OBJECTIVE: It has been reported that the non-dominant hand of patients with Charcot-Marie-Tooth disease is stronger than the dominant hand as a result of overwork weakness. The objective of this study was to determine if this hypothesis could be verified in our population.
DESIGN: Survey.
SUBJECTS: Twenty-eight patients with Charcot-Marie-Tooth disease type I or II from a rehabilitation department of a university hospital in the Netherlands.
METHODS: The strength of 3 intrinsic muscle groups of the dominant and non-dominant hand were determined using the Medical Research Council scale and the Rotterdam Intrinsic Hand Myometer. Furthermore, grip strength, pinch and key grip strength were measured.
RESULTS: We found no differences in muscle strength for the dominant and non-dominant hand, except for a stronger key grip strength of the dominant hand in patients with Charcot-Marie-Tooth disease type II.
CONCLUSION: In our population, the dominant hand of patients with Charcot-Marie-Tooth disease type I and II was equally strong as the non-dominant hand, suggesting that there is no presence of overwork weakness in the dominant hand in our group of patients. This implies that patients with Charcot-Marie-Tooth disease do not have to limit the use of their hands in daily life in order to prevent muscle strength loss.
Key words: Charcot-Marie-Tooth disease, Hereditary Motor and Sensory Neuropathies, rehabilitation, hand strength.
J Rehabil Med 2009; 41: 32–34
Correspondence address: Madlenka van Pomeren, Rehabilitation Center Sophia Revalidatie, Reinier de Graafweg 1, 2625 AD Delft, The Netherlands. E-mail: m.vanpomeren@sophiarevalidatie.nl
Submitted November 7, 2007; accepted July 9, 2008
INTRODUCTION
Charcot-Marie-Tooth (CMT) disease is characterized by loss of muscle strength and sensory function that generally progresses from distal to proximal in the extremities. It has been suggested that patients with neuromuscular disorders such as post-polio syndrome and CMT disease may have overwork weakness due to overloading of the muscles, which may be a possible cause of disease progression (1–4). However, other studies have suggested beneficial effects of training in patients with CMT disease (5–7).
Vinci et al. (1) hypothesized that, in patients with CMT disease, overwork causes additional weakness in the dominant hand, based on data from 106 patients showing that the non-dominant hand was significantly stronger than the dominant hand. This finding of Vinci et al. (1) contrasts with that of Carter et al. (8) who found that strength was equal in both hands in 53 patients with CMT disease, and is also in contrast with findings in healthy subjects (9).
The hypothesis of overwork weakness is important for training guidelines and advice on daily life activity patterns in patients with CMT disease, since it would suggest that patients should limit the use of their muscles to prevent additional weakness. Therefore, the objective of this study was to determine if this hypothesis could be verified in our population of patients with CMT disease type I and II using hand dynamometers and the Medical Research Council (MRC) scale.
METHODS
Participants
Subjects were recruited at the Rehabilitation Department of the Erasmus Medical Center in Rotterdam, the Netherlands. All patients were diagnosed with CMT disease based on clinical and electrophysiological examination and were between 18 and 80 years of age. Patients were excluded if they had co-morbidity that could interfere with muscle strength or hand function or if one or both hands had been operated on. Written informed consent was obtained from all subjects.
Thirty-three of the 34 (97%) eligible patients with CMT disease type I or II agreed to participate in this study. Data from 5 patients (2 type I and 3 type II) were excluded from analysis because one or both hands had been operated on. Thus, 28 subjects were included in the analysis. Forty-six percent of these participants had type I CMT disease (5 male, 8 female) and 54% had type II CMT disease (8 male, 7 female). The mean age of the participants was 42 years and 89% were right-handed (10 type I and 15 type II).
Measurements
All measurements were performed by the same researcher (BTJvG). Manual muscle strength testing and Rotterdam Intrinsic Hand Myometer (RIHM) dynamometer measurements were performed on 3 intrinsic muscle groups: thumb palmar abduction, index finger abduction, and little finger abduction. Muscle strength was scored based on the MRC scale, adapted for assessment of hand muscles (10, 11). Details of the RIHM and the measurement protocol have been described elsewhere (12, 13). When the MRC grade was less than 3, RIHM dynamometry was not possible because no resistance could be given and a “0” score was recorded. Maximal isometric contraction of grip strength, pinch and key grip strength were measured using Lode dynamometers (14) following the recommendations by the American Society of Hand Therapists (15). For all measurements, the mean of 3 maximum voluntary contractions was calculated.
Statistical analysis
A paired samples t-test was used to compare intrinsic hand muscle strength, grip strength, pinch and key grip between the dominant and non-dominant hand. A non-parametric Wilcoxon signed-ranks test was used to compare MRC grades. In addition, for comparison with the study of Vinci et al. (1), we also used the parametric paired samples t-test to compare the MRC grades despite the ordinal characteristic of the MRC scale. A p-value ≤ 0.05 determined significance.
RESULTS
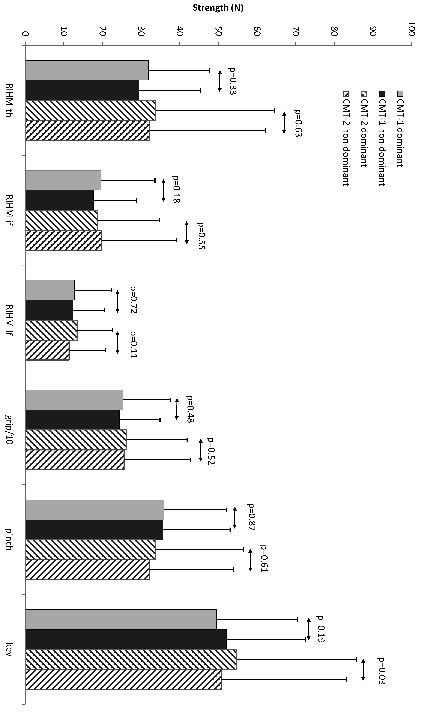
We found no significant difference between muscle strength of the dominant and non-dominant hand as measured with the MRC scale in either type of patients with CMT disease using both non-parametric (Wilcoxon signed-ranks) and parametric test (paired samples t-test) (Table I). Similarly, we found no significant difference between the RIHM measurements of the dominant and non-dominant abduction of thumb, index finger and little finger (Fig. 1). Finally, no significant differences were found in grip strength and pinch grip strength between both hands. Only key grip strength in patients with CMT disease type II was significantly higher in the dominant hand compared with the non-dominant hand (Fig. 1).
| Table I. Hand strength of the dominant and non-dominant hand in patients with type I and II Charcot-Marie-Tooth (CMT) disease measured with the Medical Research Council scale. Values indicate median, mode, mean and interquartile range (25th, 50th and 75th quartile) |
| | Dominant | | Non-dominant |
| Median | Mode | Mean | Quartiles 25/50/75 | Median | Mode | Mean | Quartiles 25/50/75 |
| CMT disease type I |
| Thumb abduction | 4 | 4 | 4.23 | 4/4/5 | 4 | 4 | 4.15 | 4/4/5 |
| Index finger abduction | 4 | 4 | 3.77 | 4/4/4 | 4 | 4 | 3.62 | 3.5/4/4 |
| Little finger abduction | 4 | 4 | 3.77 | 4/4/4 | 4 | 4 | 3.92 | 4/4/4 |
| CMT disease type II |
| Thumb abduction | 4 | 5 | 3.80 | 3/4/5 | 4 | 4 | 3.73 | 4/4/5 |
| Index finger abduction | 4 | 4 | 3.60 | 3/4/5 | 4 | 4 | 3.53 | 3/4/5 |
| Little finger abduction | 4 | 4 | 3.73 | 4/4/5 | 4 | 4 | 3.67 | 3/4/5 |
Fig. 1. Muscle strength (mean and standard deviation) of dominant and non-dominant hand in type I and II CMT patients using the Rotterdam Intrinsic Hand Myometer (RIHM) and the grip-, pinch- and key dynamometers. “RIHM-th” refers to thumb abduction, “RIHM-if” to abduction of the index finger and “RIHM-lf” to abduction of the little finger. Grip strength values were divided by 10 for reasons of comparison and representation.
DISCUSSION
In the present study, we wanted to test if we could verify in our population whether muscle strength in patients with CMT disease is lower in the dominant hand compared with the non-dominant hand, as has been reported recently by Vinci et al. (1) and which may indicate the presence of overwork weakness in the dominant hand. We found no significant differences between the dominant and non-dominant hand when using a number of different strength measurements of the hand, except for a stronger key grip of the dominant hand in patients with CMT disease type II.
Our results do not support the overwork weakness hypothesis in patients with CMT disease. In contrast, our results are similar to those of Carter et al. (8), who presented equal grip and pinch strength in both hands in 53 patients with CMT disease. A number of differences between our study and that of Vinci et al. (9) should be noted. In our study, we used a 5-point MRC scale, whereas Vinci et al. used a modified 14-point MRC scale with unknown reliability. Also, we used dynamometry in addition to MRC grading. Compared with Vinci et al. (1), our study group was relatively small. However, we did not find any trends to confirm the findings of Vinci. Finally, while differences between both studies could be related to the severity of muscle weakness, Vinci et al. (1) did not specify the level of muscle weakness in their population. In our study, the median of the MRC grade was 4. When we examined only our subjects with an MRC grade of 3 or less, we again did not find differences between both hands, suggesting no effect of the level of muscle weakness.
As mentioned earlier, the discussion on overwork weakness has important implications for advice on training and on activities of daily living in patients with CMT disease. A randomized clinical trial on lower extremities strength training in patients with myotonic dystrophy and CMT disease (5) reported no harmful effects and described a small increase in muscle strength. Furthermore, despite limited evidence, 2 systematic reviews on exercise therapy in peripheral neuropathy (16) and neuromuscular diseases (17) also suggest no risk of overwork weakness. In summary, the present study suggests no presence of overwork weakness in our patients with CMT disease type I or II, providing no ground for limiting the activities of these patients. Without the presence of such proof, it may be more appropriate to advise patients with CMT disease to keep an active role in their community, sports and work.
REFERENCES
1. Vinci P, Esposito C, Perelli SL, Antenor JV, Thomas FP. Overwork weakness in Charcot-Marie-Tooth disease. Arch Phys Med Rehabil 2003; 84: 825–827.
2. Fowler WM Jr. Importance of overwork weakness. Muscle Nerve 1984; 7: 496–499.
3. Peach FE. Overwork weakness with evidence of muscle damage in a patient with residual paralysis from polio. Arch Phys Med Rehabil 1990; 71: 348–350.
4. Borg K, Borg J, Edstrom L, Grimby L. Effects of excessive use of remaining muscle fibers in prior polio and LV lesion. Muscle Nerve 1988; 11: 1211–1218.
5. Lindeman E, Leffers P, Spaans F, Drukker J, Reulen J, Kerckhoffs M, et al. Strength training in patients with myotonic dystrophy and hereditary motor and sensory neuropathy: a randomized clinical trial. Arch Phys Med Rehabil 1995; 76: 612–620.
6. Lindeman E, Spaans F, Reulen J, Leffers P, Drukker J. Progressive resistance training in neuromuscular patients. Effects on force and surface EMG. J Electromyogr Kinesiol 1999; 9: 379–384.
7. Chetlin RD, Gutmann L, Tarnopolsky M, Ullrich IH, Yeater RA. Resistance training effectiveness in patients with Charcot-Marie-Tooth disease: recommendations for exercise prescription. Arch Phys Med Rehabil 2004; 85: 1217–1223.
8. Carter GT, Abresch RT, Fowler WM Jr, Johnson ER, Kilmer DD, McDonald CM. Profiles of neuromuscular diseases. Hereditary motor and sensory neuropathy, types I and II. Am J Phys Med Rehabil 1995; 74 Suppl 5: S140–S149.
9. Incel NA, Ceceli E, Durukan PB, Erdem HR, Yorgancioglu ZR. Grip strength: effect of hand dominance. Singapore Med J 2002; 43: 234–237.
10. Brandsma JW, Schreuders TA. Sensible manual muscle strength testing to evaluate and monitor strength of the intrinsic muscles of the hand: a commentary. J Hand Ther 2001; 14: 273–278.
11. Brandsma JW, Schreuders TA, Birke JA, Piefer A, Oostendorp R. Manual muscle strength testing: intraobserver and interobserver reliabilities for the intrinsic muscles of the hand. J Hand Ther 1995; 8: 185–190.
12. Schreuders TA, Roebroeck ME, Jaquet JB, Hovius SE, Stam HJ. Measuring the strength of the intrinsic muscles of the hand in patients with ulnar and median nerve injuries: reliability of the Rotterdam Intrinsic Hand Myometer (RIHM). J Hand Surg [Am] 2004; 29: 318–324
13. Schreuders TA, Roebroeck ME, Jaquet JB, Hovius SE, Stam HJ. Long-term outcome of muscle strength in ulnar and median nerve injury: comparing manual muscle strength testing, grip and pinch strength dynamometers and a new intrinsic muscle strength dynamometer. J Rehabil Med 2004; 36: 273–278.
14. Schreuders TA, Roebroeck ME, Goumans J, van Nieuwenhuijzen JF, Stijnen TH, Stam HJ. Measurement error in grip and pinch force measurements in patients with hand injuries. Phys Ther 2003; 83: 806–815.
15. Casanova JS, editor. American Society of Hand Therapists (ASHT). Clinical Assessment Recommendations. 2nd edn. Garner, NC: ASHT; 1992.
16. White CM, Pritchard J, Turner-Stokes L. Exercise for people with peripheral neuropathy. Cochrane Database Syst Rev 2004; 4: CD003904.
17. Cup EH, Pieterse AJ, Ten Broek-Pastoor JM, Munneke M, van Engelen BG, Hendricks HT, et al. Exercise therapy and other types of physical therapy for patients with neuromuscular diseases: a systematic review. Arch Phys Med Rehabil 2007; 88: 1452–1464.