OBJECTIVE: The aims of this study were: to follow the course of recovery of motor function following acute stroke, as assessed by the Physiotherapy Clinical Outcome Variables Scale (COVS), and; to investigate the ability of this instrument to predict length of hospital stay, discharge destination and future home facility.
METHODS: In this prospective longitudinal study, COVS was registered at admission and discharge from an acute stroke unit and at 3 months post-stroke onset.
SUBJECTS: Sixty subjects were recruited consecutively from a sample of patients after first-ever acute stroke, and of these, 50 received follow-up assessment.
Outcome variables: Length of hospital stay, discharge destination and home facility 3 months post-stroke.
RESULTS: The overall COVS scores increased significantly during the 3-month follow-up. The admission COVS score correlated negatively with length of stay. A cut-off at 50 points and 41 points could predict discharge destination and future home facility, respectively.
CONCLUSION: COVS measures improvements and can predict length of hospital stay, discharge destination and future home facility. Thus, it could be used in early prediction for effective planning of the acute stroke unit services and efficient discharge planning.
Key words: stroke, acute stroke unit, prognosis, outcome assessment, mobility.
J Rehabil Med 2008; 40: 524–528
Correspondence address: Elisabeth Ekstrand, Department of Neurology, Malmö University Hospital, Entrance 53, SE-205 02 Malmö, Sweden. E-mail: elisabeth.ekstrand@skane.se
Submitted September 13, 2007; accepted March 3, 2008
*Part of this paper was presented as a poster on the Sjukgymnastdagarna, October 11, 2007 in Stockholm, Sweden.
INTRODUCTION
Stroke is one of the most common causes of death and disability worldwide (1); in the acute stage of stroke, nearly half of all surviving patients have severe remaining functional problems (2). Stroke is a complex condition with a variety of symptoms (3), and research has shown that comprehensive stroke units, i.e. a multidisciplinary intervention including rehabilitation in the acute setting, can enhance survival and independence in functional activities, as well as reduce the length of inpatient stay and need for institutional care (4).
In Malmö, the third largest city in Sweden with 270,000 inhabitants, patients after stroke receive acute stroke treatment and inpatient rehabilitation within the comprehensive stroke unit at Malmö University Hospital, regardless of patient age or stroke severity. Patients are discharged to their homes, with early supported discharge (5) if needed, or to municipal nursing homes, some of which are aimed at rehabilitation for the elderly, if discharge to home is not possible. A few younger patients with major disabilities are referred to a rehabilitation clinic in a neighbouring county.
The greatest recovery from neurological deficits occurs during the first 3 months after the onset of symptoms, and this is the optimal time for rehabilitation (6). During the most acute phase, it is important to identify patients who are most likely to benefit from rehabilitation, in order to select the appropriate rehabilitation setting and to facilitate discharge planning. Early prediction of recovery is thus a key issue for optimizing the outcome of rehabilitation and the utilization of healthcare resources.
There is a large degree of consensus in national and international guidelines on which issues to address regarding patients after stroke; however, there are no generally accepted instruments to be used in stroke disability assessments (7). Disability assessments can be classified at different levels according to the framework of the International Classification of Functioning, Disability and Health (ICF) (8). Stroke assessments are affected by the complex relationship between impairments, activity limitations and restrictions in participation. Measurements at the level of activity have the advantage of being relevant to the patient, reasonably objective and simple to perform (9).
Previous research indicates that instruments for determining levels of activity, such as gait speed measurements, the Berg Balance Scale and the Functional Independence Measure (FIMTM), can predict discharge outcome in acute stroke (10–12). Many other prognostic factors in acute stroke, including age, gender, level of social support and type of lesion, have been proven significant (13–15).
The Physiotherapy Clinical Outcome Variables Scale (COVS) is a functional mobility scale for the assessment of patients with neuromuscular and musculoskeletal disorders (16). In stroke it has been found to correlate well with the FIMTM (rs = 0.82) and Barthel Index (rs =0.71) (17). It has also shown sensitivity to changes in acute neurology over time in a sample of a variety of diagnoses, such as stroke, traumatic brain injury, multiple sclerosis and brain tumour (18).
The aim of this study was to follow the course of recovery of motor function in acute stroke assessed by COVS and to investigate the ability of COVS to predict length of hospital stay, discharge destination and the future home facility 3 months post-stroke onset.
METHODS
Subjects
Sixty patients 50 years of age and older were recruited consecutively from a sample group of patients after first-ever stroke (cerebral infarction (I61) and intracerebral haemorrhage (I63)) treated at the stroke unit in the Department of Neurology at Malmö University Hospital between 1 September 2005 and 28 February 2006.
Patients with a severe functional disorder before stroke (inability to stand and walk and/or daily need of assistance in personal activities of daily living) or a disease or disorder in addition to the current stroke that considerably affected functional recovery and survival were not included. Exclusion criteria after stroke were non-remaining motor and/or balance deficits within 4 days after the event and inability to understand test instructions, even after repeated gesture cues. Patients who suffered a recurrent stroke or other medical condition affecting functional recovery within 3 months after discharge from the stroke unit were excluded from the follow-up.
Procedure
Age at stroke onset, gender, social situation (living alone, living with another person), type of stroke and side of lesion (right, left, unknown) were registered at admission. COVS scores were registered within 4 days from onset of stroke (admission COVS) and within 3 days prior to discharge (discharge COVS) by the treating physiotherapist. The patients were followed up with COVS after 3 months by the first author (EE), either at the hospital outpatient clinic or in their residence.
COVS has been proven valid and reliable in stroke assessment (16, 18, 19). It has been translated into Swedish and tested for inter-rater reliability with close correspondence (20). In this study the Swedish version is used. COVS consists of 13 items (rolling, transfers, ambulation and arm function), and each task is clinically assessed on an ordinal scale from 1 to 7. The overall score ranges from 13 to 91. A higher score denotes greater mobility and functional status (16). In this study the item “vertical transfers” (transfers from lying supine to standing up) was not assessed and was instead set to 1 point for all patients.
Length of stay (LOS) and discharge destination (DD) were registered as outcome variables at discharge from the acute stroke unit and future home facility at 3 months post-stroke. LOS was measured as the number of days from admission to discharge from the acute stroke unit, with the day after admission counted as day 1. Four possible discharge destinations and home facilities post-stroke were identified: own home without community support, own home with community support, rehabilitation clinic and municipal nursing home.
Statistical methods
To assess differences in sample characteristics between independent groups, the non-parametric Mann-Whitney U test was used. The ordinal data in the COVS scores between admission, discharge and follow-up were given as differences in median score. The differences in median scores were statistically evaluated by the non-parametric Wilcoxon signed-rank test. LOS was given as the median value and the variation with quartiles due to skewed data in the LOS. Differences in the median LOS and median admission COVS between the patients discharged home and the patients not discharged home were analysed using the Mann-Whitney U test. At the follow-up, differences in the median admission COVS between the patients living at home and the patients not living at home were also analysed using the Mann-Whitney U test.
The non-parametric Spearman rank correlation test was used to assess relationships between admission COVS and the outcome variables and between patient characteristics and the outcome variables. A logarithmic transformation of LOS (logLOS) produced a normal distribution of the LOS data. A linear regression analysis with logLOS as the dependent variable and admission COVS as the independent variable was performed to assess the predictability of logLOS from the admission COVS. The predicted logLOS values were then transformed back to obtain the predicted LOS. A multiple regression analysis was performed with the dependent variable logLOS and the independent variables admission COVS, age, gender, social situation, type of stroke and side of lesion to assess the impact of LOS in the multivariate model.
The ordinal data in DD and future home facility was reduced to 2 categories, and using a binary logistic regression analysis and receiver operating characteristics (ROC) curve analysis, a cut-off value was determined to assess the predictability of DD and future home facility from the admission COVS. A ROC curve is a graphical approach to determine the cut-off value that maximizes the sum of the sensitivity and specificity. Probability values less than 0.05 were considered statistically significant. Data were analysed using the SPSS 12.0.1 for Windows. The procedures in the present study were in accordance with the standards of the ethics committee responsible.
RESULTS
Sample characteristics
Sixty patients were enrolled in the study and assessed at admission and discharge. Seven patients were excluded from the follow-up: 3 patients with recurrent stroke, 2 with other acute medical conditions, one with ankle fracture, one with acute confusion and 3 who chose not to participate. Thus, 50 patients participated in the follow-up at 3 months post-stroke onset. The age of the 60 patients ranged from 54 to 97 years (mean 75.9 years), and 26 (43%) of the patients were women. There were no differences between men and women with respect to age, social situation, type of stroke and side of lesion. There were no differences between the 50 patients who were followed up at 3 months post-stroke and the initial 60 patients concerning age, gender, social situation, type of stroke, side of lesion, admission COVS, LOS or DD.
Admission COVS was assessed within 4 days post-stroke onset (mean 1.9 days, standard deviation (SD) 1.2), and discharge COVS was assessed within 3 days prior to discharge (mean 0.3 days, SD 0.7). The follow-up COVS was assessed 3 months post-stroke onset (mean 13.0 weeks, SD 0.9). The median overall COVS score increased significantly from admission to discharge (p < 0.001), from discharge to follow-up (p < 0.001) and from admission to follow-up (p < 0.001) (median 64.0 at admission, median 71.5 at discharge, median 79.5 at follow-up, n = 50).
Length of stay (LOS)
The median LOS in the stroke unit for the 60 patients was 12.0 days and ranged from 2 to 48 days (interquartile range 6.0–18.7 days). Median LOS was significantly shorter for those discharged home compared with those not discharged home (median 8.0 days and 21.0 days, respectively, p < 0.001, n = 60). The admission COVS scores, as well as all subscales except for “right-arm-function”, showed significant negative correlation with LOS (p < 0.001, n = 60). There were no significant correlations between LOS and age, gender, social situation, type of stroke or side of lesion.
In a linear regression analysis between logLOS and admission COVS, LOS could be determined by the equation LOS = 10^(1.705–0.012*admission COVS), (R2 = 0.51, p < 0.001, n = 60). The equation gives that each additional point on the admission COVS score decreases the LOS by 2.7%, and a 10-point increase in the COVS score decreases the LOS by 24.1%. A multiple linear regression analysis, where logLOS was the dependent variable and admission COVS, age, gender, social situation, type of stroke and side of lesion were independent variables, showed that admission COVS was the strongest predictor for logLOS (p < 0.001).
Discharge destination (DD)
Forty-one patients (68.3%) were discharged home from the stroke unit with or without support from the community and 19 patients (31.7%) were discharged to a rehabilitation clinic or a municipal nursing home. Of the patients discharged home, 20 were provided with early supported discharge from the stroke unit, and 4 were provided with rehabilitation in primary care. Age, gender, social situation, type of stroke and side of lesion were not significantly correlated with DD. The admission COVS score and all the subscales, except for the “right-arm-function” subscale, showed significant negative correlations with DD (p < 0.001, n = 60).
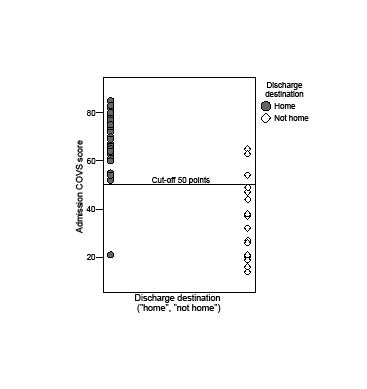
The patients discharged home had significantly higher median admission COVS scores compared with those not discharged home (median 69.0 and 32.0, respectively, p < 0.001, n = 60). Using a binary logistic regression analysis and ROC curve analysis, a cut-off was established at 50 points for admission COVS, and this cut-off correctly predicted the DD from the acute stroke unit of 93.3% of the patients (p < 0.001, n = 60; Fig. 1).
Fig. 1. Admission Clinical Outcome Variables Scale (COVS) score plotted against discharge destination (DD) with a cut-off at 50 points (n = 60). The overall percentage of correctly predicting DD using a cut-off at 50 points was 93.3% (p < 0.001).
Future home facility
Fifty patients were followed up at 3 months post-stroke onset. Forty-one patients (82.0%) were living at home and 9 patients (18.0%) were still in a rehabilitation clinic or a municipal nursing home. The factors age, gender, social situation, type of stroke and side of lesion, did not correlate significantly with the home facility 3 months post-stroke. The admission COVS score showed significant negative correlations with home facility at the follow-up (p < 0.001, n = 50). All the subscales, except for the “right-arm-function” subscale, also showed significant negative correlations with the home facility (p = 0.024 – p < 0.001, n = 50).
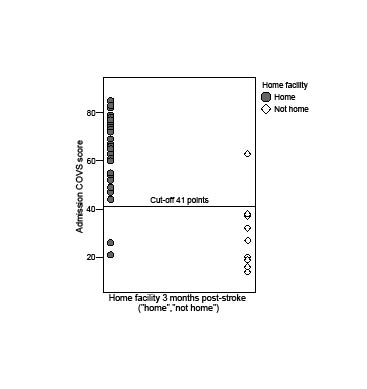
The admission COVS scores for those who lived at home at 3 months post-stroke onset were significantly higher than for those who not had been able to return home during that time (median 66.0 and 27.0, respectively, p < 0.001, n = 50). Using a binary logistic regression analysis and ROC curve analysis, a cut-off was set at 41 points for the admission COVS. This cut-off correctly predicted the home facility at 3 months post-stroke of 92.0% of the patients (p < 0.001, n = 50; Fig. 2).
Fig. 2. Admission Clinical Outcome Variables Scale (COVS) plotted against the home facility 3 months post-stroke with a cut-off at 41 points (n = 50). The overall percentage of correctly predicting future home facility using a cut-off at 41 points was 92.0% (p < 0.001).
DISCUSSION
In this study, age, gender and social support did not correlate with outcome assessments. Although the results in the literature are contradictory (13–15), older age and female sex have been reported as negative factors in recovery after stroke. Previous studies have also shown the importance of social support as a predictor of discharge destination (12, 13). The sample in this study excluded patients who were dependent in personal activities of daily living or who had a major disease or disorder prior to the stroke. This might have led to the exclusion of more older women, since women are, on average, 5 years older than men at onset of stroke and more women are dependent in personal activities of daily living prior to stroke (21). Thus, the results of this study indicate that older age and female sex per se are not associated with longer LOS and higher dependence. These findings suggest that prognostication should rely on functional status rather than age, and older patients should not be withheld rehabilitation merely because of their advanced age.
The patients improved as expected, and in accordance with earlier findings, COVS scores showed improvements in the acute setting (16). All COVS subscales were correlated with outcome assessments except for the “right arm function” subscale. This is consistent with earlier findings that the arm-function subscale of the COVS is not very responsive to changes in acute neurology (16). The strong correlations of admission COVS with LOS and DD indicate that transfers and ambulation are factors that have a strong impact in the acute phase. In previous research, gait speed has been proven to correlate with DD in acute stroke, both in a timed 5-metre walking test at comfortable and maximal speeds (10) and in a 2-minute timed walking test at a comfortable speed (11). Previous studies have shown that admission Berg Balance test scores, as well as admission FIMTM scores, correlate moderately with in-hospital LOS and DD in acute stroke (12). In comparison with the Berg Balance Scale and gait speed measurements, which are restricted mainly to ambulating patients, COVS is an instrument that can be used to assess a broad stroke population. FIMTM is a more comprehensive multidisciplinary instrument and tends to be difficult to administer in the very acute phase.
In the linear regression analysis, approximately 51% of the total variance in LOS could be explained by admission COVS. The binary logistic regression analyses showed that cut-offs at 50 points and 41 points on the admission COVS could predict with high accuracy the DD and future home facility, respectively. These findings indicate that admission COVS scores could be valuable in facilitating discharge planning in clinical practice, thus optimizing the services of an acute stroke unit. This prognostic capacity of COVS could be important from a health economic perspective, since acute hospital care is one of the major costs in stroke (22).
Methodological concerns of this study may be that assessment at admission was made within 4 days of onset of stroke. During these first days the patients may have improved functionally, and it is thus possible that late measurements do not reflect the whole degree of recovery. It is also possible that learning effects may have contributed to improvements, especially for those patients who had a short LOS, since the patients performed the test on 3 occasions. In this study the vertical transfer item in the COVS was set to 1 point for all patients. In the very acute phase, many of the patients hesitate to get down on the floor. If they are forced to accomplish the “vertical transfer”, it may affect their trust in the therapist and negatively affect the rehabilitation outcome. In clinical practice in the acute ward, it seems that this item can therefore be omitted without affecting the efficiency of the instrument.
The study population was recruited from an unselected sample of patients after stroke that is representative of the population of patients after first-time stroke; these patients were independent in personal activities of daily living prior to stroke and living in an urban area in the south of Sweden. Therefore, the results of this study may be generalized only to populations similar to the sample in this study, since previous research has shown that the factors and measurements that predict functional recovery depend, to a large extent, on the population and assessment methods chosen (13).
In conclusion, with the limited resources available in the healthcare system and an increasing number of stroke patients resulting in fewer acute care and rehabilitation beds, accurate predictive assessment instruments are essential for optimizing efficiency. This study indicates that the COVS is an assessment instrument at the level of activity that covers key factors in the very acute phase. COVS measures improvements and can predict LOS, DD and future home facility at 3 months after stroke. This instrument could therefore be used for early prediction, to enable effective planning of the services of the acute stroke unit and efficient discharge.
Acknowledgements
We wish to thank the physiotherapists at the Stroke Unit, Department of Neurology at UMAS, for their help in recruiting and assessing the patients. This study was supported by grants from Stroke-Riksförbundet and Vårdakademin, UMAS.
REFERENCES
1. WHO.int. Geneva: World Health Organization [updated 2004; cited 2007 June 5]. Available from: http://www.who.int/topics/cerebrovascular_accident/en/
2. Lawrence ES, Coshall C, Dundas R, Stewart J, Rudd AG, Howard R, et al. Estimates of the prevalence of acute stroke impairments and disability in a multiethnic population. Stroke 2001; 32: 1279–1284.
3. Kolb B, Whishaw I, editors. Fundamentals of human neuropsychology. New York: Worth Pub. Co.; 2003.
4. Stroke Unit Trialists’ Collaboration. Organised inpatient (stroke unit) care for stroke in Cochrane Library 2002; (1): CD000197. Update of Cochrane Database Syst Rev. 2000; (2): CD000197.
5. Langhorne P, Taylor G, Murray G, Dennis M, Anderson C, Bautz-Holter E, et al. Early supported discharge services for stroke patients a meta-analysis of individual patients’ data. Lancet 2005; 365: 501–506.
6. Van Peppen RPS, Kwakkel G, Wood-Dauphinee S, Hendriks HJM, Van der Wees PhJ, Dekker J. The impact of physical therapy on functional outcomes after stroke: what’s the evidence? Clin Rehabil 2004; 18: 833–862.
7. Geyh S, Cieza A, Schouten J, Dickson H, Frommelt P, Omar Z, et al. ICF Core Sets for stroke. J Rehabil Med 2004; 44: 135–141.
8. International Classification of Functioning, Disability and Health (ICF). Geneva: WHO 2001. Available from: http://www.who.int/classifications/icf/site/icftemplate.cfm
9. Duncan PW, Jorgensen HS, Wade DT. Outcome measures in acute stroke trials: a systematic review and some recommendations to improve practice. Stroke 2000; 31: 1429–1438.
10. Salbach NM, Mayo NE, Higgins J, Ahmed S, Finch LE, Richards CL. Responsiveness and predictability of gait speed and other disability measures in acute stroke. Arch Phys Med Rehabil 2001; 82: 1204–1212.
11. Meheroz HR, Blau A. Admission ambulation velocity predicts length of stay and discharge disposition following stroke in an acute rehabilitation hospital. Neurorehabil Neural Repair 2005; 19: 20.
12. Wee JYM, Bagg SD, Palpeu A. The Berg Balance Scale as a predictor of length of stay and discharge destination in an acute stroke rehabilitation setting. Arch Phys Med Rehabil 1999; 80: 448–452.
13. Kwakkel G, Wagenaar RC, Kollen J, Lankhorst G. Predicting disability in stroke – a critical review of the literature. Age Ageing 1996; 25: 479–489.
14. Meijer R, van Limbeek J, Peusens G, Rulkens M, Dankoor K, Vermulen M, et al. The stroke unit Discharge Guideline, a prognostic framework for the discharge outcome from the hospital stroke unit. A prospective cohort study. Clin Rehabil 2005; 19: 770–778.
15. Glader EL, Stegmayr B, Norrving B, Terent A, Hulter-Asberg K, Wester PO, et al. Sex differences and outcome after stroke: a Swedish national perspective. Stroke 2003; 34: 1970–1975.
16. Seaby L, Torrance G. Reliability of a physiotherapy functional assessment used in a rehabilitation setting. Physiother Can 1989; 41: 264–271.
17. Hajek VE, Gagnon S, Ruderman JE. Cognitive and functional assessments of stroke patients: an analysis of their relation. Arch Phys Med Rehabil 1997; 78: 1331–1337.
18. Barclay-Gooddard R. Physical function outcome measurement in acute neurology. Physiother Can 2000; 52: 138–145.
19. Eng JJ, Rowe SJ, McLaren LM. Mobility status during inpatient rehabilitation: a comparison of patients with stroke and traumatic brain injury. Arch Phys Med Rehabil 2002; 83: 483–490.
20. Hasselgren-Nyberg L, Omgren M, Nyberg L, Gustafson Y. S-COVS. Den svenska versionen av Physiotherapy Clinical Outcome Variables [S-COVS. The Swedich version of Physiotherapy Clinical Outcome Variables Scale]. Nordisk fysioterapi 1997; 1: 109–113 (in Swedish).
21. Riks-stroke.org. Umeå: Riks-stroke. The national Stroke Register in Sweden. c 1994 [updated 2006 October 25; cited 2006 October 30]. Available from: http://www.Riks-stroke.org.
22. Ekman M. Economic evidence in stroke: a review. Eur J Health Econom 2004; Suppl 1: 74–83.