nurse and a PT might have attended a patient at the same time during the 1 min observation period. No different from in previous studies, patients were alone most of the time (52% of the day). The most frequent contact was with therapists (17%), other patients (16%) and nurses (13%). The most common combination of professionals was nurses and nurse assistants (2.2%), followed by combinations of nurses or nurse assistants and therapists (1.2%) or combinations of therapists (1.2%).
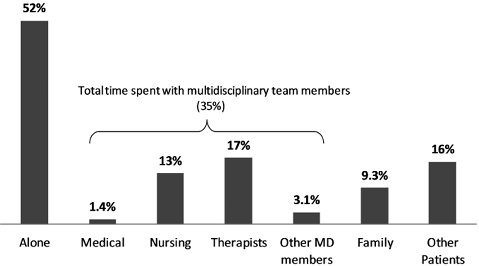
Fig. 3. Observed social interaction between 08.00 h and 17.00 h averaged across all cases. The categories are not mutually exclusive; e.g. a nurse and a physiotherapist might have attended a patient at the same time during the 1 min observation period. MD: Multidisciplinary Team.
Stroke severity and time in physical activity
Patients with moderate (NIHSS 8–16) and severe stroke (NIHSS > 16) at baseline, showed very similar activity patterns; over 40% of the day was spent in bed and less than 10% was spent in standing and walking. Comparable numbers for patients with mild stroke were 33% in bed and 18% in standing and walking. Patients with cognitive impairment (COG-4 > 0) on admission to acute hospital spent 39% of their time in bed and 11% in standing and walking. Comparable numbers for patients with no cognitive impairment were 36% and 17%, respectively.
Factors associated with high levels of physical activity
Median regression revealed that the median percentage of time spent in high levels of physical activity (AC4) was associated with gait independence and age (Table II). Adjusted for stroke severity (NIHSS) and cognition (COG-4), gait independence increased the median percentage of time spent in high levels of physical activity by 16.5%, while an increase of 1 year of age reduced the median percentage of time spent in high levels of physical activity by 0.16%.
|
Table II. Summary statistics and parameter estimates with median regression for time spent in standing and walking (high physical activity; AC4) |
|||
|
Variable |
Q1– Q3 |
Median |
Mean (SD) |
|
AC4 (%) |
2.00–18.00 |
8.00 |
12.55 (13.39) |
|
Coefficient |
p-value |
95% CI |
|
|
Age |
–0.16 |
0.041 |
–0.31 to –0.01 |
|
Gait independence |
16.51 |
< 0.0001 |
12.07 to 20.94 |
|
NIHSS score |
–0.24 |
0.259 |
–0.65 to 0.18 |
|
COG–4 |
0.26 |
0.688 |
–1.02 to 1.53 |
|
Constant |
16.52 |
0.009 |
4.28 to 28.77 |
|
CI: confidence interval. |
|||
Discussion
This is the first study to present data on patients’ levels of physical activity and social interaction in Swedish rehabilitation facilities. Our aim was to naturalistically map current stroke practice. In a study including 104 patients and over 5,000 observations, essential baseline information was obtained about patients’ physical activity and social patterns in stroke rehabilitation settings in Sweden.
It was found that, during the time patients were alone (52%), they spent the majority of time in bed (59%). A current systematic review (10) of activity in hospitalized stroke patients reported an exceptionally broad range (24–98%) of time spent in bed; however, this was in total throughout the day, and data on different activity levels whilst alone or with others were not available. An important conclusion from the review was that the description and classification of physical activity post-stroke need to be improved in order to increase the accuracy when comparing data across studies, since marked variation in definitions currently hampers the synthesis of results (10). The difficulties in comparing data became evident when we tried to compare our data with the most relevant individual study, the observational study of 4 rehabilitation centres in 4 European countries (Belgium, Switzerland, UK and Germany) (12). It is, however, important to recognize that not all therapy time is active; a current review found that 40% of the time patients spent in a PT session they were inactive (i.e. absolute rest or inactive time) (26). Furthermore, in the study by De Wit et al. (12), nursing and medical care was classified as therapy time. We found in our study that more than 80% of the time patients were observed with doctors and nurses they were lying or sitting, consequently such care is likely to occur with little physical activity for the patient. Moreover, time classified as leisure activities, such as reading a book (12), is likely to take place while lying in bed. Nevertheless, the truly important unanswered question here is whether being inactive in bed is at all beneficial to patients’ recovery.
The result that patients were alone for more than half of the time is in line with previous findings (10, 12), except from one study (15) where less time alone was reported. In this study patients spent more time in group therapy, and as a result spent more time with therapists and other patients. In our study patients spent 17% of the observed day interacting with therapists. This is similar to the findings by De Wit et al. (12), with the exception of the UK centre, where considerably less therapist contact was observed. However, in the European registers of stroke study acute stroke patients in 4 different centres (UK, France Lithuania and Russia) spent only 1–6% of the day with therapists (27). We found contact with other patients to be higher than in many other studies (10, 12) with only 1 exception (15). In addition, 13% contact time with nurses is higher than reported in other studies (12, 15). It is important to note, however, that our categories were not mutually exclusive.
Patients were more frequently engaged in walking and standing activities when PTs or OTs were present; however, they were most active with a PT. This was not surprising; according to the review (10) a greater proportion of time appeared to be spent in moderate to high levels of physical activity during physiotherapy sessions compared with occupational therapy sessions. Another study (28) comparing the content of physiotherapy and occupational therapy sessions found that ambulatory exercises, transfers and exercises in standing occurred significantly more often in physiotherapy sessions. Taking into account that most speech therapy sessions take place in a sitting position, it was not surprising that patients were not observed standing or walking while with a ST. This is not to say that activities with OTs or STs are less therapeutic (directed at improving recovery of an impairment or functional deficit), but simply that patients are less physically active during these interactions.
Previous research (29) indicates that more efficient use of human resources, rather than more staff per se, can result in higher amounts of therapy. Increased demand for therapists’ time in non-patient activities, such as meetings, documentation or registration tasks, should, even when beneficial per se, always be questioned, as therapist’s time is a scarce resource. Suggested ways to increase therapy time efficiency noted in the systematic review (10) included: increasing self-directed patient activity, modifying the environment to promote activity, and increasing time in group therapy. In our study, group therapy was rare; only observed in 1 of the 4 centres and not necessarily undertaken by a therapist. Although group therapy has been reported to stimulate interaction (12), the lack of it is in line with a previous study (30), showing that, on average, 9 out of 10 PT and OT sessions are carried out as 1-to-1 sessions. A current review (31) by the Cochrane Collaboration group found that circuit class therapy (e.g. supervised training in groups for people after stroke) was safe, time efficient, and resulted in improved mobility for people after moderate stroke. Although formal therapy time could be increased, with more time allocated for group therapy, the proportion of time spent in high level activity (walking) has been reported to be lower in group therapy than in individual sessions (32). However, this was for group therapy carried out in a large group of patients. In view of this, one suggestion is to carry out some sessions in very small groups; for example 2 patients with similar ability in walking and climbing stairs; 1 patient is resting and observing, while the other patient is active carrying out the task. This may be one strategy to increase the time patients spend in physical activity without decreasing the level of physical activity. Interestingly, there is indication that action observation (e.g. observing other individuals performing a congruent task) together with physical training can improve motor rehabilitation in patients with chronic stroke (33). To further investigate the potential benefit on action observation on patients in an earlier stage of stroke is tempting. This also raises the question as to whether patients could benefit from 1 to many sessions in a larger extent, as well as the importance of patient education and counselling.
It was, therefore, not surprising to find that time spent in high level physical activity was associated with gait independence and age; a previous study (14) investigating acute stroke found that patients with high levels of physical activity were more likely to be younger and have a high walking score (14). In addition, a current systematic review (34) identified age as a potential baseline factor in predicting mobility in the short term (within 30 days post-stroke; patients in our study had a median observation day of 19 days post-stroke). Higher NIHSS scores (more severe stroke) on admission has, in a previous study (14), been associated with low levels of physical activity in acute stroke. Moreover, a current prospective cohort study (35) reported that the admission NIHSS score was the best predictor of walking ability at discharge from hospital. However, in our study NIHSS scores were not strongly associated with level of physical activity. It is also possible to argue that patients with less independence, based on their need, should get more therapy, which seems not to be the case in our, or other previous, studies. Instead, activity levels seem to correlate with ability to move and walk. One explanation for this may be that, in our study, stroke severity (NIHSS) was assessed on admission to acute hospital and not on the day of observation (i.e. stroke patients may have made significant recovery from baseline severity levels). We did find that patients who were able to walk independently on the day of observation had higher levels of physical activity across the day. However, spending 16% (86 min) more of the day in high-level activity when you are able to walk independently raises the question as to whether further enriching the environment (i.e. providing a more stimulating environment) could promote greater physical activity for these patients. A pilot study (36) investigating the feasibility in stroke rehabilitation of using environmental enrichment (e.g. providing recreational activity groups and communal enrichment areas with reading material, jigsaw puzzles, computers, board games, music, etc.) is currently underway.
There is a lack of evidence for stating optimal activity levels in stroke rehabilitation and the largest value of our study may be establishing a baseline for further optimum-seeking studies. Although there is a large evidence base for a linear dose–response relationship of physical activity and health benefits in the general population, even the common recommendation of 30 min medium-intensity activity for 5 days a week lacks evidence for being an optimum or enough for improving function. Most likely the need for re-gaining function after stroke would require activity levels beyond these, but currently our activity level recommendations must be based on guesswork. We can be fairly sure that more is needed, but not how much more. Therefore, optimum-finding studies are extremely important, both from a patient and a health economic perspective.
Some limitations to our study should be acknowledged. Any observational study has the potential for bias as the attendance of an observer may modify personnel or patient behaviour. Nevertheless, if this was the case we have probably overestimated activity levels as we would assume increased activity in response to the observer’s presence. During 6% of observations we were unable to ascertain activity. However, the missing activity is spread across all the different categories of people present, suggesting no particular bias. We cannot be sure that our results reflect the rest of Sweden; however, studying 4 rehabilitation centres in the same country increases generalization to that country compared with a single-centre study. Our results should also be regarded in the context that we studied the patients during the times that they are presumably most active, and that including activity levels during evenings and weekends would probably show far less active patients.
In conclusion, patients in stroke rehabilitation had low levels of physical activity and social interaction, and they were rarely involved in group therapy. In view of these findings and considering previous research indicating that a higher amount of therapy is not only attributable to more staff, but also to better use of human resources, we believe that there is a huge potential to increase patients’ activity levels and augment social interaction above current levels, even in patients with decreased levels of consciousness, cognitive and physical function. Evidence for establishing optimal dosing and timing of physical activity are still lacking. Testing interventions that enrich the environment and promote activity in stroke rehabilitation is therefore highly warranted.
Acknowledgements
This study was supported by grants from the Västra Götaland and Göteborg/Södra Bohuslän R&D offices, the Ahrenberg Foundation, the Edit Jacobsson Foundation, the Per Olof Ahl Foundation for Cerebrovascular Diseases Research and the Foundation for Neurological Research in Gothenburg. The authors would like to thank Gunilla Bergkvist, Margareta Costinger, Magnus Fogelberg, Eva Friberg, Geir Gudjonsson, Elisabeth Johansson, Irene Johansson, Merja Henning, Jenny Murdvee, Carina Petersson and Ulrika Rhodin and members of the staff in the stroke rehabilitation units who supported in the performance of this study. Leonid Churilov, Li Chun Quang, Jan Chamberlain and Janice Collier at the Stroke Division, Florey Neuroscience Institutes helped with data handling and statistical analyses. Dr Bernhardt was supported by Australian Research Council Future Fellowship and the Operational Infrastructure support of the State Government of Victoria.
References
