OBJECTIVE: To determine whether there is a difference between patients with a cerebral infarction and those with an intracerebral haemorrhage with respect to the development of independence in activities of daily living over the first year post-stroke.
METHODS: Patients after first-ever stroke who were admitted to an inpatient rehabilitation programme were included. The study had a longitudinal design and measurements took place at admission, 8, 10, 12, 26 and 52 weeks post-stroke. The relationship between the development over time of activities of daily living independence, measured by the Barthel Index, and type of stroke was analysed using Generalized Estimating Equations.
RESULTS: A total of 229 patients with cerebral infarction and 45 with intracerebral haemorrhage were included. From 12 to 26 weeks post-stroke, patients with cerebral infarction showed a significantly faster recovery. The time window for recovery was more restricted for patients with intracerebral haemorrhage; a statistically significant increase in activities of daily living was found until 10 weeks post-stroke in patients with intracerebral haemorrhage, whereas patients with cerebral infarction showed statistically significant recovery until 26 weeks post-stroke.
CONCLUSION: The differences in activities of daily living recovery between patients with cerebral infarction and those with intracerebral haemorrhage should be taken into consideration in rehabilitation management.
Key words: rehabilitation, cerebrovascular accident, cerebral infarction, intracerebral haemorrhage, activities of daily living.
J Rehabil Med 2008; 40: 487–489
Correspondence address: V. P. M. Schepers, Center of Excellence for Rehabilitation Medicine Utrecht, Rehabilitation Center De Hoogstraat, Rembrandtkade 10, 3583 TM Utrecht, The Netherlands. E-mail address: V.Schepers@dehoogstraat.nl
Submitted November 27, 2007; accepted January 29, 2008
Introduction
Strokes can be divided into those with an ischaemic and those with a haemorrhagic origin. As their pathophysiological mechanisms differ, this distinction could be expected to have consequences for recovery and outcome. In the hospital population, patients after haemorrhagic stroke generally have more severe neurological impairments during the acute phase than patients after ischaemic stroke and the mortality rate from haemorrhagic stroke is higher than that from ischaemic stroke (1). From the perspective of rehabilitation medicine, it is more important to know what the differences are in terms of functional recovery. Detailed knowledge on the precise course of functional recovery, and the differences in this respect between ischaemic and haemorrhagic strokes, would be of practical use to physicians, as it would enable them to develop a more differentiated prognosis and rehabilitation programme.
Earlier studies (2–8) of functional recovery in the rehabilitation setting have assessed activities of daily living (ADL) scores, using the Functional Independence Measure or the Barthel Index (BI), at rehabilitation admission and discharge. The rate of recovery was then determined by dividing the gain in ADL scores by length of stay at the rehabilitation centre. Some studies (4, 5) showed that patients with an intracerebral haemorrhage (ICH) had a higher rate of recovery than patients with a cerebral infarction (CI), whereas other studies (2, 6, 8) found no differences in rate of recovery. Comparisons of the findings of these studies are complicated by the fact that they did not use fixed measurement times, but the moments of admission and discharge, which can vary. Consequently, both the interval from stroke onset to admission and the length of stay differed greatly between the studies, with periods of inpatient rehabilitation varying from, on average, the 2nd to the 7th week post-stroke in the study by Ween et al. (6), to, on average, the 10th until the 27th week post-stroke in the study by Inouye et al. (7). Improving our understanding of the development of ADL independence over time thus requires longitudinal studies in the rehabilitation setting with serial assessments at fixed moments in time.
Our study aimed to determine whether there is a difference between patients with CI and those with ICH in a rehabilitation setting regarding the development of ADL independence over the first year post-stroke.
Methods
Participants
Subjects were selected from patients after stroke consecutively admitted to 4 Dutch rehabilitation centres according to the following inclusion criteria: (i) admittance for inpatient rehabilitation, (ii) a first-ever stroke due to CI or ICH, (iii) a one-sided supratentorial lesion, and (iv) age over 18 years. Exclusion criteria were: (i) disabling co-morbidity (pre-stroke BI score below 18); and (ii) inability to speak Dutch. The study was approved by the medical ethics committees of University Medical Center Utrecht and the participating rehabilitation centres.
Procedure
At the start of inpatient rehabilitation, patients were asked by their rehabilitation physician whether they were willing to participate in the study. Informed consent was obtained from all patients. The assessments took place as soon as possible after admission, at 8, 10, 12, 26, and 52 weeks post-stroke. All assessments were carried out by trained research assistants.
Measures
Stroke was defined according to the World Health Organization criteria: rapidly developing clinical signs of focal disturbance of cerebral function, lasting more than 24 h, with no apparent cause other than vascular origin. Patients were classified as having a CI or an ICH based on information from the referral letter from the hospital, including the imaging findings. Data on demographic variables were derived from medical charts. The Motricity Index (MI) (9) is a brief assessment method for motor impairment. The score for the level of hemiparesis varies from 0 (paralysis) to 100 (normal strength). Cognitive impairments were assessed by the Mini Mental State Examination (MMSE) (10), a widely used brief screening instrument. A score below 24 on the MMSE indicates the presence of cognitive impairments. The level of ADL independence was measured with the BI (11), with scores ranging from 0 to 20.
Statistical analyses
The relationship between the development of ADL independence over time and the type of stroke was analysed using Generalized Estimating Equations (GEE), using STATA version 7 (12). GEE is a statistical technique for longitudinal data analysis, which takes into account that the repeated observations within one subject are not independent. A correction for these within-subject correlations was made by using an exchangeable working correlation structure. Since we assumed that the development of ADL independence over time was a non-linear function, time was added to the model as a categorical variable, modelling each time-interval separately. For each time-interval, we analysed whether the ADL independence developed differently over time in the 2 types of stroke, by including interaction terms in the model.
Results
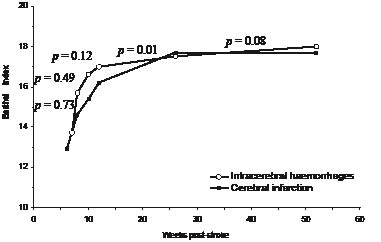
A total of 274 patients were included: 229 with CI and 45 with ICH. At 1-year post-stroke, 7 patients had died, 13 had suffered a recurrent stroke, and 20 were lost to follow-up. Baseline characteristics (Table I) were not significantly different for patients with CI and ICH, except for the time-interval between stroke onset and admission to the inpatient rehabilitation programme. The patients with CI were admitted at a median of 6 weeks post-stroke and the patients with ICH at a median of 7 weeks post-stroke (p < 0.05). Fig. 1 shows the modelled development of the BI over the first year post-stroke for patients with CI and patients with ICH, based on the GEE analysis. No significant effect of type of stroke was found, but for both the patients with CI and ICH a significant change over time was found (Table II). Among the patients with CI, BI showed a significant increase over time until 26 weeks post-stroke, whereas for the patients with ICH, a significant increase in BI was found only until 10 weeks post-stroke. The only significant effect of interaction between type of stroke and time was found between 12 and 26 weeks (Fig. 1). Over this interval, the BI of the patients with CI increased more than that of the patients with ICH.
Fig. 1. Modelled development of the Barthel Index over the first year post-stroke for patients with a cerebral infarction and those with an intracerebral haemorrhage. p-values concern the difference between the 2 patient groups in development over a particular time-period.
| Table II. Changes in Barthel Index over the first year post-stroke in patients with a cerebral infarction and those with an intracerebral haemorrhage |
| Time interval post-stroke | Cerebral infarction | Intracerebral haemorrhage |
| β | SE | p | β | SE | p |
| Baseline–8 weeks | 1.72 | 0.19 | 0.00 | 1.92 | 0.55 | 0.00 |
| 8–10 weeks | 0.74 | 0.16 | 0.00 | 0.99 | 0.32 | 0.00 |
| 10–12 weeks | 0.79 | 0.14 | 0.00 | 0.35 | 0.26 | 0.17 |
| 12–26 weeks | 1.54 | 0.20 | 0.00 | 0.48 | 0.36 | 0.19 |
| 26–52 weeks | 0.002 | 0.14 | 0.99 | 0.62 | 0.33 | 0.06 |
| β: regression coefficient for the effect of time; SE: standard error. |
Fig. 1. Modelled development of the Barthel Index over the first year post-stroke for patients with a cerebral infarction and those with an intracerebral haemorrhage. p-values concern the difference between the 2 patient groups in development over a particular time-period.
Discussion
The development of independence in ADL differed between patients with CI and those with ICH. From 12 to 26 weeks post-stroke, patients with CI showed a statistically significantly faster increase in BI scores. The time window for recovery was more restricted for patients with ICH; an increase in ADL independence was found until 10 weeks post-stroke in patients with ICH, whereas patients with CI showed recovery until 26 weeks post-stroke.
To our knowledge, this is the first longitudinal study to compare the development of ADL over time between patients with CI and ICH. Although 2 other longitudinal studies (13, 14) did examine the pattern of ADL recovery in patients after stroke among a hospital population, they did not study the differences between the types of stroke. Like these studies (13, 14), we found that the development of ADL over time shows a non-linear pattern. The cause of this non-linear pattern of recovery is insufficiently understood. Recent findings suggest that the pattern observed in the first 2 months post-stroke is determined mainly by unknown processes attributed to “spontaneous neurological recovery” and relatively less by the impact of therapy (15). Early spontaneous gains not driven by practice and learning are believed to be related with: (i) recovery of penumbral tissue in which electrical failure has occurred but membrane homeostasis is maintained, (ii) resolution of functionally suppressed areas remote from the infarcted area by diaschisis; and (iii) unmasking of latent synaptic pathways (15). An examination of the differences in the pathophysiological processes between CI and ICH, suggests a possible explanation for our findings of different recovery patterns. One may hypothesize that, in case of a haematoma, the surrounding white matter will be pushed aside more, whereas in a CI the metabolism of cerebral tissue is more directly affected by the hypo-perfusion. As the haematoma resolves and oedema diminishes, the brain tissue can partially or wholly restore its function. A relatively sudden and rapid recovery of neurological signs and symptoms can be seen in patients with ICH. In CI, by contrast, the lesioned area will partly recover as a result of tissue repair and recovery of non-irreversibly damaged, hypo-perfused areas and brain areas remote from, but anatomically related to the infracted area. A slower, more gradual and sustained pattern of recovery in the CI group is expected.
In interpreting our results, several limitations of the study must be considered. First, our study was carried out in a selected stroke population, namely those in inpatient rehabilitation. The selection criteria for inpatient rehabilitation in the Netherlands are: the patient cannot be discharged home, but is expected to return home; the patient is able to learn and motivated; and has sufficient vitality. Therefore this population has specific characteristics in that the patients are relatively young and on average moderately disabled. Differences in neurological functions between patients with CI and ICH, which are found in the hospital population (1), were not found in our rehabilitation population. This is probably the result of the selection procedure for rehabilitation (16) at the hospital. Secondly, our study provides no information on the phase of hospital stay. Data on (functional) severity at onset and medical interventions in the acute phase are lacking. Thirdly, the baseline was not at a fixed time-point, but at the moment of admission to the rehabilitation centre. As this time-point was significantly different for patients with CI and ICH, this complicates the interpretation of the differences in ADL development during the first time-interval. Fourthly, functional recovery was assessed with the BI, which is a well-known and frequently used measure to describe the functional status in patients after stroke. However, since the BI measures ADL independence, our study provides just a partial examination of the differences in functional recovery. Other areas of functioning still have to be examined to get a more comprehensive assessment of potential functional differences between CI and ICH. In addition, the BI is particularly suitable for use in the acute and subacute phases, whereas for later phases, one must consider the known ceiling effect of the BI. Finally, the number of patients with ICH included was relatively small compared with the number of patients with CI. As a result, potentially important differences between the 2 types of stroke could nevertheless be statistically non-significant.
In the acute phase, the first step in stroke management is to differentiate between ischaemic strokes and intracerebral haemorrhages, as this influences decisions about medical and surgical treatment. In view of the findings of this study, the difference in course of recovery between CI and ICH should also be considered in the inpatient rehabilitation setting.
References
1. Jorgensen HS, Nakayama H, Raaschou HO, Olsen TS. Intracerebral hemorrhage versus infarction: stroke severity, risk factors, and prognosis. Ann Neurol 1995; 38: 45–50.
2. Kelly PJ, Furie KL, Shafqat S, Rallis N, Chang Y, Stein J. Functional recovery following rehabilitation after hemorrhagic and ischemic stroke. Arch Phys Med Rehabil 2003; 84: 968–972.
3. Ring H, Feder M, Schwartz J, Samuels G. Functional measures of first-stroke rehabilitation inpatients: usefulness of the Functional Independence Measure total score with a clinical rationale. Arch Phys Med Rehabil 1997; 78: 630–635.
4. Chae J, Zorowitz RD, Johnston MV. Functional outcome of hemorrhagic and nonhemorrhagic stroke patients after in-patient rehabilitation. Am J Phys Med Rehabil 1996; 75: 177–182.
5. Paolucci S, Antonucci G, Grasso MG, Bragoni M, Coiro P, De Angelis D, et al. Functional outcome of ischemic and hemorrhagic stroke patients after inpatient rehabilitation: a matched comparison. Stroke 2003; 34: 2861–2865.
6. Ween JE, Alexander MP, D’Esposito M, Roberts M. Factors predictive of stroke outcome in a rehabilitation setting. Neurology 1996; 47: 388–392.
7. Inouye M, Kishi K, Ikeda Y, Takada M, Katoh J, Iwahashi M, et al. Prediction of functional outcome after stroke rehabilitation. Am J Phys Med Rehabil 2000; 79: 513–518.
8. Lipson DM, Sangha H, Foley NC, Bhogal S, Pohani G, Teasell RW. Recovery from stroke: differences between subtypes. Int J Rehabil Res 2005; 28: 303–308.
9. Demeurisse G, Demol O, Robaye E. Motor evaluation in vascular hemiplegia. Eur Neurol 1980; 19: 382–389.
10. Folstein MF, Folstein SE, McHugh PR. “Mini-mental State”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 1975; 12: 189–198.
11. Collin C, Wade DT, Davies S, Horne V. The Barthel ADL Index: a reliability study. Int Disabil Stud 1988; 10: 61–63.
12. Stata Reference Manual, release 7. College station, Texas: Stata Press; 2001.
13. Jorgensen HS, Nakayama H, Raaschou HO, Vive-Larsen J, Stoier M, Olsen TS. Outcome and time course of recovery in stroke. Part II: time course of recovery. The Copenhagen stroke study. Arch Phys Med Rehabil 1995; 76: 406–412.
14. Kwakkel G. Dynamics in functional recovery after stroke [Thesis]. Ponsen & Looijen BV, The Netherlands. 1998.
15. Kwakkel G, Kollen B, Lindeman E. Understanding the pattern of functional recovery after stroke: facts and theories. Restor Neurol Neurosci 2004; 22: 281–299.
16. Johnston MV, Wood K, Stason WB, Beatty P. Rehabilitative placement of poststroke patients: reliability of the Clinical Practice Guideline of the Agency for Health Care Policy and Research. Arch Phys Med Rehabil 2000; 81: 539–548.