OBJECTIVE: To quantify the long-term use of various types of healthcare services in patients with traumatic brain injury and to estimate the relative contribution of predisposing characteristics, enabling factors and health-related needs to determine whether there is equity in healthcare utilization.
DESIGN: Cross-sectional study.
Patients: Seventy-nine non-institutionalized moderate to severe patients with traumatic brain injury (age range 16–67 years).
METHODS: Healthcare use was measured at 3–5 years post-injury. The relative contribution of predisposing characteristics, enabling factors, and health-related needs to the utilization of various types of care was analysed using logistic regression to determine whether there was equity in healthcare utilization.
RESULTS: At least one healthcare service was used by 68% of the patients. Health-related needs explained most of the utilization. However, predisposing characteristics were also related to the use of other medical care and supportive care. Patients with a high internal locus of control were more likely to be users of supportive care, and patients with a high locus of control with the physician were more likely to visit medical specialists.
CONCLUSION: The results suggest that most of our patients who needed care, received care. However, inequity could not be ruled out completely as predisposing characteristics also contributed to some types of healthcare utilization.
Key words: traumatic brain injury, craniocerebral trauma, healthcare utilization, healthcare quality, healthcare access, healthcare evaluation.
J Rehabil Med 2009: 41: 59–65
Correspondence address: Agnes H. P. Willemse-van Son, Erasmus MC, Department of Rehabilitation Medicine, PO Box 2040, NL-3000 CA Rotterdam, The Netherlands. E-mail: a.vanson-willemse@erasmusmc.nl
Submitted February 26, 2008; accepted September 3, 2008.
INTRODUCTION
Approximately 1.6 million patients with traumatic brain injury (TBI) are admitted to hospital each year in Europe (1, 2). The outcome after TBI can vary from complete recovery to death, with many patients having long-term physical, cognitive and psychosocial disabilities. A Dutch follow-up study showed that the majority of patients with mild to moderate TBI experienced situational, cognitive, emotional and behavioural disabilities at 3–7 years post-injury (3). Approximately 41% experienced related participation restrictions (4) and needed various healthcare services. Information on healthcare needs and healthcare services that are used in the long-term are crucial for adequate planning of long-term care. Furthermore, it is important to determine whether healthcare is in fact delivered to patients who require services.
To ensure equity in healthcare utilization it is important to evaluate whether the limited healthcare services are used by those patients that need them the most. According to the model of Andersen (5, 6), healthcare utilization depends on: (i) predisposing characteristics, (ii) enabling factors, and (iii) health-related needs. Predisposing variables reflect a person’s preposition to use services. Predisposing variables comprise demographic variables (e.g. age, gender), health beliefs, and coping styles. Enabling factors determine whether healthcare services are available (e.g. income, availability of services where people live, insurance, etc.). Need factors represent the most immediate cause for health service use, reflected by perception of illness, symptoms, diagnosis and functioning (7). The model was designed to explain the use of services rather than to focus on important interactions that take place as people receive care, or on health outcomes (6). Andersen’s model has been used to evaluate equity in healthcare utilization in chronic diseases such as stroke, rheumatoid arthritis, heart disease and diabetes (8–11). Equitable access to healthcare services occurs when demographic and need variables account for most of the variance in utilization; this is an indication that patients receive the care they need. Inequitable access is demonstrated when care is explained by social structures, health beliefs, or enabling factors (5, 6).
A review of the predictors of healthcare utilization in the chronically ill reported that health-related needs were the most important predictors of healthcare utilization, whereas predisposing characteristics (age, sex and marital status) and most enabling factors (income, insurance and social support) were not predictive (11). In patients with myocardial infarction and in elderly patients, locus of control was also associated with healthcare utilization (12, 13). Studies in TBI populations identified the following predictors of healthcare utilization: severity of injury (14–16); physical and cognitive disability; psychosocial disability (14); sex; years of education; a longer length of stay in hospital; admittance to a hospital or rehabilitation centre for TBI (15); and motor deficits at discharge from inpatient rehabilitation (16). However, these studies had some limitations for evaluating equity in healthcare utilization: only health-related needs were investigated (14); TBI was diagnosed retrospectively with self-reports (15); and healthcare utilization was estimated from the amount that was billed to Medicaid (16).
Therefore, the aims of this study were: (i) to quantify the use of various types of healthcare services in non-institutionalized patients with moderate and severe TBI 3–5 years post-injury, and (ii) to estimate the relative contribution of predisposing characteristics, enabling factors, and health-related needs in order to determine whether there is equity or inequity in healthcare utilization. Given the principle of equity in healthcare, which is one of the basic quality indicators of the Dutch healthcare system, it is hypothesized that healthcare utilization is determined mainly by health-related needs.
MATERIAL AND METHODS
Procedure
For the present cross-sectional study a subsample of 79 patients was included from the cohort that was recruited in the Rotterdam TBI study (17). In the Rotterdam TBI study, 119 patients with TBI were consecutively enrolled from January 1999 to April 2004 in 3 medical centres: Erasmus Medical Centre in Rotterdam (entire period); University Medical Centre Utrecht (enrolment from April 2003 to February 2004); and Medical Centre Haaglanden in The Hague (enrolment from January 2003 to February 2004). The 3 centres served as treatment centres for all patients with moderate and severe TBI within their region. Patients were treated in accordance with the European Brain Injury Consortium guidelines (18).
For the Rotterdam TBI study, patients were prospectively followed-up at 3, 6, 12, 18, 24 and 36 months from April 1999 to April 2007. For this study the 36-month follow-up measurements were used. All data, except for the questionnaire on healthcare utilization, were collected in a structured interview at the participant’s home or institution of admittance by 2 study psychologists. In cases where patients suffered from serious communication impairments, a significant other or professional caregiver was interviewed. In October 2003 the questionnaire on healthcare use was added to the structured interview at the regular follow-up measurement of 3 years. As 39 patients were followed up by that time, these patients were sent the healthcare questionnaire by post up to 5 years post-injury. For these 39 patients, the other data were collected in the structural interview at the regular follow-up.
Patients
Inclusion criteria of the Rotterdam TBI study were: (i) admittance to hospital for moderate or severe TBI due to blunt or penetrating trauma (Glasgow Coma Scale (GCS) (19) score of 9–13 or 3–8, respectively); (ii) age at onset between 16 and 67 years; (iii) survival until discharge from hospital. Exclusion criteria were: (i) insufficient knowledge of the Dutch language to participate in the study; (ii) serious pre-traumatic neurological, oncological or systemic impairment (e.g. spinal cord injury, psychiatric disorders, cancer) that might interfere with the assessment of TBI-related disability. For the present study, only non-institutionalized patients were included. All patients received verbal and written information about the study and signed an informed consent form. The study was approved by the Medical Ethics Committee of Erasmus MC.
Measures
Healthcare utilization. Utilization of healthcare was assessed for a wide range of 16 healthcare services. Patients were asked if they had used these healthcare services in the last year (scored as “yes” or “no”). We selected healthcare services that were relevant in multidisciplinary care for chronic diseases, and in particular for TBI, such as a neurologist, a rehabilitation physician, a psychologist, or an activity centre. If necessary, patients were assisted by the study psychologist or a family member. Because of the small numbers, the studied healthcare services were aggregated according to care function into 4 categories: (i) general practitioner (GP); (ii) medical specialists (neurologist/neurosurgeon, urologist, eye physician, and other medical specialists); (iii) rehabilitation care (rehabilitation physician, physiotherapist, occupational therapist, speech therapist, social worker, and psychologist); and (iv) supportive care (home nurse, home help, activity centre, day care, and patient organizations). The scores were dichotomized into “use” or “no use”. In addition, the total number of all care services was calculated. Total care use was dichotomized on the fourth quartile into “high use” and “low care use”.
Independent variables. Predisposing characteristics encompass: age at injury in years, gender, living situation (with or without partner), and health beliefs. Health beliefs were assessed with the Multidimensional Health Locus of Control Scales (MHLCS) (20). The MHLCS consist of 3 separate scales: internal locus of control, locus of control with a physician, and locus of control with chance. The scales indicate how much patients believe that the health status is influenced by themselves, by a physician, or by chance. A higher score indicates that the patient attributes more influence to that factor. The scores were dichotomized into high and low locus of control on the median scores of the patients.
Enabling factors encompass: work status (working vs not working), level of education (lower or junior secondary education vs higher education), urbanization level (rural or urban), and social support. Received social support was measured with the Social Support Scale (SSL) (21). Subjects are asked to complete 4-point scale on how often they experienced a certain type of social support. The scores range from 34 to 136, where a higher score indicates more experienced social support. Scores were dichotomized on the median score into high or low social support.
Health-related factors encompass clinical aspects, and aspects of functioning and disabilities as described in the International Classification of Functioning Disability and Health (ICF) (22). The following clinical factors were assessed: TBI severity (GCS score) and co-morbidity. Co-morbidity was measured with the Cumulative Illness Rating Scale (CIRS). The CIRS is a valid and reliable instrument that rates 13 body systems on a 5-point scale (no impairment to life-threatening impairment) without using specific diagnoses (23). The numbers of body systems that had a score of 1 or higher were accumulated to a sum score for co-morbidity. Medical symptoms of TBI were not considered as co-morbidity.
Cognitive and motor functioning was determined with the Functional Independence Measure combined with the Functional Assessment Measure (FIM+FAM) (24). The FIM+FAM consists of 30 items with a 7-point scale (completely independent to totally dependent) on the domains self-care, sphincter control, transfers, locomotion, communication, psychosocial adjustment, and cognitive functioning. At 3 years post-injury many patients had ceiling effects on the FIM+FAM. Hence we used a relatively high cut-off value of 180, which corresponds to an average item score of 6 (modified independence, needing more time or devices). Scores were dichotomized into lower than 180 (indicating limitations in functioning) and 180 and above (indicating independence in functioning).
The presence of depression was measured with the Wimbledon Self-Report Scale (WSRS) (25). The WSRS consists of 30 questions on how often a certain feeling was felt in the past 4 weeks (most of the time, quite often, only occasionally, not at all). The items are transformed into a 2-point scale with a maximum score of 30. Scores ranging from 0 to 7 are considered as normal functioning, scores ranging from 8 to 10 are considered as borderline for mood disorders, and scores of 11 to 30 are considered as cases with a clinically significant mood disorder. The scores were dichotomized into lower or equal to 7 indicating no depression, and above 7 indicating depression.
Participation restrictions were assessed with the Sickness Impact Profile-68 (SIP-68) (26), which has 68 statements on behaviour, feelings, and functions. The Respondents are asked if these statements apply to their current situation (yes/no) and whether they are health-related. The SIP-68 score is calculated by summing all positively scored items (range 0–68). A higher score indicates more participation restrictions. The SIP-68 has excellent test-retest reliability (intra class correlation = 0.97) (27). The SIP-68 was originally not primarily intended to measure participation restrictions but was developed to measure functional health status. However, the version from which the SIP-68 was derived, the Sickness Impact Profile-136 (SIP-136) (28), covered a broad bandwidth of different ICF categories and among them the category activities and participation is represented most extensive (29). Despite the limitation that the SIP-68 also measures other ICF categories, we considered it suitable for measuring participation restrictions. The SIP-68 was dichotomized on the median into restricted or not restricted in participation.
Community integration was determined with the Community Integration Questionnaire (CIQ) (30), which assesses daily activities in the home, social environment, and in work or education. It contains 15 questions on how activities are usually performed (alone, with another person, by someone else) and how frequently activities are performed. The score ranges from 0 to 29, with a higher score indicating better community integration. The reliability of the CIQ is sufficient (31). The CIQ was dichotomized on the median into high or low community integration.
Statistical analysis
Descriptive statistics were performed with SPSS 12.0.1. Four separate logistic regression analyses were performed for each of the 4 types of care and for high total use of care.
First, the association between the independent variables and the 4 types of care and total care were tested with χ2 tests. Effect sizes were expressed with odds ratios (ORs) and 95% confidence intervals (95% CI). Because the sample was relatively small and there were many independent variables, we set the criterion for inclusion in the multivariate model at 0.10. Variables that were selected for the multivariate model were tested for interrelations with Spearman’s rho. If there were interrelations (Spearman’s rho ≥ 0.80) between a type of factors (pre-disposing, enabling, or need factors), than the highest contributor was selected for the multivariate model. If there were interrelations (Spearman’s rho ≥ 0.80) between types of factors, multiple models were built in order to investigate the influence of the contributing factors.
Secondly, the selected independent variables were analysed with a backward logistic regression analyses. A p-value below 0.05 was considered significant.
RESULTS
Study population
Of the 119 patients with TBI included in the Rotterdam TBI study, 4 patients were institutionalized, 3 were deceased, and 16 patients were lost at the time of follow-up. Of the 96 eligible patients, 79 (82%) completed the healthcare utilization questionnaire at 3–5 years post-injury. Seventeen patients had not returned their questionnaire. Compared with non-participants, participants had a more severe initial injury (lower GCS score) (p = 0.040) and higher education levels (p = 0.034). There were no significant differences for the other independent variables between participants and non-participants.
Table I presents the characteristics of the study participants: mean age was 35 years, there were twice as many males as females, and the majority (72%) lived with a partner or parent. The mean GCS score was 6.7 (standard deviation = 3.0). Co-morbidity was present in 57 patients (74%). Limitations in functioning were found for 7 patients (9%) and depression was present in 10 patients (13%).
| Table I. Characteristics of all participating patients (n = 79) |
| Patient characteristics | |
| Age, years, mean (SD) | 35 (13.3) |
| Gender | |
| Male, n (%) | 54 (68) |
| Female, n (%) | 25 (32) |
| Living situation | |
| Without partner or parent, n (%) | 22 (28) |
| With partner or parent, n (%) | 57 (72) |
| Internal locus of control (n = 67) | |
| Low (MHLCS internal ≤ 22), n (%) | 32 (48) |
| High (MHLCS > 22), n (%) | 35 (52) |
| Locus of control with a physician (n = 67) | |
| Low (MHLCS physician ≤ 15), n (%) | 31 (46) |
| High (MHLCS physician > 15), n (%) | 36 (54) |
| Locus of control with chance (n = 67) | |
| Low (MHLCS chance ≤ 17), n (%) | 32 (48) |
| High (MHLCS chance > 17), n (%) | 35 (52) |
| Education (n = 78) | |
| Low (lower or junior secondary education), n (%) | 31 (40) |
| High (higher than junior secondary education), n (%) | 47 (60) |
| Work status at follow-up | |
| Not working, n (%) | 34 (43) |
| Working, n (%) | 45 (57) |
| Urbanization level | |
| Rural, n (%) | 26 (33) |
| Urban, n (%) | 53 (67) |
| Social support | |
| Low (SSL ≤ 72), n (%) | 34 (50) |
| High (SSL > 72), n (%) | 34 (50) |
| Glasgow Coma Scale, mean (SD) | 6.7 (3.0) |
| Co-morbidity (n = 77) | |
| Present, n (%) | 57 (74) |
| Absent, n (%) | 20 (26) |
| Functioning | |
| Limitations (FIM+FAM < 180), n (%) | 7 (9) |
| Independent (FIM+FAM ≥ 180), n (%) | 72 (91) |
| Depression (n = 77) | |
| Present (WSRS > 7), n (%) | 10 (13) |
| Absent (WSRS ≤ 7), n (%) | 67 (87) |
| Participation (n = 78) | |
| Restricted (SIP-68 > 9), n (%) | 40 (51) |
| Not restricted (SIP ≤ 9), n (%) | 38 (49) |
| Community integration | |
| Low integration (CIQ ≤ 19), n (%) | 44 (56) |
| High integration (CIQ > 19), n (%) | 35 (44) |
| SD: standard deviation; FIM+FAM: Functional Independence Measure combined with the Functional Assessment Measure; CIQ: Community Integration Questionnaire; WSRS: Wimbledon Self-Report Scale; SIP-68: Sickness Impact Profile-68; MHLCS: Multidimensional Health Locus of Control Scales; SSL: Social Support Scale. |
Healthcare utilization
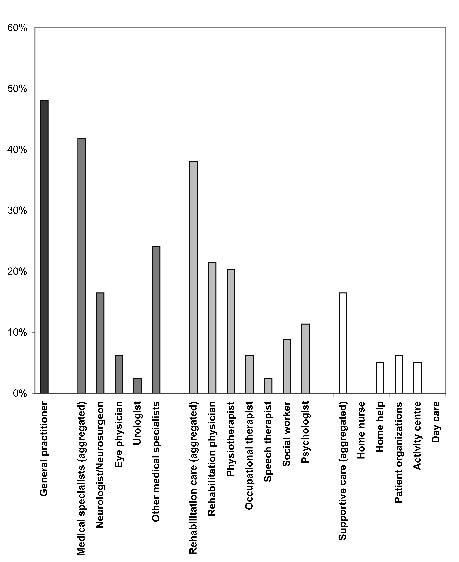
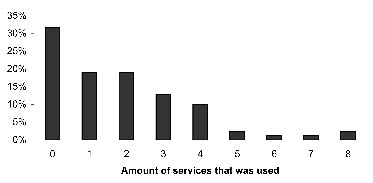
Fig. 1 presents the long-term utilization by the 79 patients of the different types of healthcare. Of these 79 patients, 26 (32%) did not use any care at all and the remainder received various types of care. Of all healthcare services, the GP was contacted most frequently (48%). Rehabilitation care was used by 38% of the patients; 42% visited medical specialists, and 16.5% had supportive care. Within rehabilitation care, most contacts were with the rehabilitation physician, followed by the physical therapist. Several supportive care services were equally received: home help, support from other TBI victims, and activity centres. Fig. 2 shows the amount of different type of services that were used; high overall care use (3 or more services) was found in 24 patients (30%).
Fig. 1. Long-term healthcare utilization by the group of patients with moderate to severe traumatic brain injury (n = 79). General practitioner; Medical specialists; Rehabilitation care; Supportive care.
Fig. 2. Amount of different types of healthcare services used at 3–5 years post-injury by the group of patients with moderate to severe traumatic brain injury (n = 79).
Determinants of healthcare utilization
The results of the univariate and multivariate analyses for utilization of the 4 types of care and high overall care use are presented in Table II. Only significant univariate results (p < 0.10) and significant multivariate results (p < 0.05) are presented.
| Table II. Univariate and multivariate analyses for long-term utilization of high overall care use and all aggregated care types |
| Variables | Univariate analyses | Multivariate analyses |
| Odds ratio | 95% CI | p-value | Odds ratio | 95% CI | p-value |
| High overall care use |
| Work status (not working) | 2.451 | 0.919–6.536 | 0.070 | | | |
| Functioning (limitations) | 6.974 | 1.247–39.005 | 0.024 | | | |
| Participation (restrictions) | 3.268 | 1.166–9.174 | 0.021 | 3.273 | 1.166–9.190 | 0.24 |
| General practitioner |
| Co-morbidity in number of systems | 1.631 | 1.093–2.435 | 0.017 | | | |
| Medical specialists |
| Gender (male) | 2.387 | 0.858–6.623 | 0.091 | | | |
| Participation (restrictions) | 2.392 | 0.951–6.024 | 0.062 | | | |
| Locus of control with physician (high) | 2.444 | 0.887–6.739 | 0.081 | 3.759 | 1.191–11.862 | 0.024 |
| Co-morbidity in number of systems | 1.473 | 1.012–2.145 | 0.043 | 1.767 | 1.106–2.823 | 0.017 |
| Rehabilitation care |
| Functioning (limitations) | 3.448 | 1.325–8.929 | 0.010 | | | |
| Supportive care |
| Functioning (limitations) | 4.650 | 0.903–23.952 | 0.083 | | | |
| Participation (restrictions) | 15.873 | 1.945–125.000 | 0.001 | 14.373 | 1.611–128.212 | 0.017 |
| Internal locus of control (high) | 10.731 | 1.274–90.363 | 0.010 | 11.693 | 1.298–105.352 | 0.028 |
| TBI-severity (GCS score) | 0.762 | 0.582–0.997 | 0.048 | | | |
| Only the significant (p < 0.10) results for the univariate analyses and significant results (p < 0.05) for the multivariate analyses are reported here. 95% CI: 95% confidence interval; GCS: Glasgow Coma Scale; TBI: traumatic brain injury. |
High overall care use. In the univariate analyses, not working at follow-up, limitations in functioning, and restrictions in participation were risk factors for a high overall use of healthcare. Significant intercorrelations were found between limitations in functioning and respectively work (Spearman’s rho = 0.36) and restrictions in participation (Spearman’s rho = 0.31); patients with limitations in functioning were less likely to work and were more likely to have participation restrictions. Because these values were below the cut-off value of 0.80, all variables were entered in the multivariate model. In the multivariate model, only restrictions in participation were significant (OR = 3.273, 95% CI: 1.166–9.190, p < 0.024).
General practitioner. In the univariate analyses, more co-morbidity was the only significant determinant for GP use (OR = 1.631 per impaired body system, 95% CI: 1.093–2.435, p = 0.017). Therefore, no multivariate model was tested.
Medical specialists. In the univariate analyses, male gender, more co-morbidity, restrictions in participation, and a high locus of control for a physician were risk factors for utilization of medical specialists. The spearman correlations between these variables were all not significant and therefore all variables were entered in the multivariate model. In the multivariate model, more co-morbidity (OR per extra impaired body system = 1.767, 95% CI: 1.106–2.823, p = 0.017) and a high locus of control for a physician (OR = 3.759, 95% CI: 1.191–11.862, p = 0.024) remained significant risk factors.
Rehabilitation care. In the univariate analyses, only dependence in functioning was significant (OR = 4.700, 95% CI: 0.850–25.988, p = 0.098); therefore no multivariate model was tested.
Supportive care. In the univariate analyses, a more severe initial injury, dependence in functioning, restrictions in participation, and a high internal locus of control were risk factors for use of supportive care. Interrelations were found between dependence and functioning and restrictions in participation (Spearman’s rho = 0.31) and between initial severity and internal locus of control (Spearman’s rho = 0.26); patients who were dependent in functioning were more likely to have restrictions in participation and more severely injured patients were more likely to have higher internal locus of control. Because these values were below the cut-off value of 0.80, all variables were entered in the multivariate model. In the multivariate model, restrictions in participation (OR = 14.373, 95% CI: 1.611–128.212, p = 0.017) and a high internal locus of control (OR = 11.693, 95% CI: 1.298–105.352, p = 0.028) remained significant risk factors.
DISCUSSION
This study investigated which healthcare facilities were used by patients with moderate to severe TBI 3–5 years post-injury. equity or inequity was determined by analysing which factors contributed to the use of healthcare services, i.e. predisposing characteristics, enabling factors, or health-related needs.
At least one healthcare service was used by 68% of the patients in the long term, which is similar to the results of earlier studies on patients with TBI (14, 15). The GP, medical specialists, and rehabilitation care were contacted most frequently. Compared with the general Dutch population, a smaller percentage of the study population had visited a GP (32). The GP is the first contact and gatekeeper in the Dutch healthcare system and referrals are generally made by GPs; however, because our patients were already in the system they probably needed fewer referrals from the GP. A remarkable finding was that physical therapists were contacted more frequently than psychologists or social workers, despite that in the long term most patients with TBI experience psychosocial problems rather than physical problems (3, 33). Perhaps rehabilitation programmes focused more on regaining physical capacity than on psychosocial issues. Another explanation might be that patients have organized care themselves and were more inclined to arrange physical support than psychosocial support because they were more familiar with this type of care. Many patients with TBI were using a variety of healthcare services: 49% visited at least 2 services, and 30% received 3 or more services. The utilization of multiple services underscores the importance of good collaboration and coordination between these services.
Andersen’s model (5, 6) was used to evaluate equity or inequity of care. equity in healthcare use is demonstrated when this use is mainly determined by health-related factors and not by enabling or predisposing factors. Health-related needs (such as restrictions in participation and co-morbidity) explained most of the variance of healthcare utilization. Hence, these results seem to suggest that most patients who needed care, received care. However, for medical specialists and supportive care, inequity could not be ruled out, as predisposing factors also contributed to healthcare utilization.
Patients with a high locus of control with the physician were more likely to visit medical specialists than other patients, despite comparable health-related factors. Patients with a high internal locus of control were more likely to use supportive care than other patients, despite comparable health-related factors. In contrast, a study in patients after a myocardial infarct reported that a lower belief in personal control was related to more physician visits (13). However, the differences in findings might be explained by the fact that this study used a one-dimensional health locus of control scale, in which internal and external orientations were not separate scales but opposites on the same dimension. Therefore, their results are in agreement with our findings for the use of medical specialist care but in disagreement with the use of supportive care. A study of elderly people found no association between internal locus of control and hospitalization and physician use (12). An internal orientation might lead to different actions in health behaviour, which can explain differences in findings between studies. Usually, patients with a high internal orientation control their own health by performing healthy behaviour, while patients with an external control rely on others for their health. However, their behaviour also depends on what patients expect to be effective for their health (34). If patients believe that a treatment will be beneficial, then internal orientated patients can choose an active problem-solving approach by seeking support to overcome health problems. An alternative explanation is that health locus of control might change as a consequence of continued healthcare utilization. A study of elderly people found that a continued period of hospitalization or an increase in physician visits over time, was associated with an increase in powerful others (physician) and an increase in a chance health locus of control orientation. However, internal health locus of control was not affected by continued healthcare utilization (12).
An interesting item is whether the influence of health beliefs on care use point to inequity or whether patient preferences should be considered in healthcare utilization. Andersen originally stated that there was inequity when health beliefs determined healthcare utilization (5), but later reported that it also depended on the circumstances (6). It is a matter of concern when patients refuse or do not seek healthcare because they have insufficient insight into their sickness or are unaware of their problems. However, it is now common knowledge that healthcare is more effective if patients are involved in the management of care (35, 36). With a patient-centred approach, health beliefs and expectations of patients and professionals can be matched, and patients can be activated to take some control in disease management (37). Particularly in chronic illness, this approach was found to result in a better satisfaction, adherence to treatment, and outcome (37). In the future, professionals might pay more attention to the influence of health beliefs on healthcare utilization, which might prevent some patients failing to receive the care they need because of their health beliefs.
Some caution is warranted in interpreting these results. First, because the results are based on a small study sample, some small but important associations might not have been identified. However, we assume that the sample was representative because the procedure stipulated that all patients with moderate and severe TBI be referred to the 3 recruitment centres. It is not known whether the results can also be generalized to patients with mild TBI.
Secondly, we assessed only whether the patients had contact with healthcare services, and not the frequency or intensity of the provided healthcare. Therefore, it was beyond the scope of this study to determine whether the quantity and quality of the delivered healthcare services were sufficient to deal with all experienced health problems.
Thirdly, we evaluated equity in aggregated care types and not for individual services. On the individual level, patients with TBI might still have unmet needs for healthcare services. In the long-term, patients with TBI may disappear from the healthcare system. New healthcare needs, created by altered circumstances, might therefore remain undetected. TBI research has a strong focus on short-term outcome, whereas it is a lifelong problem. Because we have not yet succeeded in identifying which patients need intensive long-term follow-up, it is recommended that TBI rehabilitation be a lifelong, well-coordinated process focusing on both the patient and their family (38).
ACKNOWLEDGEMENTS
This study was performed as part of the “Long-term prognosis of functional outcome in neurological disorders”, supervised by the Department of Rehabilitation Medicine of the VU Medical Centre, Amsterdam, and supported by the Netherlands Organisation for Health Research and Development (project: 1435.0020).
On behalf of the FuPro-II study group: G. J. Lankhorst, J. Dekker, A. J. Dallmeijer, M. J. IJzerman, H. Beckerman, V. de Groot: VU University Medical Centre Amsterdam (project coordination); E. Lindeman, G. Kwakkel, I. G. L. van de Port: University Medical Centre Utrecht/Rehabilitation Centre De Hoogstraat, Utrecht; H. J. Stam, A. H. P. Willemse-van Son: Erasmus Medical Centre, Rotterdam; G. M. Ribbers: Rehabilitation Centre Rijndam, Rotterdam; G. A. M. van den Bos: Department of Social Medicine, Academic Medical Centre, University of Amsterdam, Amsterdam, The Netherlands.
REFERENCES
1. Andlin-Sobocki P, Jonsson B, Wittchen HU, Olesen J. Cost of disorders of the brain in Europe. Eur J Neurol 2005; 12 Suppl 1: 1–27.
2. Tagliaferri F, Compagnone C, Korsic M, Servadei F, Kraus J. A systematic review of brain injury epidemiology in Europe. Acta Neurochir (Wien) 2006; 148: 255–268; discussion 268.
3. van Balen HG, Mulder T, Keyser A. Towards a disability-oriented epidemiology of traumatic brain injury. Disabil Rehabil 1996; 18: 181–190.
4. van Balen HGG, editor. Mensen met een traumatisch hersenletsel: probleeminventarisatie. [Traumatically brain-damaged people: an investigation of their problems]. Utrecht: Gehandicaptenraad; 1992 (in Dutch).
5. Andersen RM, editor. Behavioral model of families’ use of health services. Chicago: Center for health Administration Studies, University of Chicago; 1968.
6. Andersen RM. Revisiting the behavioral model and access to medical care: does it matter? J Health Soc Behav 1995; 36: 1–10.
7. van den Bos GAM, Triemstra AHM. Quality of life as an instrument for need assessment and outcome assessment of healthcare in chronic patients. Qual Health Care 1999; 8: 247–252.
8. Jacobi CE, Triemstra M, Rupp I, Dinant HJ, Van Den Bos GA. Health care utilization among rheumatoid arthritis patients referred to a rheumatology center: unequal needs, unequal care? Arthritis Rheum 2001; 45: 324–330.
9. van den Bos GA, Smits JP, Westert GP, van Straten A. Socioeconomic variations in the course of stroke: unequal health outcomes, equal care? J Epidemiol Community Health 2002; 56: 943–948.
10. Jacobi CE, Mol GD, Boshuizen HC, Rupp I, Dinant HJ, Van Den Bos GA. Impact of socioeconomic status on the course of rheumatoid arthritis and on related use of healthcare services. Arthritis Rheum 2003; 49: 567–573.
11. de Boer AG, Wijker W, de Haes HC. Predictors of healthcare utilization in the chronically ill: a review of the literature. Health Policy 1997; 42: 101–115.
12. Goldsteen RL, Counte MA, Goldsteen K. Examining the relationship between health locus of control and the use of medical care services. J Aging Health 1994; 6: 314–335.
13. Maeland JG, Havik OE. Use of health services after a myocardial infarction. Scand J Soc Med 1989; 17: 93–102.
14. Hodgkinson A, Veerabangsa A, Drane D, McCluskey A. Service utilization following traumatic brain injury. J Head Trauma Rehabil 2000; 15: 1208–1226.
15. High WM, Gordon WA, Lehmkuhl LD, Newton C, Vandergoot D, Thoi L, et al. Productivity and service utilization following traumatic brain injury: results of a survey by the RSA regional TBI centers. J Head Trauma Rehabil 1995; 10: 64–80.
16. Vangel SJ, Jr., Rapport LJ, Hanks RA, Black KL. Long-term medical care utilization and costs among traumatic brain injury survivors. Am J Phys Med Rehabil 2005; 84: 153–160.
17. van Baalen B, Ribbers GM, Medema-Meulepas D, Pas MS, Odding E, Stam HJ. Being restricted in participation after a traumatic brain injury is negatively associated by passive coping style of the caregiver. Brain Inj 2007; 21: 925–931.
18. Maas AI, Dearden M, Teasdale GM, Braakman R, Cohadon F, Iannotti F, et al. EBIC-guidelines for management of severe head injury in adults. European Brain Injury Consortium. Acta Neurochir (Wien) 1997; 139: 286–294.
19. Teasdale G, Jennett B. Assessment of coma and impaired consciousness. A practical scale. Lancet 1974; 2: 81–84.
20. Wallston KA, Wallston BS, DeVellis R. Development of the Multidimensional Health Locus of Control (MHLC) Scales. Health Educ Monogr 1978; 6: 160–170.
21. Van Sonderen E, editor. Measuring social support with the Social Support List-Interactions (SSL-I) and Social Support List-Discrepancies (SSL-D). A manual [Het meten van sociale steun met de Sociale Steun Lijst – Interacties (SSL-I) en Sociale Steun Lijst - Discrepanties (SSL-D), een handleiding]. Groningen: Noordelijk centrum voor gezondheidsvraagstukken, Rijksuniversiteit Groningen; 1993 (in Dutch).
22. World Health Organization. International classification of functioning, disability and health. Geneva: World Health Organization; 2002.
23. de Groot V, Beckerman H, Lankhorst GJ, Bouter LM. How to measure comorbidity. a critical review of available methods. J Clin Epidemiol 2003; 56: 221–229.
24. Turner-Stokes L, Nyein K, Turner-Stokes T, Gatehouse C. The UK FIM+FAM: development and evaluation. Functional Assessment Measure. Clin Rehabil 1999; 13: 277–287.
25. Coughlan AK. The Wimbledon self-report scale: emotional and mood appraisal. Clin Rehabil 1988; 2: 207–213.
26. de Bruin AF, Diederiks JP, de Witte LP, Stevens FC, Philipsen H. The development of a short generic version of the Sickness Impact Profile. J Clin Epidemiol 1994; 47: 407–418.
27. de Bruin AF, Buys M, de Witte LP, Diederiks JP. The sickness impact profile: SIP68, a short generic version. First evaluation of the reliability and reproducibility. J Clin Epidemiol 1994; 47: 863–871.
28. Bergner M, Bobbitt RA, Carter WB, Gilson BS. The Sickness Impact Profile: development and final revision of a health status measure. Med Care 1981; 19: 787–805.
29. Geyh S, Cieza A, Kollerits B, Grimby G, Stucki G. Content comparison of health-related quality of life measures used in stroke based on the international classification of functioning, disability and health (ICF): a systematic review. Qual Life Res 2007; 16: 833–851.
30. Willer B, Ottenbacher KJ, Coad ML. The community integration questionnaire. A comparative examination. Am J Phys Med Rehabil 1994; 73: 103–111.
31. Dijkers M. Measuring the long-term outcomes of traumatic brain injury: a review of the community integration questionnaire. J Head Trauma Rehabil 1997; 12: 74–91.
32. Centraal Bureau voor de Statistiek. Use of medical services [Gebruik medische voorzieningen] (database on the internet). Voorburg/Heerlen: Centraal Bureau voor de Statistiek. 1981–2006 [Updated 2007 Mar 20; cited 2007 Nov 30]. Available from: http://statline.cbs.nl/
33. Dikmen SS, Machamer JE, Powell JM, Temkin NR. Outcome 3 to 5 years after moderate to severe traumatic brain injury. Arch Phys Med Rehabil 2003; 84: 1449–1457.
34. Wallston KA. The importance of placing a measure of health locus of control beliefs in a theoretical context. Health Educ Res 1991; 6: 251–252.
35. Von Korff M, Glasgow RE, Sharpe M. Organising care for chronic illness. BMJ 2002; 325: 92–94.
36. Holman H, Lorig K. Patients as partners in managing chronic disease. Partnership is a prerequisite for effective and efficient healthcare. BMJ 2000; 320: 526–527.
37. Michie S, Miles J, Weinman J. Patient-centredness in chronic illness: what is it and does it matter? Patient Educ Couns 2003; 51: 197–206.
38. Ribbers GM. Traumatic brain injury rehabilitation in the Netherlands: dilemmas and challenges. J Head Trauma Rehabil 2007; 22: 231–235.