OBJECTIVE: To investigate whether muscle strength is selectively more affected at shorter lengths of thigh muscles as result of stroke and to determine whether this is associated with impaired neural activation at shorter muscle lengths.
DESIGN: Case-control study.
SUBJECTS: Fourteen patients with sub-acute stroke, and 12 able-bodied controls.
METHODS: In patients (bilaterally) and able-bodied controls torque and activation was measured during maximal voluntary isometric contractions of knee extensors and flexors at 30°, 60° and 90° knee flexion.
RESULTS: The paretic knee extensors showed lower normalized maximal torques (73%) and lower normalized activation (71%) compared with control at 30°. The paretic knee flexors showed lower normalized maximal torques at 60° (64%) and 90° (45%) with non-significantly lower normalized activation (~ 79% and ~ 67%, respectively).
CONCLUSION: As a result of stroke, lower muscle torque at shorter muscle lengths of the knee extensors is associated with a length-dependent lower voluntary activation, which may also be the case for the flexor muscles, but not with altered co-activation. We recommend investigating the role of specific training of paretic knee extensors and flexors in especially shortened positions to improve recovery of function after stroke.
Key words: cerebrovascular accident, muscle weakness, electromyography.
J Rehabil Med 2009; 41: 317–321
Correspondence address: Astrid Horstman, Faculty of Human Movement Sciences, VU University Amsterdam, Van der Boechorststraat 9, NL-1081 BT Amsterdam, The Netherlands. E-mail: a.horstman@fbw.vu.nl
Submitted May 29, 2008; accepted October 31, 2008
INTRODUCTION
The majority of people after stroke have motor impairments, e.g. hemiparesis, muscle weakness of the upper and lower limbs contralateral to the brain lesion. This causes problems in functional tasks, such as balancing, climbing stairs, walking and transfers. Therefore, the severity of strength loss after stroke is an important consideration in regaining locomotor skills and improving functional performance during rehabilitation (1).
In addition to a reduction in muscle strength, length-dependency of strength is an important feature, since a whole range of muscle lengths is used during daily activities. This length-dependency of force generation is reflected in the torque-angle relationship as found in healthy humans (2). In patients with stroke, muscle weakness is present to a greater extent at relatively short muscle lengths for elbow extensors and flexors (3–5). However, the mechanisms responsible for the impaired strength at short muscle length after stroke are presently unknown. Ada et al. (3) excluded contracture as a major contributing factor to selective weakness at short elbow muscle lengths. Apart from possible changes in biomechanical properties of muscles, Koo et al. (4) showed for arm muscles that a reduced central drive to the agonist muscles could be responsible for the lower torque at shorter muscle lengths. According to Ada et al. (6), a differential effect of muscle length on strength after stroke may contribute to the problem of poorer muscle function at shorter muscle lengths rather than loss of control.
To the best of our knowledge, the length-dependency of torque of knee flexors and extensors of individuals after stroke has not been investigated previously. In fact, the consequences of this length-dependent weakness could be much larger in the lower limb muscles compared with, for example, elbow muscles, since it is impossible to walk with one leg. Besides, in the upper limbs, more degrees of freedom are available, so the unfavourable lengths could be avoided or the other arm could be used for certain movements. Except for some compensation strategies, this is impossible with the lower limb muscles during locomotion. Moreover, it is known that after stroke individuals have difficulties functioning when the affected knee is in the last 0–15° of extension (7). This possible length-dependent muscle weakness could explain the clinical observation of an inability to stand with a straight knee (short muscle length of knee extensors) despite the ability to bear weight when the knee is slightly flexed (longer knee extensor muscles) (6). The quadriceps muscles, and the hamstrings during the swing phase of gait, are very important for locomotion. The knowledge that force is more impaired after stroke at a certain thigh muscle length and, moreover, which mechanisms are responsible for this, may help to improve rehabilitation after stroke. The aim of the present study was therefore to investigate whether muscle strength is impaired at particular lengths of the thigh muscles as result of stroke. We hypothesized that patients with stroke show selective muscle weakness at relatively short lengths of knee extensor and flexor muscles of the contralateral paretic lower limb. Because the ipsilateral non-paretic limb also shows muscle weakness (1, 8, 9) it may not be valid as control. Therefore, we assessed muscle strength in both lower limbs at different knee angles and compared this with able-bodied control subjects. Furthermore, we measured muscle activity from both knee extensor and flexor muscles to determine whether impaired function was associated with neural activation at knee angles in which muscles were at a short length.
METHODS
Subjects
Fourteen patients after their first-ever stroke (lesion side: 6 left, 8 right; type of stroke: 5 haemorrhagic, 9 ischaemic; sex: 4 women, 10 men) with a minimum age of 18 years and hemiparesis of the lower extremity (mean age 55.9 years (standard deviation (SD) 10.4), height 1.74 m (SD 0.10) and weight 74.9 kg (SD 14.3)) and 12 able-bodied control subjects (mean age 58.1 years (SD 12.2), height 1.76 m (SD 0.06) and weight 75.7 kg (SD 11.9), 5 women) volunteered to join this study. The patients entered the study on average 3.5 months after stroke and 2 months after admission to the rehabilitation centre. They were all inpatient and all (except one) ambulatory. The median and interquartiles on the Functional Ambulation Categories (FAC)-score were 4 (2.25–4), evaluating the measure of independence of walking of the patient and categories are scored on a 6-point scale (0–5) (10, 11) in our study. The exclusion criteria were: medical complications; severe cognitive and/or communicative problems preventing the ability to follow verbal instructions or limiting the ability to perform the requested tasks, e.g. aphasia, hemi-neglect; and contra-indications for electrical stimulation, such as unstable epilepsy, cancer, skin abnormalities, or pacemaker.
The project was approved by the institutional review board of the VU University Medical Centre, Amsterdam, The Netherlands, and all subjects signed an informed consent before participation.
Experimental design
Maximal voluntary isometric knee extensions and flexions in 30°, 60° and 90° knee angles were performed. The studies were spread over 2 days; at day one the familiarization session was performed and the other day the real measurements took place, with at least one day of rest in between.
Torque measurements
Maximal voluntary isometric forces of the knee extensors and knee flexors at different knee angles were measured on a custom-built Lower EXtremity System (LEXS). Subjects were placed in a supine position and were tilted to the measuring position: back 10° backwards from upright position and a hip-angle of 100° (180° being fully extended hips). The position of LEXS was adjusted, subjects were stabilized with a hip and trunk belt to avoid changes in hip and knee angle during isometric contractions and the external moment arm was measured as described in Horstman et al. (1). Briefly, the lower leg was strapped tightly to a force transducer (KAP, E/200Hz, Bienfait B.V. Haarlem, The Netherlands, range: 0–2 kN) just above the ankle by means of a cuff and active knee angle was determined with a handheld goniometer (model G300, Whitehall Manufacturing, California, USA). The 3 different knee angles were 30°, 60° and 90° (0° corresponds to a fully extended knee).
Electromyographic measurements
To assess the potential effect of neural activation on the torque-angle relationship we used electromyography (EMG) on both extensor and flexor muscles. For these EMG measurements (Biotel 99, Almere, The Netherlands) the skin was shaved and cleaned with alcohol. Subsequently, surface (Ag-AgCl) electrodes were placed in a bipolar figuration, consistent with the muscle’s overall direction of pull, with a centre to centre inter-electrode distance of 25 mm on the muscle bellies of the vastus lateralis muscle (VL), rectus femoris muscle (RF), vastus medialis muscle (VM), biceps femoris muscle (BF) and gastrocnemius medialis muscle (GM). One reference electrode was placed on the patella of the corresponding lower limb.
Experimental procedures
Familiarization session. The measurements of the familiarization session were performed with the non-paretic lower limb (NL), in order to check whether the subjects understood the instructions. After a warm-up (comprising 5 submaximal contractions) subjects were trained to perform maximal isometric knee flexion and extension contractions.
Muscle torque. Subjects started with their paretic lower limb (PL) and were asked to maximally generate isometric knee extensions for 3–4 seconds to determine maximal voluntary knee extension torque (MVCe). Two to 4 attempts were allowed, separated by 2 min of rest. MVC was taken as the highest value of these attempts, which did not exceed preceding attempts by > 10%, allowing a maximum of 4 attempts. Real-time force production was visible on a computer screen. Subjects were vigorously encouraged to exceed their maximal value, which was also displayed to confirm the subject’s achievement throughout the test. Alternately, MVCs with the knee flexors (MVCf) were performed. For this purpose, the lower limb was fixed tightly by a top restraining bar that was secured on the thigh, just proximal to the knee joint to minimize the movement of the leg and to avoid changes in hip and knee angles during isometric flexion contractions (1). In addition, an ankle brace was placed to keep the ankle in plantar flexion position, in order to reduce the contribution of the gastrocnemius medialis muscle to the knee flexion.
Muscle activity was measured by means of EMG, as described above (see Torque measurements). Measurements always started at 60° knee angle, at which angle the highest torque is usually produced (12). Subsequently, measurements were repeated at 30° and 90° knee angles, which were randomly assigned. At the end of the session, one or 2 maximal attempts were carried out again at 60° to assure that fatigue, which could have influenced the maximal force values at the previous knee angle as well, had not occurred.
The measurements performed on the PL were repeated with the NL, carried out on a separate day. Control subjects only performed 1 of these sessions, with the right lower limb.
Data analysis
Real-time force applied to the force transducer was digitally stored (1 kHz) on computer disk. The force signals were automatically corrected for gravity of the leg; the average force applied to the transducer during the first 50 ms after the start of the recording, was set to zero force by the computer program. All force signals were low-pass filtered (4th order Butterworth, 50Hz). MVC torque (Nm) was defined as the peak force from the stable part of the force plateau multiplied by the external moment arm. Overall, all torques (and accompanying EMG signals) higher than 0.3 Nm are included.
To assess the potential effect of neural activation on the torque-angle relationship we obtained EMG from both extensor and flexor muscles during MVCfs and MVCes at the 3 knee angles. EMG signals of the voluntary maximal knee extensions and flexions were amplified (×100), digitized (1 kHz) and stored with the synchronized force signal on computer disk. All EMG signals were band-pass filtered (10–400 Hz) and rectified. Rectified surface EMG amplitude (rsEMG) was calculated for the RF, VL, VM, BF and GM for 1000 ms of the plateau of the force signal. rsEMG signals served to assess activation levels of the knee extensors (summed rsEMG of VL, VM and RF) and flexors (summed rsEMG of BF and GM) during MVCs at the different knee angles. Moreover, rsEMG signals served to determine the co-activation of the thigh muscles during those MVCs, to check whether an increased neural drive to antagonist muscles causes reduced net torques. To determine whether activation was selectively more affected after a stroke at different muscle lengths, absolute rsEMG was normalized to maximum rsEMG at the angle with the highest torque, making the results independent of absolute rsEMG-values. To evaluate the level of co-activation, rsEMG of the antagonist muscles was normalized to the maximum rsEMG during its primary function (working as an agonist). For instance, co-activation during flexion was defined as the ratio of rsEMG flexors during extension divided by maximal rsEMG flexors during maximal flexion (rsEMG/rsEMGmax) (13).
Statistics
All results are presented as means and SD. Repeated measures analysis of variance (ANOVA) were used (group × knee angle) for the comparison of normalized MVCe and MVCf, agonist and antagonist rsEMG in the 30°, 60° and 90° knee angle and between NL, PL and control lower limb. In case of significance, Bonferroni post-hoc tests were used. In each statistical analysis, the level of significance was set at p < 0.05. For our main hypothesis a sample size calculation was performed and the design, given a large effect size (Δ2 = 0.9) and a power of 70% would result in 13 participants per group to be able to show a significant, relevant difference within angles between groups. A post-hoc power analysis showed a power of 99%.
RESULTS
Data were not complete for some patients due to discomfort or unreliable data, e.g. concentration problems, one subject dozed off a few times during the measurements. These were excluded from the data. In addition, one of the 14 patients with stroke was not able to flex the non-paretic lower limb in any of the angles. One subject could not perform a knee extension with the non-paretic lower limb in 30°. With the paretic lower limb, 7 subjects were unable to perform a knee flexion in all angles and were therefore not included. One subject could not perform a knee flexion with the paretic leg in the 90° knee angle only. The number of patients analysed are indicated in the text or in the figure legends.
Torque-angle relationship
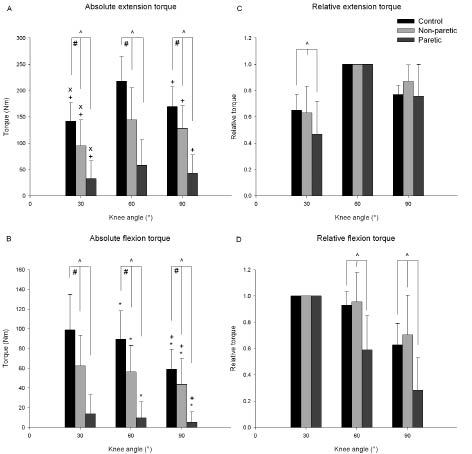
For both knee extensors and flexors, significant main effects for group (p = 0.000, p = 0.000) and angle (p = 0.000, p = 0.000) were found (Fig. 1A and B). More important, significant interaction effects (angle × group) were found for the absolute maximal voluntary torque of the knee extensors (p = 0.000) and knee flexors (p = 0.002). Thus, for both extensors and flexors a significant difference is found in the angle-torque relationships between groups. To compare angle effects among groups independent of absolute torque we normalized torques for torque at the “optimum” knee angle (i.e. angle at which torque was highest) (Fig. 1C and D). Most importantly, a main effect for group was found (p = 0.001): at 30° knee angle (at short muscle length) normalized knee extensor torque in PL (n = 12) was significantly lower than in NL (n = 13) and control (n = 12) (Fig. 1C). NL did not differ significantly from control. Also for knee flexors, at shorter muscle lengths (60° and 90° knee angle), normalized torque in PL (n = 7) was significantly lower than in NL (n = 13) and control (n = 12) (Fig. 1D main effect group, p = 0.001). NL again did not differ significantly from control.
Fig. 1. Left: absolute maximal voluntary torques of controls (n = 12), non-paretic lower limb (n = 13) and paretic lower limb (n = 12 for extension and n = 7 for flexion) at 3 different knee angles (30°, 60° and 90°) for (A) knee extensors and (B) knee flexors. An interaction-effect (knee angle × group) was found. Right: normalized (C) knee extensor and (D) knee flexor torque at the 3 knee angles for the 3 groups. ^Lower than non-paretic lower limb and control; #lower than control; *: lower than 30°; +: lower than 60°; ×: lower than 90°.
Thus, lower normalized torques at shorter muscle lengths were found for both knee extensors (73% of control at 30°) and knee flexors (64% and 45% of control at 60° and 90°, respectively) in PL compared with control. No significant differences were found between NL and control.
Agonists muscle activity
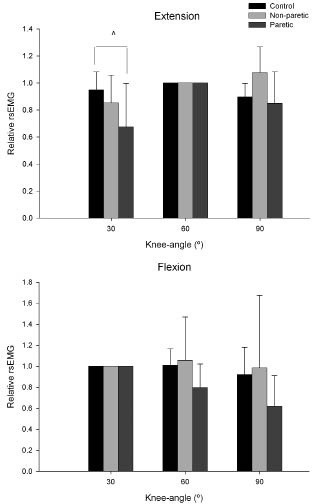
For absolute rsEMG of the knee extensors (sum of VL, VM and RF) during extension an interaction effect (angle × group) was found (p = 0.05), while for absolute rsEMG of the knee flexors (sum of BF and GM) during knee flexion no significant interaction effect was found (p = 0.876). Thus, the angle-rsEMG relation of the knee extensors is different among groups, whereas no differences were found for knee flexors.
Normalized knee extensor rsEMG values (Fig. 2, upper) were significantly lower in PL than in controls (~71%) at 30° knee angle (p = 0.006), indicating lower activation at short muscle lengths compared with controls. Mean values for the knee flexors muscles in PL (Fig. 2, lower) at 60° and 90° were 79% and 67% of control, respectively, but were not significantly different from control (p = 0.226). No significant differences between control and NL were found.
Co-activation
No interaction effects (angle × group) were found for co-activation values during extension (p = 0.419) or flexion (p = 0.052); hence, no differences were found among PL, NL and control in the different angles (rsEMG/rsEMGmax). So, the level of co-activation was not different between PL, NL and controls and this was not different among knee angles.
DISCUSSION
The most important finding of the present study was the selectively impaired muscle function at shorter muscle lengths of both knee extensors and flexors of the paretic lower limb, but not the non-paretic lower limb of individuals after stroke. For the knee extensors, this may be attributed to a length-dependent lower voluntary activation capacity, which may also be the case for the flexor muscles. Co-activation did not seem to play a significant role herein.
Normalized angle-torque relationship
After normalization, lower torques at shorter muscle lengths were found for both knee extensors (73% of control at 30°) and knee flexors (64% and 45% of control at 60° and 90°, respectively) in PL compared with control. Bohannon (14) found lower knee flexion torque in supine position (shorter knee flexor muscle length) compared with sitting position. The sitting-to-supine ratio was 0.56 for the non-paretic lower limb and 0.70 for the paretic side, but this difference was not significant.
As stated previously, in the present study patients with stroke seem to show relative muscle weakness of knee extensors at more extended knee angle (shorter knee extensor muscle lengths) of the paretic lower limb. This is in agreement with Koo et al. (4), who found smaller extension torques of the upper limb at shorter muscle lengths in patients with stroke compared with controls. They also found slightly smaller maximal flexion torques in the more flexed positions (shorter muscle lengths) for the paretic compared with the control (upper) limb. In addition, the non-paretic lower limb was weaker than control, but no length-dependent differences in extensor and flexor strength were observed.
Activation
An impaired drive to voluntarily activate the quadriceps muscles in patients with stroke (1, 8, 9) might be a cause of the length-dependent weakness after stroke. Indeed, our results (Fig. 2) show that normalized knee extensor rsEMG in PL was significantly lower compared with controls (~71%) at 30° knee angle, which corresponds with the 73% lower torque. Although not significantly different, mean values for the PL knee flexors muscles at 60° and 90° were 79% and 67% of control, respectively, which may explain the torque decreases of 64% and 45%. Note that our patients have a wide range of motor impairments from very severe to very mild, based on the scores at tests of functional performance (1). This leads to a large variation within the subject group, which may explain the lack of significance. Another reason for the lower torque at shorter muscle length could be more co-activation. Despite a tendency during flexion we did not find significant differences in co-activation between groups during the maximal voluntary contractions in isometric conditions. However, during (submaximal) dynamic contractions, this may be different.
Fig. 2. Normalized rectified surface electromyography (rsEMG) at the 3 knee angles (30°, 60° and 90°) for the 3 groups (control (n = 12), non-paretic lower limb (n = 13) and paretic lower limb (n = 12 for extension and n = 7 for flexion) for knee extensors (upper graph) and knee flexors (lower graph). ^Lower than control.
Thus, the voluntary activation capacity is more impaired at shorter lengths of paretic knee extensor muscle compared with control, which may also be the case for the flexor muscles. This is in agreement with Koo et al. (4), who found smaller elbow extension torques at shorter muscle lengths in patients with stroke compared with controls, also accompanied by a reduced central drive (rsEMG) to agonist muscles at extended positions. In addition, Koo et al. (4) found that, during maximal flexion contraction, the rsEMG of patients with stroke tended to be lower than that of controls, as the elbow joint was flexed beyond 90° (short muscle lengths). During submaximal contractions, Tang & Rymer (15) found more EMG per force level in shortened paretic elbow flexors. The authors conclude that this did not result from co-activation, but most likely from a reduction in mean motor unit discharge rate.
A possible explanation for the disproportionate reduction in muscle strength at short lengths might be a differentially affected (impaired) excitability – muscle length relationship in patients with stroke, which corresponds with difficulties in producing higher motor unit firing rates required to produce fusion of twitches, and therefore maximum torque, at shorter muscle lengths after stroke (16). This decreased neural activation is a primary result of the stroke and explains why already 3.5 months after stroke, muscle weakness at especially short lengths is found, in contrast to adaptations in intrinsic muscle properties, which are often used as an explanation in chronic patients with stroke with spastic upper limbs (4, 6, 17).
In conclusion, as result of stroke the lower muscle torque at shorter muscle lengths of the knee extensors is associated with a length-dependent lower voluntary activation capacity, which may also be the case for the flexor muscles. Co-activation does not seem to play a role in the length-dependency of muscle weakness after stroke. From these results, we recommend investigating the role of specific training of the knee flexors and extensors of the (paretic) lower limb, especially in the shortened position, to improve the recovery of function after stroke. Hence, it may be important not only to strengthen muscles after stroke, but to strengthen them at shortened lengths in order to counterbalance the impaired voluntary activation at those muscle lengths.
REFERENCES
1. Horstman AM, Beltman MJ, Gerrits KH, Koppe P, Janssen TW, Elich P, et al. Intrinsic muscle strength and voluntary activation of both lower limbs and functional performance after stroke. Clin Physiol Funct Imaging 2008; 28: 251–261.
2. Gordon AM, Huxley AF, Julian FJ. The variation in isometric tension with sarcomere length in vertebrate muscle fibres. J Physiol 1966; 184: 170–192.
3. Ada L, Canning CG, Low SL. Stroke patients have selective muscle weakness in shortened range. Brain 2003; 126: 724–731.
4. Koo TK, Mak AF, Hung L, Dewald JP. Joint position dependence of weakness during maximum isometric voluntary contractions in subjects with hemiparesis. Arch Phys Med Rehabil 2003; 84: 1380–1386.
5. Levin MF, Selles RW, Verheul MH, Meijer OG. Deficits in the coordination of agonist and antagonist muscles in stroke patients: implications for normal motor control. Brain Res 2000; 853: 352–369.
6. Ada L, Canning C, Dwyer T. Effect of muscle length on strength and dexterity after stroke. Clin Rehabil 2000; 14: 55–61.
7. Carr JH, Shepherd RB, editors. Neurological rehabilitation: optimizing motor performance. Oxford: Butterworth-Heinemann; 1998.
8. Newham DJ, Hsiao SF. Knee muscle isometric strength, voluntary activation and antagonist co-contraction in the first six months after stroke. Disabil Rehabil 2001; 23: 379–386.
9. Harris ML, Polkey MI, Bath PM, Moxham J. Quadriceps muscle weakness following acute hemiplegic stroke. Clin Rehabil 2001; 15: 274–281.
10. Holden MK, Gill KM, Magliozzi MR, Nathan J, Piehl-Baker L. Clinical gait assessment in the neurologically impaired. Reliability and meaningfulness. Phys Ther 1984; 64: 35–40.
11. Holden MK, Gill KM, Magliozzi MR. Gait assessment for neurologically impaired patients. Standards for outcome assessment. Phys Ther 1986; 66: 1530–1539.
12. de Ruiter CJ, Kooistra RD, Paalman MI, de Haan A. Initial phase of maximal voluntary and electrically stimulated knee extension torque development at different knee angles. J Appl Physiol 2004; 97: 1693–1701.
13. Macaluso A, Nimmo MA, Foster JE, Cockburn M, McMillan NC, De Vito G. Contractile muscle volume and agonist-antagonist coactivation account for differences in torque between young and older women. Muscle Nerve 2002; 25: 858–863.
14. Bohannon RW. Decreased isometric knee flexion torque with hip extension in hemiparetic patients. Phys Ther 1986; 66: 521–523.
15. Tang A, Rymer WZ. Abnormal force – EMG relations in paretic limbs of hemiparetic human subjects. J Neurol Neurosurg Psychiatry 1981; 44: 690–698.
16. Renner CI, Woldag H, Hummelsheim H. Central compensation at short muscle range is differentially affected in cortical versus subcortical strokes. Stroke 2006; 37: 2076–2080.
17. Hu XL, Tong KY, Hung LK. Firing properties of motor units during fatigue in subjects after stroke. J Electromyogr Kinesiol 2006; 16: 469–476.