OBJECTIVE: To determine the efficacy of botulinum toxin type A for the treatment of spastic shoulder pain in patients after stroke.
DESIGN: Double-blind randomized clinical trial.
Patients: Of 31 patients enrolled from an acute-care hospital in Spain, 2 cases dropped out (drop-out rate 6.5%). Fourteen subjects were treated with infiltration of 500 units of botulinum toxin type A in the pectoralis major muscle of the paretic side, and 15 with a placebo.
METHODS: After infiltration, both groups received transcutaneous electrical nerve stimulation for 6 weeks. Patients were assessed by the use of the Visual Analogue Scale for pain. A good result concerning pain was considered when the Visual Analogue Scale score was below 33.3 mm or less than half the initial score. The patients were followed-up for 6 months.
RESULTS: The patients treated with botulinum toxin type A showed a significantly greater pain improvement from the first week post-infiltration. Persistent shoulder pain was observed more frequently in the placebo group, with relative risks in the range 0.32–0.41 during the follow-up period.
CONCLUSION: Patients with spastic shoulder pain treated with a botulinum toxin type A infiltration in the pectoralis major muscle of the paretic side have a higher likelihood of pain relief (between 2.43- and 3.11-fold).
Key words: shoulder pain, botulinum toxin, cerebrovascular disease, muscle spasticity, rehabilitation.
J Rehabil Med 2007; 39: 440–447
Correspondence address: Ester Marco, Physical Medicine and Rehabilitation Department, Hospital de l’Esperança, Sant Josep de la Muntanya 12, ES-08024 Barcelona, Spain. E-mail: emarco@imas.imim.es
Submitted July 13, 2006; accepted January 8, 2007
INTRODUCTION
Shoulder pain is a common complication in patients suffering from hemiplegia secondary to a cerebrovascular accident (CVA) (1). Its prevalence oscillates between 34% and 84%, it is age- and sex-independent and it usually appears from the second week post-CVA (2–3).
Spasticity is outstanding (2) among the various factors involved in shoulder pain occurrence and in the upper limb. Spasticity shows a pattern involving adduction and internal rotation of the shoulder. If this pattern persists, it can cause pain and limit the shoulder range of motion, interfering with the patient’s rehabilitation schedule and considerably restricting their functional outcome.
Different preventive and therapeutic options have been suggested, from orthotic supports and electric therapy techniques to various drugs. Most of the drugs proposed (baclofen, benzodiazepines, tricyclic antidepressants, dantrolene, tizanidine, etc.) have a concomitant depressing effect on the central nervous system, which limits their tolerance and efficacy (4). In cases of moderate spasticity, the traditional treatment of choice has been a combination of kinesiotherapy techniques (exercises to maintain or improve joint mobility), orthoses and oral medication. Among the different electric therapy modes, functional electric stimulation and transcutaneous electrical nerve stimulation (TENS) have shown their usefulness in the management of shoulder pain in patients with hemiparesis (5–7).
When spasticity affects certain muscular groups it is preferable to apply local techniques as they prevent overactivity, contractures and muscle shortening. For muscle “stretching” to be effective, it should be maintained for several hours a day, and therefore isolated physiotherapy techniques are not enough. Local pharmacotherapy, in the form of infiltrations with neurolytic agents in the muscles suffering from spasticity, has been used for many years. Perineural or intramuscular blockade with local anaesthetics acts by reducing the evident motor activity for only a few hours. Local infiltrations of alcohol (> 10%) and phenol have shown a longer effect (2–36 months), but they are painful and have low selectivity (8–9).
Botulinum toxin type A (BTA) is a drug with widely shown efficacy in the treatment of focal spasticity (10–15) secondary to different causes such as multiple sclerosis, brain traumatisms, cerebral palsy in children, spinal cord injuries or CVA.
In the treatment of the vascular hemiplegic patient’s spastic upper extremity, some studies show BTA’s usefulness for spasticity affecting the elbow, the wrist and the hand (16–19). A recent study (20) has pointed out the beneficial effects of BTA injections in the subscapular muscle on pain and shoulder mobility, but the authors are not aware of controlled clinical trials published in the literature about its efficacy on spastic shoulder pain in patients after hemiplegic stroke. The length of the clinical response in the spastic muscle depends upon factors such as dose, size and activity of the treated muscle, even though the mean doses currently being used are 500 units of Clostridium botulinum type A toxin-haemagglutinin complex (Dysport®; Ipsen Pharma, SA, Barcelona, Spain) per “large” muscle (for instance the triceps surae or the biceps brachii muscles) with an anticipated length of the clinical effect of 2–6 months. The treatment acts by diffusion from the injection site to the adjacent areas, and the estimated diffusion area is 4.5 cm2 for this reason the application of multiple injections is more effective than the administration of the whole dose at a single site.
The pectoralis major is one of the muscles of the shoulder girdle involved in the motor pattern of adduction and internal rotation characteristic of the spastic shoulder in patients with hemiparesis. There were 2 reasons why we chose to treat only the pectoral muscle. First, the lack of studies of the effects of BTA injection in the pectoralis muscle regarding pain and mobility and secondly, from a technical point of view, infiltrating this muscle seems to be easier than infiltrating the subscapular muscle. Therefore, a better knowledge of its effects could be useful in clinical practice. An increase in the tone of the pectoralis major can be detected easily when exploring abduction and external rotation in these patients; a manoeuvre that usually increases their pain. Considering that the chemical muscle denervation produced by the BTA injection improves spasticity, it would be logical to think that, consequently, it also relieves pain. Therefore, the primary end-point of this study was to determine the efficacy of BTA injections in the pectoralis major for the treatment of spastic shoulder pain of vascular hemiparetic patients. The secondary end-point was to assess BTA efficacy on the degree of spasticity and shoulder joint mobility.
METHODS
Trial design and participants
A prospective, double-blind, randomized controlled clinical trial was established to determine the efficacy of BTA for spastic shoulder pain in rehabilitation patients after stroke, and as a secondary end-point, to assess its effect on spasticity, shoulder range of motion and disability.
Patients were eligible for inclusion if they met the following criteria:
• Age over 18 years, of either sex.
• Having spastic hemiparesis due to CVA of 3 or more months of evolution.
• Moderate-severe spastic shoulder pain:
• Visual Analogue Scale (VAS) for pain equal to or greater than 40 mm
• Spasticity of 3 or more points as determined by the Modified Ashworth Scale (MAS) (21).
• Ability to understand and accept the trial procedures and to sign an informed consent form in accordance with national legislation.
Participants were characterized according to their level of hemiparetic severity, identified on the basis of the Brunnstrom motor recovery stages (22). Patients with mild hemiparesis (defined as Brunnstrom stage 6) were excluded. Patients were also excluded if they presented previous concomitant shoulder pathology, were fitted with pacemakers (contra-indication for TENS), had peripheral nervous system diseases, had hypersensitivity to botulinum toxin or were pregnant. Treatment with anticoagulants was not considered an exclusion criterion, and anticoagulated patients were included in the study if following a haemostasis control made on the same day as the infiltration, their international normalized ratio (INR) was shown to be within therapeutic margins.
The setting was a rehabilitation unit in an acute-care general university hospital in Barcelona, Spain. The clinical trial was approved by the Institut Municipal d’Assistència Mèdica (IMAS)’s Ethics Committee for Clinical Research, and by the Spanish Agency of Medicines (registration code: RHBESPE/TOXIN/1). The trial received no funding from the pharmaceutical industry.
Interventions
Patients were distributed between 2 treatment groups:
• Group I received treatment with TENS + BTA infiltration.
• Group II received treatment with TENS + placebo infiltration (2.5 ml of physiological saline solution).
Participants were informed about the trial and its risks and gave their written consent. After having been allocated randomly to 1 of the 2 groups, the patients were treated by means of intramuscular injection, at 4 sites, of 500 units of BTA (Dysport®) vs a placebo in the pectoralis major muscle of the paretic side, under electromyographic monitoring. The injection site was located at the upper front of the chest next to the shoulder where the muscle fibres converge towards their insertion on a line arising from the coracoid apophysis and passing downward. Subsequently, all the patients were treated with conventional TENS, consisting of short pulses (250 μsec) of high frequency (75 megahertz) and low intensity for a 6-week period. Subjects were then evaluated 4 more times: at 7 days, 1 month, 3 months and 6 months after infiltration. Although all participants were still undergoing training in daily living activities and different aspects of mobilitity, none of them was following any specifical treatment for alleviating pain or improving shoulder mobility.
Randomization and blinding
Treatment blinding and randomization were carried out in the hospital’s pharmacy service using a program to generate random numbers. The injections were also prepared in the pharmacy service, thus the rehabilitation specialist in charge of infiltrations was not aware of administering BTA or placebo. At the time of BTA infiltration, neither the patients, the doctors (rehabilitation specialist and electromyographist) nor the physiotherapists were aware of which group they had been allocated to. At one month follow-up, clinical and electromyographic controls were conducted separately, since at that phase it might be possible to differentiate between patients injected with BTA or placebo. From then on, clinical assessment was conducted exclusively by the same trained rehabilitation specialist who had no knowledge of the electromyography measurements, in order to ensure the blind status of the trial.
Outcomes
The primary outcome measurement of the study was pain, measured with the VAS for pain (23) from 0 to 100 mm when mobilizing the shoulder. The minimum difference in pain decrease considered clinically significant was 33.3 mm. The treatment was considered to have yielded good results concerning pain when the patient’s VAS score was below 33.3 mm (mild pain) or when the score was less than half the initial score.
Secondary outcome measures collected were:
• Spasticity measured with the MAS (ranging from 0 to 5).
• Shoulder range of motion expressed in degrees: flexion, abduction and external rotation.
The assessment of range of movement was (24):
• Flexion: supine, arm at side with hand pronated.
• Abduction: supine, arm at side.
• External rotation: supine, arm abducted to 90º and elbow off table, elbow flexed to 90º and hand pronated, forearm perpendicular to floor.
The assessment of pain and spasticity was performed in the same position as abduction. The examiner performed a passive stretch in abduction of the arm until the maximal range of movement supported by the patient was reached.
Sample size
The sample size was calculated so that a minimum of 14 subjects were necessary in each group by accepting a difference between treatments of 33.3 mm in the VAS for pain, with a 30-mm standard deviation (SD), and for an alpha risk of 0.05 and a beta risk of 20% on a bilateral contrast. The sample size was overestimated in order to allow for up to 15% of potential drop-outs.
Procedures
Patients were referred to a rehabilitation and neurophysiology clinic. The data collected at the first evaluation, immediately before injection, were: age, sex, disease history, type of CVA (ischaemic or haemorrhagic), time of CVA progression, time of shoulder pain progression (in months), pharmacological treatments of spasticity over the last month, other treatments, VAS for pain at rest and on mobilization, shoulder joint function (flexion, abduction, external rotation) and spasticity according to the MAS. A neurographic study (amplitude and latency) of the pectoral nerve with muscular response determination was performed prior to the infiltration. The preparation was then administered by the intramuscular route (either BTA or placebo) under electromyographic monitoring. All patients underwent passive shoulder mobilizations in order to encourage diffusion of the preparation in the infiltrated muscle, and were instructed by the same physiotherapist to start a daily home treatment with TENS for 6 weeks. A second electromyographic control was performed by the same neurophysiologist one month after the injection in order to monitor the pharmacological effect of the drug.
Statistical analysis
Categorical variables are given in absolute and percentage values. Quantitative variables are given together with the mean and SD, or else with the median and 25th and 75th percentiles (P25–P75) when they did not meet normality criteria. In the case of quantitative variables, the assumption of normality was analysed through the normal probability graphs and using the Kolmogorov-Smirnov test corrected by the Lilliefors test. Only one variable was outside the normal distribution for time of CVA progression.
Patients were classified according to pain improvement into: patients with a good outcome (when the VAS for pain was below 33.3 mm or when the score was less than half the initial score) and patients with a bad outcome. To best explain differences in efficacy between treatments regarding pain, the relative risk (RR) and 95% confidence interval (CI) were calculated for every time interval. An RR of less than one indicated that treatment with TBA protected the patient from shoulder pain. In these cases, the inverse of the RR was calculated to quantify this protection.
Univariate analysis used either the χ2 test or the Fisher’s exact test for categorical variables, and Student t-test or Mann-Whitney U test for quantitative variables. Analysis of variance was performed using a repeated-measures mixed design (intra-subjects) and one factor (inter-subjects) for the analysis of values over time. When the sphericity criteria were not complied with, the degrees of freedom were corrected using Greenhouse-Geisser’s method. The level of statistical significance was 0.05 for all hypothesis contrasts.
Role of funding source
The trial received no funding from the pharmaceutical industry. The Institut Municipal d’Investigacio Mèdica in Barcelona provided a grant to fund this study. The funding source has no role in the study design, data collection, data analysis, data interpretations or writing of the report. The corresponding author had full access to all the data in the study and had final responsibility for the decision to submit for publication.
RESULTS
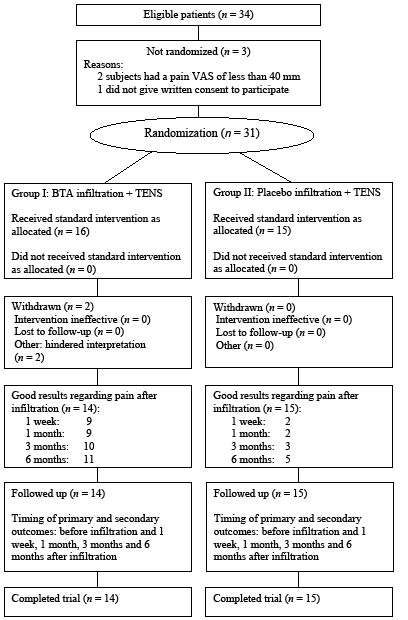
The trial procedure is described in Fig. 1. Thirty-one patients were enrolled in the trial between August 2001 and July 2003. Two patients were excluded upon completion; one for presenting neuropathic pain of the whole paretic half of the body, and 1 for presenting a personality disorder associated with a drinking habit that greatly hindered interpretation of his progress during follow-up. The final sample was therefore 29 patients (14 in Group I and 15 in Group II).
Fig. 1. Trial Profile (CONSORT flow diagram).
VAS: visual analogue scale; BTA: Botulinum toxin type A; TENS: transcutaneous electrical nerve stimulation.
The mean age of the sample was 65.6 (SD 9.1) years, and the gender distribution was 21 (72.4%) men and 8 (27.6%) women. All the patients had presented ischaemic strokes with a slight predominance of right hemiparesis (16 vs 13). The baseline demographic and clinical characteristics of each group are shown in Table I. No statistically significant differences were observed between the groups regarding any of the clinical and demographic characteristics.
| Table I. Sample baseline characteristics. All values are non-significant. |
| | Total sample n = 29 | Group I n = 14 | Group II n = 15 |
| Age, years: mean (SD) | 65.6 (9.06) | 63.9 (10.6) | 67.2 (7.4) |
| Distribution by sexes: n (%) |
| Men | 21 (72.4) | 10 (71.4) | 11 (73.3) |
| Women | 8 (27.6) | 4 (28.6) | 4 (26.7) |
| Type of CVA: n (%) |
| Ischaemic | 29 (100) | 14 (100) | 15 (100) |
| Haemorrhagic | 0 (0) | 0 (0) | 0 (0) |
| CVA laterality: n (%) |
| Right | 16 (55.2) | 8 (57.1) | 8 (53.3) |
| Left | 13 (44.8) | 6 (42.9) | 7 (46.7) |
| Concomitant disease: n (%) |
| Hypertension | 20 (69.0) | 9 (64.3) | 11 (73.3) |
| Diabetes mellitus | 8 (27.6) | 3 (21.4) | 5 (33.3) |
| Prior CVA | 5 (17.2) | 2 (14.3) | 3 (20.0) |
| Heart disease | 6 (20.) | 3 (21.4) | 3 (20.0) |
| Respiratory disease | 2 (6.9%) | 2 (14.3) | 0 (0) |
| Neoplasias | 4 (13.8) | 0 (0) | 4 (26.7) |
| Patients on oral antispastic agents, n (%) | 5 (17.2) | 2 (14.3) | 3 (20.0) |
| Time of CVA evolution (days)* | 141, 107–241 | 174, 89–263 | 133, 112–210 |
| Time of SP appearance: n (%) |
| First month post-CVA | 18 (62.1) | 9 (64.3) | 9 (60) |
| After first month post-CVA | 11 (37.9) | 5 (35.7) | 6 (40) |
| CVA: cerebrovascular accident; SP: shoulder pain; SD: standard deviation. *Data expressed by the median and 25th–75th percentile. |
Mean pain VAS on passive mobilization was 73.1 (SD 15.5) mm. The degree of spasticity in 16 of 29 cases was 4 on the MAS. Table II gives a detailed description of the main variables measured during screening prior to infiltration and the follow-up period and here again there were no significant differences found between the groups.
| Table II. Two-factor ANOVA of main measure outcomes prior to starting treatment and during the follow-up period. Quantitative variables are expressed by the mean and standard deviation in parentheses. |
| | Before infiltration | 1 week post-infiltration | 1 month post-infiltration | 3 months post-infiltration | 6 months infiltration | Two-factor ANOVA |
| | Group I | Group II | Group I | Group II | Group I | Group II | Group I | Group II | Group I | Group II | Time effects | Group effects | Interaction effects |
| VAS of pain | 76.4 (15.6) | 70.1 (15.3) | 44.4 (25.9) | 59.3 (21.0) | 38.7 (27.0) | 60.1 (22.1) | 35.4 (25.3) | 56.7 (23.4) | 30.1 (26.9) | 48.3 (29.4) | < 0.001 | 0.048 | 0.035 |
| Passive shoulder range: | | | | | | | | | | | | | |
| Flexion | 84.6 (23.2) | 83.7 (25.2) | 91.4 (30.4) | 91.3 (18.9) | 95.0 (28.7) | 92.3 (21.4) | 98.6 (32.6) | 91.0 (17.1) | 97.9 (31.4) | 96.7 (19.1) | 0.015 | NS | NS |
| Abduction | 69.6 (18.4) | 66.7 (18.5) | 80.7 (19.0) | 66.3 (22.3) | 81.1 (10.8) | 65.3 (17.5) | 81.1 (10.8) | 67.7 (23.3) | 77.5 (27.9) | 72.7 (26.3) | NS | NS | NS |
| External rotation | 7.9 (27.9) | 6.7 (18.1) | 20.0 (26.9) | 8.3 (20.0) | 21.4 (28.9) | 13.7 (19.1) | 23.9 (35.0) | 22.3 (26.3) | 38.9 (34.9) | 19.3 (23.8) | < 0.001 | NS | 0.041 |
| Modified Ashworth Scale | 3.1 (0.7) | 3.13 (0.6) | 2.79 (1.0) | 3.13 (0.7) | 2.9 (1.0) | 3.1 (0.8) | 2.9 (1.0) | 3.2 (0.9) | 2.9 (1.2) | 3.2 (0.9) | NS | NS | NS |
| Neurographic study | | | | | | | | | | | | | |
| Motor potential | | | | | | | | | | | | | |
| Amplitude | 4.3 (2.0) | 3.4 (1.8) | – | – | 2.3 (1.2) | 3.8 (2.4) | – | – | – | – | 0.015 | NS | <0.001 |
| Latency | 2.4 (0.5) | 2.4 (0.7) | – | – | 3.0 (1.3) | 2.9 (1.3) | – | – | – | – | 0.33 | NS | NS |
| VAS: visual analogue scale; NS: not significant. |
The main measure outcomes under study (intra-subjects) were analysed by intention-to-treat and over the follow-up period showed the results described below.
Visual Analogue Scale for pain
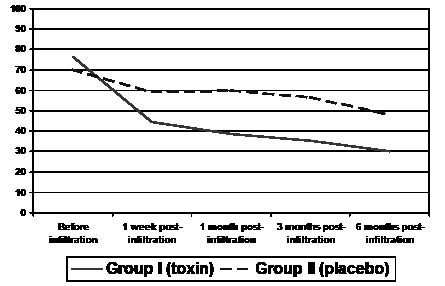
Decreased pain VAS values when mobilizing the shoulder were observed in both groups from the first week post-infiltration, but the magnitude of this decrease was greater in Group I (mean VAS one week post-infiltration: 44.4 mm, SD 26.0) compared with Group II (mean VAS 59.3 mm, SD 21.0). On subsequent controls, the VAS values continued to diminish in Group I until reaching 38.7 (SD 26.9) mm on the first month, 35.4 (SD 25.3) mm on the third month, and 30.1 (SD 26.9) mm on the sixth month. As for Group II, the mean scores for the VAS were higher: 60.1 (SD 22.1) mm on the first month, 56.7 (SD 23.4) mm on the third month, and 48.3 (SD 29.2) mm on the sixth month, constituting a statistically significant difference between both groups (p = 0.035) (Fig. 2). In Group I there was a mean reduction of 46.2 (SD 34.2) mm on completion of the study, whereas the Group II reduction was 21.9 (SD 29.4). Treatment with BTA protected patients from shoulder pain, and resulted in patients presenting a RR of less than one in all the follow-up periods (Table III).
Fig. 2. Pain progression at 1 week, 1 month, 3 months and 6 months, as measured with the Visual Analogue Scale for pain.
| Table III. Contingency tables to show differences in efficacy of pain treatment between groups. |
| | Persistent pain 1 week post-infiltration | Persistent pain 1 month post-infiltration | Persistent pain 3 months post-infiltration | Persistent pain 6 months post-infiltration |
| Yes | No | Yes | No | Yes | No | Yes | No |
| Group I (toxin) Group II (placebo) | 5 13 | 9 2 | 5 13 | 9 2 | 4 12 | 10 3 | 3 10 | 11 5 |
| Relative risk (95% CI) | 0.41 (0.20–0.86) | 0.41 (0.20–0.86) | 0.36 (0.15–0.85) | 0.32 (0.11–0.93) |
| CI: confidence interval. |
Shoulder range of motion
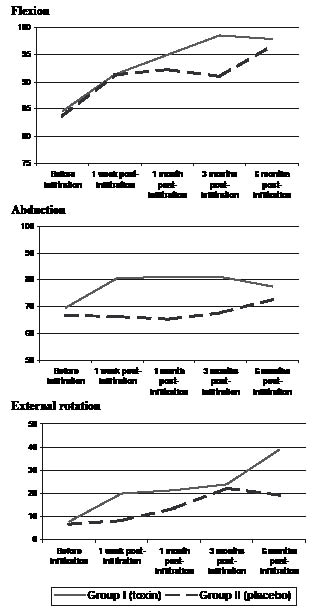
Flexion. The mean flexion at study initiation was 84.6 (SD 23.2) degrees in Group I and 83.7 (SD 25.2) degrees in Group II. The mean values at 6 months post-infiltration were 97.9 (SD 31.4) degrees and 96.7 (SD 19.1) degrees, respectively. No statistically significant differences were found between both groups throughout the follow-up period (p > 0.05) (Table II and Fig. 3).
Abduction. The mean abduction at study initiation was 69.6 (SD 18.4) degrees in Group I and 66.7 (SD 18.5) degrees in Group II. Increased abduction was appreciated from the first week post-infiltration, and maintained with little variation until the sixth month of follow-up in the BTA-treated group. No statistically significant differences were found between both treatment groups at the 6 month follow-up (Table II and Fig. 3).
External rotation. The mean external rotation improved from 7.9 (SD 27.9) degrees at the start of the study to 38.9 (SD 34.8) degrees after 6 months in Group I. The improvement in Group II was more moderate: from 6.7 (SD 18.1) to 19.3 (SD 23.8) degrees. These differences were indeed statistically significant between both treatment groups (p = 0.041). The overall improvement in external rotation was 31.1 (SD 24.4) degrees in Group I and 12.7 (SD 16.0) degrees in Group II (Table II and Fig. 3).
Fig. 3. Progression of the shoulder joint mobility (flexion, abduction and external rotation) expressed in degrees during follow-up.
Spasticity
Even though a moderate decrease in the MAS occurred among the toxin-treated group, no statistically significant differences were observed between both groups throughout the follow-up (p > 0.05).
Neurographic study of the pectoral nerve and muscular response measure
The mean amplitude of the motor potential prior to infiltration was 4.3 (SD 2.0) mV in the BTA-treated group, and 3.4 (SD 1.8) mV in the placebo-treated group. One month post-infiltration, the mean amplitude was reduced to 2.3 (SD 1.2) mV in Group I, whereas it increased to 3.8 (SD 2.4) mV in Group II, a statistically significant difference (p < 0.001). No significant differences were found between both groups regarding the latency of the potential. Before infiltration, Group I presented a mean latency of 2.4 (SD 0.5) ms, and Group II 2.4 (SD 0.7) ms. One month post-infiltration, the mean latency was 3.0 (SD 1.3) ms in Group I, and 2.9 (SD 1.3) ms in Group II.
Adverse effects
No important adverse effects occurred. Two patients in the placebo group reported fatigue and a moderate loss of strength of the upper limb, which was self-limited in just a few days.
DISCUSSION
This trial suggests that infiltration of BTA into the pectoralis major muscle is effective in the treatment of spastic shoulder pain in patients after stroke. Furthermore, it also improves external rotation range of motion. Even though the use of BTA for the treatment of upper extremity focal spasticity is currently accepted, we have found no other works published in the clinical literature assessing the results of treatment of spastic shoulder pain of the vascular hemiplegic patient by means of intramuscular infiltration of BTA in the pectoralis major muscle.
Before attempting any further discussion of these findings, some limitations of the present study should be noted. First, it is well known that samples of patients from rehabilitation units tend to be pre-selected for the patients’ potential to follow a rehabilitation programme. The sample presents an initial bias due to the fact that not all the patients suffering from hemiplegia after a stroke were admitted to follow an intensive in-patient rehabilitation programme and some were transferred to other facilities. Admission criteria to the trial setting were: moderate-severe hemiparesis without cognitive impairment and stabilization of medical conditions. Regarding the kind of toxin used, we decided to apply Dysport® BTA because it was the only drug approved in Spain for treating upper limb spasticity at the time of trial initiation. As for the dose used, the decision was based on current international recommendations. However, a higher or lower dose could have produced a greater or lesser effect. It is necessary to investigate whether the same effect could have been obtained if patients had been treated with lower doses. Although no adverse effects occurred, the cost of the treatment would be considerably reduced with lower doses.
With regard to the pain relief observed, we should first consider why pain improvement is taking place: Is it because the spasticity is treated and consequently, a secondary symptomatic alleviation occurs? Or is it because BTA acts at the nociceptive pathway neurotransmission level? The causality is difficult to establish. In fact, no improvement is seen in the MAS. It is true that spasticity is a difficult state to evaluate, particularly due to the great intra-subject variability, and also due to the multiple factors that may trigger it, often in a transient manner. In the case of the shoulder, assessment of the spasticity is hindered by the presence of pain and limitation of range of movement in a practically constant manner. Despite the shortcomings and lack of consensus in the exact scoring, the MAS with all of its variants remains the standard with which other measures of spasticity are compared. The lack of improvement on the MAS questions the validity of this scale to measure spasticity and indicates the need for further studies regarding assessment of spasticity in the shoulder. In spite of there being scant direct evidence that BTA might have an analgesic effect on humans (25), some previous works have reported an association between pain and BTA (26–29). This association was initially thought to be due to the effect of the toxin on muscle contraction. However, the fact that in many cases muscular relaxation does not coincide with pain improvement suggests the existence of alternative mechanisms that favour BTA’s analgesic effects. Experimental evidence shows that botulinum toxin acts on neurotransmission afferent pathways (30), and this could partially explain such improvement. On the other hand, BTA also inhibits P-substance production (a neuropeptide that participates in pain perception, vasodilatation and neurogenic inflammation) and, potentially, the production of other neuromodulators (31).
The muscles most often contributing to the adduction pattern and to the shoulder’s internal rotation in patients after stroke are the latissimus dorsi, teres major, subscapular and pectoralis major muscles. The tendon of the pectoralis major is usually prominent when the examiner tries to obtain passive movement in abduction and external rotation, although the role played by the rest of the musculature should not be ignored. Some works have demonstrated that the subscapular muscle could be a significant source of internal rotation (32–33), but this muscle is both difficult to study by electromyography, and difficult to produce chemical denervation in. Since the pectoralis major is a large easily accessible muscle, electromyography is not indispensable in clinical practice. However, for the purpose of this study, using it allows for verification of whether the motor amplitude of the inherent activity potential of the denervated muscle has diminished. It would be expected that the latency of the motor potential is not modified following the treatment, as there is no reason why the time needed by the motor potential to pass through the preserved nerve fibres should vary. The result inferred from neurography is that a significant reduction in the muscular response occurs in almost 50% of its amplitude, with normal response time. That is, an axon injury occurs that entails a loss of response of almost 50% of the muscular fibres of the pectoralis major, with the relevant spasticity-related functional response. In view of these results, perhaps it should be questioned whether 50% of the C-nociceptive fibres are also affected.
When analysing the differences in joint range improvement between both treatment groups, there was significant improvement in external rotation in the BTA-treated group. Not finding significant differences in flexion and abduction ranges requires additional comments. From a biomechanical point of view, it is surprising that such an effect was not significant for abduction. However, abduction did improve in the BTA group in the 3 first months after injection (Fig. 3 and Table II). This observation seems to be due to the fact that the biological effect of the BTA lasts for less than 6 months.
Further investigations should be conducted before generalizing the results observed in this trial regarding infiltration of the subscapular in association or not with the pectoralis major muscles. Another aspect to explore would be the lack of a function scale for shoulder assessment in hemiplegic patients. It should also be taken into account that the trial setting was a rehabilitation unit in an acute-care hospital (where samples tend to be pre-selected) and that the subjects eligible for inclusion were consecutive hemiparetic patients with moderate-severe shoulder pain. Therefore, conclusions should only be applied to the population of hemiparetic patients who have followed a rehabilitation programme after suffering a CVA. In addition, different generalizability contexts (times and places) should also be contemplated. Probably the best approach to improve external validity would be replicating the trial in different contexts.
It should be remembered, however, that the most important approach regarding this pathology is prevention by means of postural and joint hygiene measures. When spastic shoulder pain persists and initial traditional treatment (kinesiotherapy and/or pharmacological treatment) fails, we believe that BTA infiltration is a good option for consideration in these patients for reducing pain and improving shoulder mobility.
In summary, this trial concludes that BTA is more effective than placebo in reducing pain and improving external rotation in patients with vascular hemiplegia with spastic shoulder pain. The infiltration of BTA into the pectoralis major muscle increases the probability of alleviating shoulder pain by 2.46–3.11-fold.
ACKNOWLEDGEMENTS
This paper was presented at the 42nd Congreso de la Sociedad Española de Rehabilitación y Medicina Física, 9 June 2004 in La Coruña, Spain, the Mediterranean Congress of Physical and Rehabilitation Medicine, 1 October 2004 in Antalya, Turkey, and the Evidence for Stroke Rehabilitation – Bridging into the Future meeting, 26–28 April 2006 in Göteborg, Sweden.
The authors thank Dr Josep M. Manresa (Biostatistics from the Institut Municipal d’Investigació Mèdica, Barcelona, Spain) for his expert statistical assistance and Rod V. Bowman for language revisions. The content of this trial is solely the responsibility of the authors and does not necessary represent the view of official institutions.
REFERENCES
1. Joynt RL. The source of shoulder pain in hemiplegia. Arch Phys Med Rehabil 1992; 73: 409–413.
2. Van Ouwenaller C, Laplace PM, Chantraine A. Painful shoulder in hemiplegia. Arch Phys Med Rehabil 1986; 67: 23–26.
3. Zorowitz RD, Hughes MB, Idank D, Ikai T, Johnston MV. Shoulder pain after subluxation after stroke: correlation or coincidence? Am J Occup Ther 1996; 50: 194–201.
4. Abruzzesse G. The medical management of spasticity. Eur J Neurol 2002; 9: 30–34.
5. Chantraine A, Baribeault A, Uebelhart D, Gremion G. Shoulder pain and dysfunction in hemiplegia: effects of functional electrical stimulation. Arch Phys Med Rehabil 1999; 80: 328–331.
6. Leandry M, Bouter LM, Beckerman H. Comparison of TENS treatments in hemiplegic shoulder pain. Scand J Rehabil Med 1990; 22: 69–71.
7. Sonde L, Geip F, Fernaeus SE, Nilsson CG, Viitanen M. Stimulation with low frequency (1.7 Hz) transcutaneous electric nerve stimulation (low TENS) increases motor function of the post-stroke paretic arm. Scand J Rehabil Med 1998; 30: 95–99.
8. Petrillo CR, Chu DS, Davis SW. Phenol block of the tibial nerve in the hemiplegic patient. Orthopedics 1980; 3: 871–874.
9. Zafonte RD, Munin MC. Phenol and alcohol blocks for the treatment of spasticity. Phys Med Rehabil Clin N Am 2001; 12: 817–832.
10. Das TK, Park DM. Effect of treatment with botulinum toxin on spasticity. Postgrad Med J 1989; 65: 208–210.
11. Snow BJ, Tsui JKC, Bhatt MH, Varelas M, Hashimoto SA, Calne DB. Treatment of spasticity with botulinum toxin: a double-blind study. Ann Neurol 1990; 28: 512–515.
12. Yablon SA. Botulinum toxin A: experience in the management of spasticity among patients with traumatic brain injury. Eur J Neurol 1995; 2: 47–55.
13. Koman LA, Mooney JF, Smith BP. Botulinum toxin: potential role in the management of cerebral palsy during childhood. In: Janovic J, Hallet M, editors. Therapy with botulinum toxin. New York: Marcel Dekker; 1994, pp. 511–522.
14. Cosgrove AP, Graham HK. Botulinum Toxin A in the management of spasticity with cerebral palsy. Br J Surg 1992; 74B: 135–136.
15. Dykstra DD, Sidi AA: Treatment of detrusor-sphincter dyssynergia with botulinum A toxin: a double-blind study. Arch Phys Med Rehabil 1990; 71: 24–26.
16. Grazko MA, Polo KB, Jabbari B. Botulinum toxin A for spasticity, muscle spasms, and rigidity. Neurology 1995; 45: 712–717.
17. Das TK, Park DM: Botulinum toxin in treating spasticity. Br J Clin Pract 1989; 43: 401–403.
18. Simpson DM, Alexander DN, O’Brien CF, Tagliati M, Aswad AS, Leon JM, et al. Botulinum toxin type A in the treatment of upper extremity spasticity: a randomized, double-blind, placebo-controlled trial. Neurology 1996; 46: 1306–1310.
19. Van Kuijk AA, Geurts ACH, Bevaart BJW, van Limbeek J. Treatment of upper extremity spasticity in stroke patients by focal neuronal or neuromuscular blockade: a systematic review of the literature. J Rehabil Med 2002; 12: 51–61.
20. Yelnik AP, Colle FM, Bonan IV. Treatment of pain and limited movement of the shoulder in hemiplegic patients with botulinum toxin A in the subescapular muscle. Eur Neurol 2003; 50: 91–93.
21. Bohannon RW, Smith MB. Interrater reliability of a Modified Ashworth Scale of Muscle Spasticity. Phys Ther 1987; 67: 206–207.
22. Brunnstrom S, editor. Movement therapy in hemiplegia: a neurophysiological approach. Hagerstown, MD: HarpeRow; 1970.
23. Collins SL, Moore RA, McQuay HJ. The visual analogue pain intensity scale: what is moderate pain in millimetres? Pain 1997; 72: 95–97.
24. Erickson RP, McPhee MC. Clinical evaluation. In: DeLisa JA, Gans BM, editors. Rehabilitation medicine: principles and practice, 3rd edn. Philadelphia: Lippincott-Raven Publishers; 1998, p. 74–75.
25. Voller B, Sycha T, Gustorff B, Schmetterer L, Lehr S, Eichler HG, et al. A randomized, double-blind, placebo controlled study on analgesic effects of botulinum toxin A. Neurology 2003; 61: 940–944.
26. Dengler R, Neyer U, Wohlfarth K, Betting U, Janzik H. Local botulinum toxin in the treatment of spastic foot drop. J Neurol 1992; 239: 375–378.
27. Dunne JW, Heye N, Dunne SL. Treatment of chronic limb spasticity with botulinum toxin A. J Neurol Neurosurg Psychiatry 1995; 58: 232–235.
28. Foster L, Clapp L, Erickson M, Jabbari B. Botulinum toxin A and chronic low back pain: a randomized, double-blind study. Neurology 2001; 56: 1290–1293.
29. Porta M. A comparative trial of botulinum toxin type A and methylprednisolone for the treatment of myofascial pain syndrome and pain from chronic muscle spasm. Pain 2000, 85: 101–105.
30. Rosales RL, Arimura K, Takenaga S, Osame M. Extrafusal and intrafusal muscle effects in experimental botulinum toxin type A injection. Muscle Nerve 1996; 19: 488–496.
31. Brin MF, Aoki KR. Botulinum toxin type A: Pharmacology. In: Mayer NH, Simpson DM, editors. Spasticity: etiology, evaluation, management and the role of botulinum toxin. New York: WE Move; 2002, p. 110–125.
32. Chironna RL, Hecht JS. Subscapularis motor point block for the painful hemiplegic shoulder. Arch Phys Med Rehabil 1990; 71: 428–429.
33. Hecht JS. Subscapular nerve block in the painful hemiplegic shoulder. Arch Phys Med Rehabil 1992; 73: 1036–1039.