OBJECTIVE: To analyse the effects of gait therapy for patients after acute stroke in a randomized controlled trial.
METHODS: Fifty-six patients with a mean of 8 days post-stroke participated in: (i) gait trainer exercise; (ii) walking training over ground; or (iii) conventional treatment. Patients in the gait trainer exercise and walking groups practiced gait for 15 sessions over 3 weeks and received additional physiotherapy. Functional Ambulatory Category and several secondary outcome measures assessing gait and mobility were administered before and after rehabilitation and at 6-month follow-up. Patients also evaluated their own effort.
RESULTS: Walking ability improved more with intensive walk training compared with conventional treatment; median Functional Ambulatory Category was zero in all patients at the start of the study, but it was 3 in both walk-training groups and 0.5 in the conventional treatment group at the end of the therapy. Median Functional Ambulatory Category was 4 in both walk-training groups and 2.5 in conventional treatment group at 6-month follow-up. Mean accomplished walking distance was not different between the gait trainer exercise and over ground walking groups. Borg scale indicated more effort in over ground walking. Secondary outcomes also indicated improvements.
CONCLUSION: Exercise therapy with walking training improved gait function irrespective of the method used, but the time and effort required to achieve the results favour the gait trainer exercise. Early intensive gait training resulted in better walking ability than did conventional treatment.
Key words: stroke, exercise therapy, rehabilitation, gait disorders, neurology, training, body-weight support.
J Rehabil Med 2009; 41: 166–173
Correspondence address: Sinikka H. Peurala, The Finnish Centre for Interdisciplinary Gerontology, Faculty of Sport and Health Sciences PO Box 35 (V374), University of Jyväskylä, FI-40014, Finland. E-mail: sinikka.peurala@sport.jyu.fi
Submitted July 28, 2008; accepted September 23, 2008
*Presented in part at the International Society for Posture and Gait, 14–18 July 2007, in Burlington, Vermont, USA.
INTRODUCTION
Despite improved management of patients in the acute phase after stroke, the majority of surviving patients are disabled (1). Recovery of walking function to obtain independence in daily life is one of the main goals of patients after stroke. Treadmill training with partial body weight support was introduced as a strategy for gait rehabilitation more than 10 years ago (for example, 2, 3); subsequently various gait rehabilitation devices have been shown to be effective in regaining and improving walking function in patients after stroke (4). The electromechanical “Gait Trainer” (GT) and the Driven Gait Orthosis (DGO) were the first such devices in this field (5, 6). Subsequently, more prototypes have been designed (7, 8). On the GT, the patient is supported with a harness and his or her feet are placed on motor-driven footplates. On the DGO, the patient moves on the treadmill operated according to a pre-programmed physiological gait pattern and an exoskeleton-type robot. A Cochrane Review of electromechanical-assisted training for walking after stroke reported that the use of the GT and DGO could reduce dependency in walking by 25% (4). The authors concluded that further research is needed to determine what frequency or duration of training might be most effective, and at what time to begin training after stroke and to find out how long the benefit of training persists.
In patients with stroke, randomized controlled trials comparing gait rehabilitation devices with other physiotherapy methods have been conducted in the subacute or chronic stage (9–12). In our previous study (9), 45 patients, all more than 6 months post-stroke, practiced walking for 20 min, either on the gait trainer, on the gait trainer with functional electrical stimulation (FES), or on level ground daily. In Hong Kong (10), the same amount of walking exercises were provided for 3 similar groups for a period of 4 weeks (these patients were on average 2.7 weeks post-stroke). In patients with chronic stroke (9) no group differences were observed in improvements of gait speed and endurance, balance or mobility, whereas in the subacute stage (10) the patients who trained on the gait trainer, with or without FES, gained faster gait, better mobility, and greater improvement in functional ambulation than the patients who practiced on level ground.
In the Deutsche Gangtrainer Studie, 155 patients (on average one month post-stroke) participated either in 20 min of walking exercises on the gait trainer followed by 25 min physiotherapy (group A) or 45 min physiotherapy (group B) for 4 weeks (11). In group A, patients practiced walking and mounting stairs on average for 34 min out of each 45 min session. In group B, patients practiced walking and mounting stairs on average for 20 min out of each 45 min session. Patients in group A performed more walking exercise and in outcome had better gait ability and better daily living competence.
The purpose of the present study was to evaluate gait ability and gait rehabilitation very early after stroke, in 3 groups. Our rehabilitation groups were: body-weight-supported exercise on the gait trainer (GT); walking exercise over ground (WALK); and conventional treatment (CT). To our knowledge, no randomized controlled trials in which rehabilitation has started this early after stroke (less than 10 days after stroke onset) have been published in the peer review literature.
METHODS
Subjects
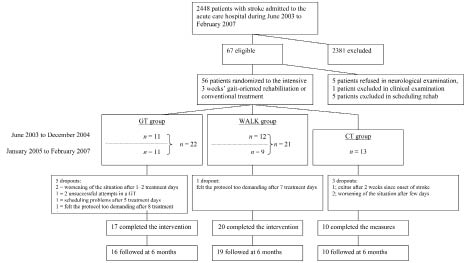
A total of 56 patients early after stroke participated in this trial (see Fig. 1). The criteria for patient selection were: (i) first supratentorial stroke or no significant disturbance from an earlier stroke (Modified Ranking Scale 0–2); (ii) Functional Ambulatory Category (FAC) 0–3 (13); (iii) voluntary movement in the leg of the affected side; (iv) Barthel Index (BI) 25–75 points (14); (v) age 18–85 years; (vi) no unstable cardiovascular disease; (vii) body mass index (BMI) < 32; (viii) no severe malposition of joints; and (ix) no severe cognitive or communicative disorders. All patients were initially diagnosed by magnetic resonance imaging or computerized tomography. Patients who fulfilled the criteria in a neurological examination within 10 days of stroke onset and who provided informed consent between June 2003 and December 2004 were randomly allocated to 2 walking exercise groups. They received a 3-week period of intensive inpatient rehabilitation in the acute care hospital. Between January 2005 and February 2007 patients fulfilling the criteria and providing informed consent were randomly allocated to 3 groups; either of the walking exercise groups or a conventional treatment group. For the total duration of the study the envelopes indicating the groups were sealed separately for patients with FAC 0 or 1 and with FAC 2 or 3. Stratified allocation was performed by an independent person who was not otherwise involved with the patients. The study was approved by the Hospital District of Northern Savo Research Ethics Committee (#148/2001, updated February 2005).
Neurologists used the Scandinavian Stroke Scale (SSS) (15) and the BI to assess the functional status of the patients with acute stroke. The SSS contains items on consciousness, orientation, eye movements, facial palsy, motor function of the arm, hand and leg, gait and speech. Each item is scored from 0 to 12 and the maximum score is 48 (see Table I for patient characteristics). The BI scale contains items on feeding, moving to and from a wheelchair, personal toileting, bathing oneself, walking on a level surface, ascending and descending stairs, dressing, and bowel and bladder control. Each item is scored from 0 to 15 and the maximum score is 100. Position sense was measured with the patient supine and the observer moving the paretic ankle in different directions. The patient repeated the movements with the unaffected leg. Thirty-eight of the patients had normal position sense in the ankle on the paretic side. Thirty-nine of the patients were not able to walk or needed 2 assistants to help them walk (FAC = 0) (13). Eight patients needed the constant attention of one assistant in walking (FAC = 1). Five patients needed someone for balance support (FAC = 2), and 4 patients needed to have someone walking beside them to give them confidence (FAC = 3). None of the patients were in FAC 4 or 5 (independent, but in FAC 4 need help with stairs of on uneven ground). Unfortunately, one patient did not fulfil inclusion criteria within 10 days, her programme started 17 days post-stroke. One patient with BMI above the criteria limit (39.6) was accepted.
| Table I. Characteristics of patients in gait trainer (GT) group, walking (WALK) exercise group, conventional treatment (CT) group, and dropouts |
| | GT (n = 17) | GT dropouts (n = 5) | WALK (n = 20) | WALK dropouts (n = 1) | CT (n = 10) | CT dropouts (n = 3) | p* |
| Age, years, mean (SD) | 65.7 (9.2) | 73.1 (9.2) | 65.3 (9.9) | 81.6 | 69.5 (11.0) | 73.1 (9.2) | 0.52 |
| Post-stroke, days, mean (SD) | 8.6 (2.3) | 4.2 (3.4) | 7.8 (3.0) | 6 | 9.5 (1.9) | 6.0 (4.6) 0.21 | |
| BMI, kg/m2, mean (SD) | 26.8 (4.3)† | 26.0 (5.1) | 26.5 (3.0) | 20.3 | 30.0 (5.1) | 29.3 (2.7) | 0.07 |
| SSS, scores, mean (SD) | 36.8 (11.8) | 38.8 (8.7) | 37.0 (8.7) | 42 | 33.2 (9.8) | 32.7 (14.0) | 0.59 |
| BI, scores, mean (SD) | 45.5 (23.7) | 54.0 (23.6) | 44.5 (19.8) | 20 | 31.6 (13.6) | 28.3 (14.4) | 0.23 |
| Men/women, n | 8/9 | 3/2 | 11/9 | 1/0 | 5/5 | 1/2 | |
| Infarction/haemorrhage, n | 11/6 | 5/0 | 16/4 | 1/0 | 8/2 | 1/2 | |
| Left/right hemiparesis, n | 9/8 | 2/3 | 12/8 | 0/1 | 6/4 | 2/1 | |
| Aphasia, no/yes, n | 13/4 | 4/1 | 12/8 | 1/0 | 7/3 | 3/0 | |
| Neglect, no/yes, n | 13/4 | 4/1 | 17/3 | 1/0 | 8/2 | 2/1 | |
| Patients in FAC 0, n | 12 | 3 | 14 | 1 | 7 | 2 | 0.99‡ |
| Patients in FAC 1, n | 3 | 1 | 3 | NA | 1 | NA | |
| Patients in FAC 2, n | NA | 1 | 3 | NA | 1 | 1 | |
| Patients in FAC 3, n | 2 | NA | NA | NA | 1 | NA | |
| *p-values between GT, WALK, and CT groups obtained using one-way analysis of variance or Pearson χ2. p < 0.05 is considered significant. †One missing value. ‡FAC and group table. SD: standard deviation; BMI: body mass index; SSS: Scandinavian Stroke Scale; BI: Barthel Index; FAC: Functional Ambulatory Category 0–3 (0 = not able to walk or need 2 assistants, 1 = need constant attention of 1 assistant, 2 = need someone to support, 3 = need to have someone walking beside to give confidence); NA: not applicable. |
Intervention
The objective of our 3-week in-patient rehabilitation for acute patients was to enhance their motor abilities and help them regain their walking independence as soon as possible. For 3 weeks each patient spent a maximum of 1 h/day to obtain 20 min actual walking either in the electromechanical gait trainer (Gait Trainer, Reha-Stim, Berlin, Germany) or over ground. Each patient also received additional gait-oriented physiotherapy for 55 min daily.
In the GT, the patient was supported with a harness and his or her feet were placed on motor-driven footplates. The amount of body weight support (BWS) provided by the harness was chosen according to the patient's individual needs. The percentage of BWS was recorded (kg). In the WALK group, the patients practiced walking over ground with 1 or 2 physiotherapists, using their individual walking aids. The training in the GT and WALK groups was progressed by increasing the speed and decreasing the amount of BWS or manual guidance and reliance on walking aids (for more details see (16)). The patients in the CT group were most often transferred to a health centre after the first set of measurements. Thereafter, they visited the hospital on testing days. While in the health centre, the patients normally had 1 or 2 physiotherapy sessions daily, but not at the same intensity as in the GT and WALK groups. The content of physiotherapy in the CT group was determined according to individually set goals.
Assessments
Training assessments. The patients in the GT and WALK groups evaluated their effort using the Borg Rating of Perceived Exertion Scale (17) (from 6 to 20; 7 = very marginally strenuous, 19 = extremely strenuous). Ratings were recorded each time during the last minute of the 20 min walking exercise and during the last minute of the other physiotherapy sessions. Heart rate (HR) was recorded with a heart rate monitor (Polar®, Polar-electro Oy, Kempele, Finland). HR was monitored continuously during the 20 min walking exercise and the data for the last minute was recorded.
During the 20 min walking exercise on the gait trainer, walking speed, number of steps, distance, and amount of BWS were recorded. In the WALK group, walking duration, distance walked and the use of walking aids were recorded. We allowed each patient a daily maximum of 1 h to achieve the target of 20 min of real walking time. Unfortunately, we did not have a formal record of the time spent to achieve the 20 min of walking during 1 h in the walking training groups. After interviewing the physiotherapist and analysing her notes and walking training records, we estimated the time used, but no statistical analysis was relevant to perform.
Outcome assessments. The primary outcome to assess the efficacy of the 3-week physiotherapy programme was the FAC (13). In addition, we used secondary outcomes to assess walking and other motor abilities. In the 10-m walk test (10MWT) (18), patients were asked to walk as quickly as possible. In the 6-min walk test (6MWT) (19), patients were asked to walk back and forth for 6 min on a 30-m-long track as quickly as possible while pacing themselves so as to be able to complete the task. In both walking tests, patients were allowed to use an orthosis. Partial support was allowed. For example, a physiotherapist could hold the walking belt during the test, but not push forward or move the patient’s legs. The assessment of other motor abilities were performed with the Modified Motor Assessment Scale (MMAS) (20), Rivermead Motor Assessment Scale (RMA) (18), and Rivermead Mobility Index (RMI) (21). The MMAS assessments included 8 items each scored from 0 to 6, maximum 48. Two out of 3 sections of the RMA were used: gross motor function (RMA g) and lower limb function plus trunk control (RMA l&t). Performance of each item scored 1 point. The testing was discontinued if the patient was unable to perform 2 consecutive items. The 15-item RMI was completed in the same way, except that the RMI scores were obtained by asking the patient if she or he could perform the activity in question.
Patients were assessed at the start, at 2 weeks (not reported), at the end of the 3 weeks of rehabilitation, and at 6 months. During the first 1.5 years both the physiotherapist and an independent observer performed the outcome measures. After analysing the inter-rater reliability (ranging from good to excellent) of the measures, the physiotherapist or an independent observer alone performed the set of measurements.
Statistical analysis
Statistical analyses were performed with SPSS 14.0. The mean values of age, time since onset of stroke, BMI, SSS, and BI in the GT, WALK, and CT groups were compared using one-way analysis of variance (ANOVA) (Table I). At start the distribution of gender, diagnosis, side of hemiparesis, aphasia, neglect and FAC between the groups was compared using the Mann-Whitney U test or Kruskal–Wallis test. The means in walking exercise time, total distance (in m) covered during the 15 gait sessions, HR during exercise, and Borg Scale in GT and WALK groups were compared using the independent-samples t-test to assess the similarity of the GT and WALK groups. For the GT group, mean values for speed and amount of BWS on the gait trainer were also calculated.
The FAC changes before and after the treatment was analysed with the Friedman and Wilcoxon signed-rank test, and the groups were compared with Kruskal-Wallis test. Analysis of the repeated measures data, using both within and between factors, was used to evaluate the changes in secondary measures. Many CT group patients were unable to walk at the start of rehabilitation and thus no statistical comparisons could be made for their secondary walking test data. Secondary walking test comparisons were performed for the GT and WALK groups. For these 2 groups, the walking test data were restructured to take into account patients who were unable to walk at the start of rehabilitation. Data transformation was performed by calculating the relative difference between the walking parameters at the start and at the end of rehabilitation, using the following formula. The following example is from the 10MWT (18):
10 MWT relative difference (10MWTrf):
(mean 10MWT at the end – mean 10WMT at the start)
mean 10MWT at the end
Missing parameters at the starting point were then calculated as follows:
(100–10MWTrf) × mean 10MWT at the end
100
The relative differences were calculated separately for the GT and WALK groups in the 10MWT, but for the combined GT and WALK patients in the 6MWT. The reason for combining the patient groups in the latter test was the low number of parameters in each separate group. It was not necessary to reconstruct the secondary walking test data for the follow-up comparisons, because more patients performed the test at the end and at follow-up. However, we were still not able to perform statistical analysis on the secondary walking test data of the patients in the CT group. The results were considered significant if p < 0.05.
RESULTS
Fifty-six patients, all early after stroke, participated in the study, and 47 patients completed the rehabilitation period. Only one patient in each of the 2 walking training groups felt the protocol to be too demanding after 7–8 days of treatment and 7 patients dropped out for other reasons (Fig. 1). A total of 45 patients participated in the follow-up visits and performed the tests. The demographic characteristics of the patients in the GT, WALK, and CT groups were comparable (Table I, for age: F2,44 = 0.657, p = 0.523; for post-stroke time: F2,44 = 1.640, p = 0.206); for BMI: F2,43 = 2.806, p = 0.072; for SSS: F2,44 = 0.532, p = 0.591: for BI: F2,44 = 1.494, p = 0.236).
Fig. 1. Consort flow diagram. GT: walking exercise in a gait trainer; WALK: walking over ground; CT: control group.
Exercise intensity
Effective duration of gait training in the GT and WALK groups. The actual walking exercise time achieved was near the maximum of 300 min in both GT and WALK groups (Table II). However, only 9 of the 17 patients in the GT group were able to walk for the whole 20 min at the start of the study. In 7 patients, the first walking session in the gait trainer did not last 20 min despite 1 h of effort, the next 4–6 sessions were completed with 1–3 short breaks, and in the remaining 8–10 sessions, the patients were able to walk for the full 20 min. Overall, the patients in the GT group performed their 20-min walk in much less than 1 h, 9 of them in 20 min in every session, and another 7 in 20 min in most sessions. In the WALK group, none of the patients was able to walk for target 20 min at the beginning of rehabilitation. Eight patients accomplished in sessions 1–5 less than 20 min walking time despite 1 hour of effort. Seven patients were able to walk for 20 min in the last 5–7 sessions. Except for the last sessions of these 7 patients, a large number of breaks was needed (often more than 5 breaks, and in some cases up to 17 breaks) and often the whole 1 h period was needed to achieve 20 min walking.
Other parameters of gait training in the GT and WALK groups. The total distance covered during 15 gait sessions, the mean HR during the last minute of walking exercise, and the perception of exertion of the patients are presented in Table II. Also, the training parameters of the gait trainer are presented. In both gait training groups, 2 therapists were usually needed to assist patients in the beginning. In the gait trainer, one assistant was mainly needed when moving to the gait trainer or from it. Later days during the rehabilitation, manual guidance by one therapist was sufficient for most patients in both groups. In the WALK group, a walking belt was used by half of the patients during the walking exercises. When patients were able to increase their walking speed, a knee orthosis was often used to prevent over-extension of the knee. A peroneus orthosis or an elastic bandage was used in 16/20 patients in the WALK group. Details of the additional physiotherapy content recorded in GT and WALK groups are published elsewhere (16).
| Table II. Total amount of walking exercise and exertion in both walking groups during rehabilitation (n = 37). The development of training speed (maximum 2 km/h) and body-weight support (BWS) is presented for the gait trainer group (GT). The walking exercise group (WALK) received intensive traditional walking training |
| | GT Mean (SD) | WALK Mean (SD) | p* | 95% CI |
| Walking exercise time, min | 292 (16) | 287 (30) | 0.536 | –11 to 22 |
| Distance, m | 8512 (1695) | 10382 (5662) | 0.174 | –4624 to 884 |
| Heart rate during exercise† | 97.2 (14.3) | 99.0 (17.8) | 0.740 | –12.7 to 9.1 |
| Borg Scale, score | 13.9 (1.1) | 14.9 (0.9) | 0.009 | –1.6 to –0.2 |
| Speed_1, km/h | 1.5 (0.2) | | NA | |
| Speed_2, km/h | 1.8 (0.3) | | NA | |
| Speed_3, km/h | 1.9 (0.2) | | NA | |
| BWS_1, % | 34.1 (32.4) | | NA | |
| BWS_2, % | 11.6 (18.4) | | NA | |
| BWS_3, % | 5.3 (10.1) | | NA | |
| *p-values obtained using independent samples t-test. p < 0.05 is considered significant. †Mean of heart rate in the last minute of 20 min exercise over 15 sessions. SD: standard deviation; 1: in the first session; 2: in the tenth session; 3: in the last session. CI: confidence interval; NA: not applicable. |
Conventional treatment path. For all patients in the CT group, physiotherapy continued with 1 or 2 physiotherapy sessions daily according to individual needs. The patients in the CT group were transferred from the acute care hospital 0–8 days (mean 3.7 (standard deviation (SD) 3) days) after the first measurements at the start of the study, whereas the patients in the GT and WALK group remained for 3 weeks. Patients in the CT group were transferred to the health centre (n = 4), home (n = 3) or to the rehabilitation hospital (n = 3), subsequently visiting the hospital for the same tests as the patients in the GT and WALK groups.
Effects of rehabilitation
Primary outcome FAC. The intensive therapy with gait training (either GT or WALK) resulted in better improvement in walking ability compared with the CT group. It reached significant group difference in repeated measures ANOVA (Table III, rehabilitation improvement: F1,44 = 59.508, p < 0.0001; group difference: F2,44 = 4.036, p = 0.025), but not in the Kruskal-Wallis test for different time-points (rehabilitation improvement: Wilcoxon Z = –5.079, p < 0.0001; group difference: Kruskal-Wallis χ2 at start 0.022, p = 0.989, and at 3 weeks 4.684, p = 0.096). Median FAC change was 2 (interquartile range 1–3) in GT, 3 (0.25–3) in WALK, and 0 (0–1.5) in CT by 3 weeks.
None of the patients were in FAC 4 or 5 at start, but 5/17 (29.4%) in GT, 4/20 (20.0%), and 1/10 (10%) achieved either FAC 4 or 5 at the end.
Secondary outcomes. Secondary walking test data were statistically analysed for the GT and WALK groups (see above). Table IV shows patients’ participation in the different measurements at the different time-points. At the end of 3 weeks, the number of patients who were tested for the 10MWT and 6MWT increased by 41% and 47%, respectively, in the GT group; 50% and 75% in the WALK group, and 10% in the CT group. For example, 14/17 patients in the GT group, and 17/20 patients in the WALK group, but only 2/10 patients in the CT group were able to perform the 10 MWT at the end of 3 weeks.
| Table IV. Participation of 47 patients in different measurements at start of rehabilitation, after 3 weeks, at end of rehabilitation, and at 6-month follow-up |
| Group | n | Time | FAC | 10MWT | 6MWT | Motor scales† | RMI |
| GT | 17 | At start | 17 | 7 | 4 | 17 | 16 |
| | 17 | 3 weeks | 17 | 14 | 12 | 17 | 17 |
| | 16 | Follow-up | 16 | 15 | 13 | 15 | 15 |
| WALK | 20 | At start | 20 | 7 | 1 | 20 | 20 |
| | 20 | 3 weeks | 20 | 17 | 16 | 20 | 20 |
| | 19 | Follow-up | 19 | 18 | 17 | 18 | 18 |
| CT | 10 | At start | 10 | 1 | 1 | 10 | 9 |
| | 10 | 3 weeks | 10 | 2 | 2 | 10 | 10 |
| | 10 | Follow-up | 10 | 8 | 8 | 10 | 10 |
| †Motor scales include Modified Motor Assessment Scale, Rivermead Motor Assessment subscales of gross motor function and leg&trunk. FAC: Functional Ambulatory Category; 10MWT: 10-metre walk test; 6MWT: 6-minute walk test; RMI: Rivermead Mobility Index; GT: intensive therapy with gait trainer exercise; WALK: intensive therapy with walking training over ground; CT: conventional treatment. |
There were no differences between the GT and WALK groups in secondary walking test data (Table III, group differences for 10MWT: F1,21 = 0.588, p = 0.452; for 6MWT: F1,19 = 0.375, p = 0.547). Mean 10MWT decreased by 16 sec in GT and 30 sec in WALK (rehabilitation improvement: F1,21 = 6.862, p = 0.016), and mean 6MWT increased by 125 m and by 151 m (rehabilitation improvement: F1,19 = 45.675, p < 0.0001) during 3 weeks of rehabilitation.
| Table III. Gait and motor task performance of patients with acute stroke at start of rehabilitation, and at end of rehabilitation |
| Parameters | Group (n) | At start, mean (95% CI) | 3 weeks, mean (95% CI) | p-value* |
| FAC | GT (17) | 0 (0–1†) | 3 (1–4.5†) | |
| | WALK (20) | 0 (0–1†) | 3 (1.25–3†) | |
| | CT (10) | 0 (0–1.25†) | 0.5 (0–3†) | < 0.001 |
| | Group difference p-value | | | 0.025 |
| 10MWT time, sec | GT (9) | 29.4 (0.0–62.1) | 13.2 (7.0–19.4) | |
| | WALK (14) | 44.2 (18.0–70.4) | 14.6 (9.6–19.5) | 0.016 |
| | Group difference p-value | | | 0.452 |
| 6MWT distance, m | GT (9) | 196.4 (143.9–248.9) | 321.2 (218.1–424.3) | |
| | WALK (12) | 220.1 (174.7–265.6) | 370.8 (280.5–459.1) | < 0.001 |
| | Group difference p-value | | | 0.547 |
| MMAS score, points | GT (17) | 19.9 (14.6–25.3) | 33.5 (27.9–39.1) | |
| | WALK (20) | 16.4 (11.4–21.3) | 30.8 (25.6–35.9) | |
| | CT (10) | 15.3 (8.3–22.3) | 20.9 (13.6–28.2) | < 0.001 |
| | Group difference p-value | | | 0.003 |
| RMAg score, points | GT (17) | 4.5 (3.0–6.0) | 8.4 (6.7–10.2) | |
| | WALK (20) | 3.7 (2.3–5.1) | 8.4 (6.8–10.0) | |
| | CT (10) | 2.3 (0.4–4.2) | 6.3 (4.0–8.6) | < 0.001 |
| | Group difference p-value | | | 0.513 |
| RMAl&t score, points | GT (17) | 4.0 (2.8–5.2) | 6.8 (5.4–8.2) | |
| | WALK (20) | 3.2 (2.1–4.3) | 6.9 (5.6–8.3) | |
| | CT (10) | 2.7 (1.1–4.3) | 4.5 (2.6–6.4) | < 0.001 |
| | Group difference p-value | | | 0.100 |
| RMI score, points | GT (16) | 5.7 (4.1–7.3) | 9.8 (7.7–11.9) | |
| | WALK (20) | 5.0 (3.6–6.4) | 9.8 (7.9–11.6) | |
| | CT (10) | 3.2 (1.1–5.3) | 8.0 (5.2–10.8) | < 0.001 |
| | Group difference p-value | | | 0.703 |
| *p-values are obtained using analysis of variance for repeated measures (reconstructed data for 10MWT and 6MWT). p < 0.05 is considered significant. † Values are median and interquartile ranges. CI: confidence interval; FAC, Functional Ambulatory Category; GT, intensive therapy with gait trainer exercise; WALK, intensive therapy with walking training over ground; CT, conventional treatment; 10MWT, 10-metre walk test; 6MWT, 6-minute walk test; MMAS, Modified Motor Assessment Scale; RMAg, Rivermead Motor Assessment subscale of gross motor function; RMAl&t, Rivermead Motor Assessment subscale of leg&trunk; RMI, Rivermead Mobility Index. |
Motor scale scores improved in all groups (Table III, rehabilitation improvement for MMAS: F1,44 = 123.952; for RMA g: F1,44 = 165.749; for RMA l&t: F1,44 = 65.090; for RMI: F1,42 = 142.867, p for all motor scales < 0.0001). Mean MMAS improved by 14 points in walking training groups compared with 5 points in CT (group difference: F2,44 = 6.513, p = 0.003). No significant differences were found between the groups in other motor scale improvements (group differences for RMA g: F2,44 = 0.678, p = 0.513); for RMA l&t: F2,44 = 2.424, p = 0.100; for RMI: F2,42 = 0.355, p = 0.703). The analysis of all 3 time-points (including 2 weeks) during intensive gait rehabilitation indicated that the improvements in motor ability continued throughout the 3-week rehabilitation period (data now shown).
Follow-up measures. Of the 47 patients studied, 45 participated in the follow-up at 26.4 (SD 1.1) weeks (Fig. 1). One patient in the GT group cancelled her scheduled time, and one patient in the WALK group cancelled because of illness. Motor ability continued to improve from the end of rehabilitation period to the follow-up. The improvement was seen in primary outcome as well as in the motor ability scales in all patients, and in the 10MWT in the GT and WALK groups (Table V).
Median FAC change was 0 (interquartile range 0–1) in GT, 1 (0–1.75) in WALK, and 1 (0–2.25) in CT (Table V, rehabilitation improvement: F1,42 = 33.052, p < 0.0001; group difference: F2,42 = 1.061, p = 0.355 and rehabilitation improvement: Wilcoxon Z = –4.374, p < 0.0001; group difference: Kruskal-Wallis χ2 for follow-up 3.240, p = 0.198). More independent walking ability as seen in final FAC 4 or 5 was achieved by 10 of 16 (62.5%) in GT, 10 of 19 (52.6%) in WALK, and 2 of 10 (20.0%) in CT. The secondary walking test data was calculated for those patients who were able to perform the tasks at both time-points. Statistical analysis of the walking test data was possible only for the GT and WALK groups. Mean 10MWT decreased by 4.3 sec in GT and 0.4 sec in WALK (rehabilitation improvement: F1,27 = 4.432, p = 0.045; group interquartile range difference: F1,27 = 3.084, p = –0.090). Other measures of motor abilities also continued to show improvement, although the groups behaved somewhat differently (Table V).
| Table V. Motor task performance of patients with acute stroke at end of rehabilitation and at 6 month follow-up. Values are given as mean (95% confidence interval) if not otherwise stated |
| Parameters | Group (n) | 3 weeks | 6 months | p-value* |
| FAC, median (IQR) | GT (16) | 3 (1–4.75†) | 4 (1–5†) | |
| | WALK (19) | 3 (1–3†) | 4 (3–4†) | |
| | CT (10) | 0.5 (0–3†) | 2.5 (1–3.25†) | < 0.001 |
| | Group difference p-value | 0.355 |
| 10MWT time, sec | GT (13) | 17.4 (12.1–22.7) | 13.1 (7.9–18.3) | |
| | WALK (16) | 15.0 (10.2–19.8) | 14.6 (9.9–19.3) | 0.045 |
| | Group difference p-value | 0.090 |
| 6MWT distance, m | GT (11) | 289.8 (201.8–377.9) | 385.0 (306.3–463.7) | |
| | WALK (15) | 355.5 (280.1–430.9) | 337.1 (269.7–404.5) | 0.082 |
| | Group difference p-value | 0.013 |
| MMAS score, points | GT (16) | 33.6 (27.8–39.5) | 33.0 (28.1–37.9) | |
| | WALK (19) | 31.4 (26.0–36.7) | 34.2 (29.7–38.8) | |
| | CT (10) | 20.9 (13.5–28.3) | 27.1 (20.8–33.4) | 0.004 |
| | Group difference p-value | 0.024 |
| RMAg score, points | GT (16) | 8.4 (6.5–10.2) | 10.7 (9.3–12.1) | |
| | WALK (19) | 8.4 (6.7–10.1) | 10.8 (9.5–12.1) | |
| | CT (10) | 6.3 (4.0–8.6) | 8.8 (7.0–10.6) | < 0.001 |
| | Group difference p-value | 0.980 |
| RMAl&t score, points | GT (16) | 6.9 (5.4–8.5) | 7.8 (6.4–9.2) | |
| | WALK (19) | 6.9 (5.5–8.4) | 7.8 (6.6–9.1) | |
| | CT (10) | 4.5 (2.6–6.4) | 5.8 (4.1–7.5) | < 0.001 |
| | Group difference p-value | | | 0.764 |
| RMI score, points | GT (16) | 9.8 (7.7–11.9) | 12.3 (10.8–13.7) | |
| | WALK (19) | 9.8 (7.9–11.8) | 12.4 (11.1–13.7) | |
| | CT (10) | 7.8 (5.1–10.5) | 10.(9 9.1–12.7) | < 0.001 |
| | Group difference p-value | 0.817 |
| *p-values are obtained using analysis of variance for repeated measures. p < 0.05 is considered significant. † Values are median and interquartile ranges. FAC: Functional Ambulatory Category; GT: intensive therapy with gait trainer exercise; WALK: intensive therapy with walking training over ground; CT: conventional treatment; 10MWT: 10-metre walking test; 6MWT: 6-minute walk test; MMAS: Modified Motor Assessment Scale; RMAg: Rivermead Motor Assessment subscale of gross motor function; RMAl&t: Rivermead Motor Assessment subscale of leg&trunk; RMI: Rivermead Mobility Index; IQR: interquartile range. |
DISCUSSION
Motor ability improved in all 3 groups during the 3 weeks of study very early after stroke, but intensive gait-oriented rehabilitation resulted in better walking ability and better MMAS scores compared with conventional treatment. In spite of similar improvements in the early phase in both walking training groups, the time and effort required to achieve the results were different, favouring the patients in the GT group.
Previous studies have shown that a large amount of walking training can be accomplished while patients are in the subacute (10, 11) or chronic (9) stages of stroke. The present study shows that also a large amount of walking training can be accomplished during very early acute stage of stroke. The onset of stroke was only 8 days (mean and median) before the initiation of intensive therapy in the present study. The earliest start of training was as early as at the third day, and 9 patients started before the eighth day following stroke onset. In addition, 33 of the 47 patients (70%) were non-ambulatory at the start of the study. However, the patients rated their walking training as only slightly strenuous or strenuous. Furthermore, patients in the GT group gave lower perceived exertion ratings than those in the WALK group. Moreover, the patients in the GT group achieved their 20 min walking exercise in much less time than the 1 h allocated, compared with the often full 1 h needed by the patients in the WALK group. Electromechanically assisted walking training was less strenuous. A harness and motor-driven footplates in the gait trainer eased the effort needed compared with walking over ground and thus also decreased the need for breaks. The distance walked by the GT and WALK groups did not differ significantly. The patients in the CT group were transferred on average after 3.7 days of the first set of measurements, either to the health centre, home or to the rehabilitation hospital. The 3 patients transferred to the rehabilitation hospital may have received more physiotherapy. However, even in rehabilitation hospitals the conventional treatment protocol does not include as large amount of walking exercise therapy as was delivered in the present study.
The walking test data showed clearly better walking ability outcomes in those patients in walking training groups. Patients in the GT and WALK groups achieved median FAC 3 at 3 weeks, whereas all patients in the CT group needed either the continuous support of 2 assistants or someone for balance support (median FAC 0.5). Furthermore, 29% of the patients in GT, 20% in WALK, and only 10% in CT achieved the more independent walking ability, i.e. FAC 4 or 5, at 3 weeks. The secondary walking test data supported the primary outcome, and walking velocity and endurance increased in both walking training groups. Walking velocity in the 10MWT increased by 0.4 m/sec in the walking training groups, improving to 0.76 m/sec in the GT group and 0.68 m/sec in the WALK group by the end of the rehabilitation period. The mean walking distance achieved in the 6MWT increased by 125–151 m in the GT and WALK groups, reaching 321 m in the GT group and 371 m in the WALK group. In addition to gait, improvement in other motor abilities was seen in all groups. MMAS differentiated the motor ability between the groups, other motor scales showing similar improvements. MMAS indicated 2.5 times better improvements in walking training groups compared with CT.
A recent Cochrane Review of electromechanical-assisted training for walking raises a number of important issues (4). The authors concluded that patients who receive electromechanical-assisted gait training in combination with physiotherapy after stroke are more likely to achieve independent walking than patients receiving gait training without these devices. However, the interventions used in different studies were not always comparable. In order to study whether electromechanical-assisted gait training is more efficient than walking training with traditional methods, we should compare these 2 interventions. The Bobath concept by itself does not guarantee that the training content is always gait-oriented. In the present study, the 2 intervention groups performed strictly gait exercises in different ways, yet showed no significant differences in outcome. However, the improvements achieved using the gait trainer required much less effort. It is important to emphasize that in the present study, the patients were approximately one month since stroke onset at the end of study period. In the Cochrane Review, most patients were likely to be only starting training at this stage post-stroke. It would be interesting to see what influence the time since stroke onset would have as a covariate on the Cochrane results.
The follow-up measures at 6 months showed that all patients continued to improve their gait and other motor ability, yet the walking ability of the CT group remained poorer compared with GT and WALK. All patients in the walking training groups continued to improve also in terms of increased gait velocity. The mean increase in over ground walking velocity was 0.19 m/sec in the GT and 0.01 m/sec in the WALK group suggesting a tendency to dissociation of improvement between GT and WALK. The mean increase in 6MWT by 95 m in the GT group and decrease in the WALK group showed differences between our groups in later recovery phase. A positive development was seen in all motor scales with somewhat different speeds of recovery in 3 groups. The later phase, however, is not the focus of the current report.
The limitation of our study is that we did not have a formal record of the time used to achieve the 20 min of walking during 1 h in the training groups. Mostly physiotherapists recorded the time used for real practice and that used for breaks. Although not formal, the aforementioned time-use data were obtained. We also missed to formally record effort of physiotherapists. The reconstructed model of secondary walking tests biased the values towards the means, thus possibly decreasing the walking test differences between the GT and WALK group. However, it made comparison of the secondary walking test data possible in patients in the early acute stage of stroke, and the model treated both groups similarly. Another possibility would have been to assign zero to non-performance of the task; however, from our point of view the reconstructed model resulted in a more accurate assessment of improvements.
Clinical message. In patients early after stroke intensive walking training in combination with other physiotherapy resulted in significant improvements in walking. Gait and motor task performance improved irrespective of whether the walking exercise was performed using the gait trainer or over ground. However, the time and effort required to achieve the results were different, favouring the gait trainer group. The patients receiving conventional treatment requiring far less physiotherapy time and effort also improved their motor ability; however, their walking ability remained poor. Our aim in the present study was to motivate patients to make a greater effort with more repetitions of training and to start gait therapy as early as possible after stroke. Where electromechanical-assisted gait devices are available, they should be used, as they diminish the burden that heavy exercise imposes on both patients and therapists. However, where they are not available, both patients and physiotherapist should consider making more effort to achieve the same level of repetitions compared with that obtained with the use of devices.
ACKNOWLEDGEMENTS
We thank the physicians at the Department of Neurology, Kuopio University Hospital, and Dorota Musialowicz, PT, for her help during the data collection and Markku Kauppinen, MSc, at the Finnish Centre for Interdisciplinary Gerontology, University of Jyväskylä, for statistical assistance.
This study was supported by the Brain Research and Rehabilitation Center Neuron, Kuopio, Finland, the Department of Neurology, University of Kuopio, and Kuopio University Hospital, Kuopio, Finland (grant # EVO477338, 57/2003, 36/2004), and Academy of Finland (grant # 114291). No commercial party having a direct financial interest in the results of the research supporting this article has or will confer a benefit upon the authors or upon any organization with which the authors are associated. The Gait Trainer exercise equipment was purchased at the standard price. No author has any financial interest in the company manufacturing the equipment.
REFERENCES
1. Hankey GJ, Jamrozik K, Broadhurst RJ, Forbes S, Anderson CS. Long-term disability after first-ever stroke and related prognostic factors in the Perth community stroke study, 1989–1990. Stroke 2002; 33: 1034–1040.
2. Hesse S, Bertelt C, Schaffrin A, Malezic M, Mauritz KH. Restoration of gait in nonambulatory hemiparetic patients by treadmill training with partial body-weight support. Arch Phys Med Rehabil 1994; 75: 1087–1093.
3. Malouin F, Potvin M, Prevost J, Richards CL, Wood-Dauphinee S. Use of an intensive task-oriented gait training program in a series of patients with acute cerebrovascular accidents. Phys Ther 1992; 72: 781–789; discussion 789–793.
4. Mehrholz J, Werner C, Kugler J, Pohl M. Electromechanical-assisted training for walking after stroke. Cochrane Database Syst Rev 2007; (4): CD006185.
5. Hesse S, Uhlenbrock D. A mechanized gait trainer for restoration of gait. J Rehabil Res Dev 2000; 37: 701–708.
6. Colombo G, Joerg M, Schreier R, Dietz V. Treadmill training of paraplegic patients using a robotic orthosis. J Rehabil Res Dev 2000; 37: 693–700.
7. Schmidt H, Werner C, Bernhardt R, Hesse S, Kruger J. Gait rehabilitation machines based on programmable footplates. J Neuroengineering Rehabil 2007; 4: 2.
8. Veneman JF, Kruidhof R, Hekman EE, Ekkelenkamp R, Van Asseldonk EH, van der Kooij H. Design and evaluation of the LOPES exoskeleton robot for interactive gait rehabilitation. IEEE Trans Neural Syst Rehabil Eng 2007; 15: 379–386.
9. Peurala SH, Tarkka IM, Pitkanen K, Sivenius J. The effectiveness of body weight-supported gait training and floor walking in patients with chronic stroke. Arch Phys Med Rehabil 2005; 86: 1557–1564.
10. Tong RK, Ng MF, Li LS. Effectiveness of gait training using an electromechanical gait trainer, with and without functional electric stimulation, in subacute stroke: a randomized controlled trial. Arch Phys Med Rehabil 2006; 87: 1298–1304.
11. Pohl M, Werner C, Holzgraefe M, Kroczek G, Mehrholz J, Wingendorf I, et al. Repetitive locomotor training and physiotherapy improve walking and basic activities of daily living after stroke: a single-blind, randomized multicentre trial (DEutsche GAngtrainerStudie, DEGAS). Clin Rehabil 2007; 21: 17–27.
12. Mayr A, Kofler M, Quirbach E, Matzak H, Frohlich K, Saltuari L. Prospective, blinded, randomized crossover study of gait rehabilitation in stroke patients using the lokomat gait orthosis. Neurorehabil Neural Repair 2007; 21: 307–314.
13. Holden MK, Gill KM, Magliozzi MR, Nathan J, Piehl-Baker L. Clinical gait assessment in the neurologically impaired. Reliability and meaningfulness. Phys Ther 1984; 64: 35–40.
14. Mahoney FI, Barthel DW. Functional evaluation: the Barthel Index. Md State Med J 1965; 14: 61–65.
15. De Haan R, Horn J, Limburg M, Van Der Meulen J, Bossuyt P. A comparison of five stroke scales with measures of disability, handicap, and quality of life. [see comment]. Stroke 1993; 24: 1178–1181.
16. Peurala SH, Airaksinen O, Jakala P, Tarkka IM, Sivenius J. Effects of intensive gait-oriented physiotherapy during early acute phase of stroke. J Rehabil Res Dev 2007; 44: 637–648.
17. Borg GA. Psychophysical bases of perceived exertion. Med Sci Sports Exerc 1982; 14: 377–381.
18. Wade DT, editor. Measurement in neurological rehabilitation. Oxford: Oxford University Press; 2000, p. 388.
19. Guyatt GH, Pugsley SO, Sullivan MJ, Thompson PJ, Berman L, Jones NL, et al. Effect of encouragement on walking test performance. Thorax 1984; 39: 818–822.
20. Carr JH, Shepherd RB, Nordholm L, Lynne D. Investigation of a new motor assessment scale for stroke patients. Phys Ther 1985; 65: 175–180.
21. Collen FM, Wade DT, Bradshaw CM. Mobility after stroke: reliability of measures of impairment and disability. Int Disabil Stud 1990; 12: 6–9.